Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Neuraminidase
Ligand
BDBM4998
Substrate
BDBM4702
Meas. Tech.
Neuraminidase Inhibition Assay
IC50
120±n/a nM
Citation
 Kim, CU; Lew, W; Williams, MA; Wu, H; Zhang, L; Chen, X; Escarpe, PA; Mendel, DB; Laver, WG; Stevens, RC Structure-activity relationship studies of novel carbocyclic influenza neuraminidase inhibitors. J Med Chem 41:2451-60 (1998) [PubMed] Article
Kim, CU; Lew, W; Williams, MA; Wu, H; Zhang, L; Chen, X; Escarpe, PA; Mendel, DB; Laver, WG; Stevens, RC Structure-activity relationship studies of novel carbocyclic influenza neuraminidase inhibitors. J Med Chem 41:2451-60 (1998) [PubMed] Article More Info.:
Target
Name:
Neuraminidase
Synonyms:
Influenza B Virus Neuraminidase | NA | NRAM_INBLE | Neuraminidase | Neuraminidase B
Type:
Enzyme
Mol. Mass.:
51446.67
Organism:
Influenza B virus (B/Lee/40)
Description:
n/a
Residue:
466
Sequence:
MLPSTVQTLTLLLTSGGVLLSLYVSASLSYLLYSDVLLKFSSTKTTAPTMSLECTNASNAQTVNHSATKEMTFPPPEPEWTYPRLSCQGSTFQKALLISPHRFGEIKGNSAPLIIREPFVACGPKECRHFALTHYAAQPGGYYNGTRKDRNKLRHLVSVKLGKIPTVENSIFHMAAWSGSACHDGREWTYIGVDGPDNDALVKIKYGEAYTDTYHSYAHNILRTQESACNCIGGDCYLMITDGSASGISKCRFLKIREGRIIKEILPTGRVEHTEECTCGFASNKTIECACRDNSYTAKRPFVKLNVETDTAEIRLMCTKTYLDTPRPDDGSIAGPCESNGDKWLGGIKGGFVHQRMASKIGRWYSRTMSKTNRMGMELYVKYDGDPWTDSDALTLSGVMVSIEEPGWYSFGFEIKDKKCDVPCIGIEMVHDGGKDTWHSAATAIYCLMGSGQLLWDTVTGVDMAL
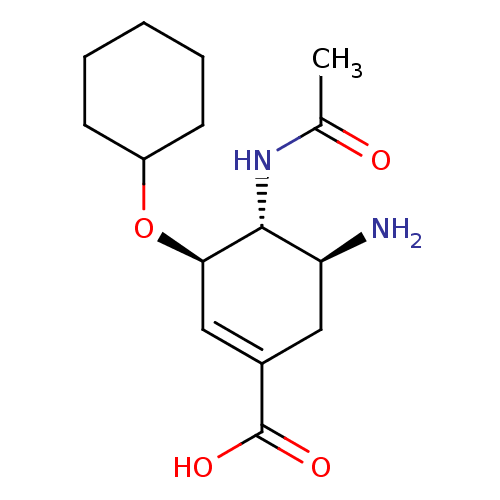
Inhibitor
Name:
BDBM4998
Synonyms:
(3R,4R,5S)-5-amino-3-(cyclohexyloxy)-4-acetamidocyclohex-1-ene-1-carboxylic acid | Carbocyclic Analogue 48 | carbocyclic analogue 8
Type:
Small organic molecule
Emp. Form.:
C15H24N2O4
Mol. Mass.:
296.3621
SMILES:
CC(=O)N[C@@H]1[C@@H](N)CC(=C[C@H]1OC1CCCCC1)C(O)=O |r,c:8|
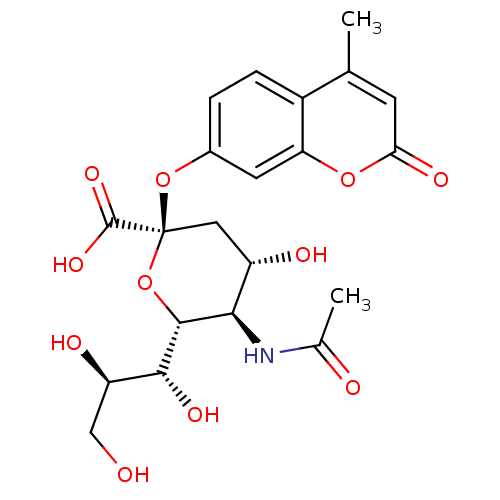
Substrate
Name:
BDBM4702
Synonyms:
(2R,4S,5R,6R)-5-acetamido-4-hydroxy-2-[(4-methyl-2-oxo-2H-chromen-7-yl)oxy]-6-[(1R,2R)-1,2,3-trihydroxypropyl]oxane-2-carboxylic acid | 2 -(4-methylumbelliferyl)-alpha-D-acetylneuraminic acid | neuraminidase substrate
Type:
Small organic molecule
Emp. Form.:
C21H25NO11
Mol. Mass.:
467.4233
SMILES:
CC(=O)N[C@@H]1[C@@H](O)C[C@](Oc2ccc3c(C)cc(=O)oc3c2)(O[C@H]1[C@H](O)[C@H](O)CO)C(O)=O |r|