Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Histone-lysine N-methyltransferase, H3 lysine-79 specific
Ligand
BDBM297391
Substrate
n/a
Meas. Tech.
Assays of Enzymatic Activity
IC50
0.590±n/a nM
Citation
 Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018
Olhava, EJ; Chesworth, R; Pollock, RM; Jin, L Inhibitors of protein methyltransferase DOT1L and methods of use thereof US Patent US10112968 Publication Date 10/30/2018 More Info.:
Target
Name:
Histone-lysine N-methyltransferase, H3 lysine-79 specific
Synonyms:
2.1.1.43 | DOT1-like protein | DOT1-like protein (Dot1L) | DOT1L | DOT1L_HUMAN | H3-K79-HMTase | Histone H3-K79 methyltransferase | Histone H3-K79 methyltransferase (DOT1L) | Histone Methyltransferase DOT1L | Histone-lysine N-methyltransferase, H3 lysine-79 specific (DOT1L) | KIAA1814 | KMT4 | Lysine N-methyltransferase 4
Type:
Protein
Mol. Mass.:
184911.91
Organism:
Homo sapiens (Human)
Description:
Q8TEK3
Residue:
1537
Sequence:
MGEKLELRLKSPVGAEPAVYPWPLPVYDKHHDAAHEIIETIRWVCEEIPDLKLAMENYVLIDYDTKSFESMQRLCDKYNRAIDSIHQLWKGTTQPMKLNTRPSTGLLRHILQQVYNHSVTDPEKLNNYEPFSPEVYGETSFDLVAQMIDEIKMTDDDLFVDLGSGVGQVVLQVAAATNCKHHYGVEKADIPAKYAETMDREFRKWMKWYGKKHAEYTLERGDFLSEEWRERIANTSVIFVNNFAFGPEVDHQLKERFANMKEGGRIVSSKPFAPLNFRINSRNLSDIGTIMRVVELSPLKGSVSWTGKPVSYYLHTIDRTILENYFSSLKNPKLREEQEAARRRQQRESKSNAATPTKGPEGKVAGPADAPMDSGAEEEKAGAATVKKPSPSKARKKKLNKKGRKMAGRKRGRPKKMNTANPERKPKKNQTALDALHAQTVSQTAASSPQDAYRSPHSPFYQLPPSVQRHSPNPLLVAPTPPALQKLLESFKIQYLQFLAYTKTPQYKASLQELLGQEKEKNAQLLGAAQQLLSHCQAQKEEIRRLFQQKLDELGVKALTYNDLIQAQKEISAHNQQLREQSEQLEQDNRALRGQSLQLLKARCEELQLDWATLSLEKLLKEKQALKSQISEKQRHCLELQISIVELEKSQRQQELLQLKSCVPPDDALSLHLRGKGALGRELEPDASRLHLELDCTKFSLPHLSSMSPELSMNGQAAGYELCGVLSRPSSKQNTPQYLASPLDQEVVPCTPSHVGRPRLEKLSGLAAPDYTRLSPAKIVLRRHLSQDHTVPGRPAASELHSRAEHTKENGLPYQSPSVPGSMKLSPQDPRPLSPGALQLAGEKSSEKGLRERAYGSSGELITSLPISIPLSTVQPNKLPVSIPLASVVLPSRAERARSTPSPVLQPRDPSSTLEKQIGANAHGAGSRSLALAPAGFSYAGSVAISGALAGSPASLTPGAEPATLDESSSSGSLFATVGSRSSTPQHPLLLAQPRNSLPASPAHQLSSSPRLGGAAQGPLPEASKGDLPSDSGFSDPESEAKRRIVFTITTGAGSAKQSPSSKHSPLTASARGDCVPSHGQDSRRRGRRKRASAGTPSLSAGVSPKRRALPSVAGLFTQPSGSPLNLNSMVSNINQPLEITAISSPETSLKSSPVPYQDHDQPPVLKKERPLSQTNGAHYSPLTSDEEPGSEDEPSSARIERKIATISLESKSPPKTLENGGGLAGRKPAPAGEPVNSSKWKSTFSPISDIGLAKSADSPLQASSALSQNSLFTFRPALEEPSADAKLAAHPRKGFPGSLSGADGLSPGTNPANGCTFGGGLAADLSLHSFSDGASLPHKGPEAAGLSSPLSFPSQRGKEGSDANPFLSKRQLDGLAGLKGEGSRGKEAGEGGLPLCGPTDKTPLLSGKAAKARDREVDLKNGHNLFISAAAVPPGSLLSGPGLAPAASSAGGAASSAQTHRSFLGPFPPGPQFALGPMSLQANLGSVAGSSVLQSLFSSVPAAAGLVHVSSAATRLTNSHAMGSFSGVAGGTVGGN
Inhibitor
Name:
BDBM297391
Synonyms:
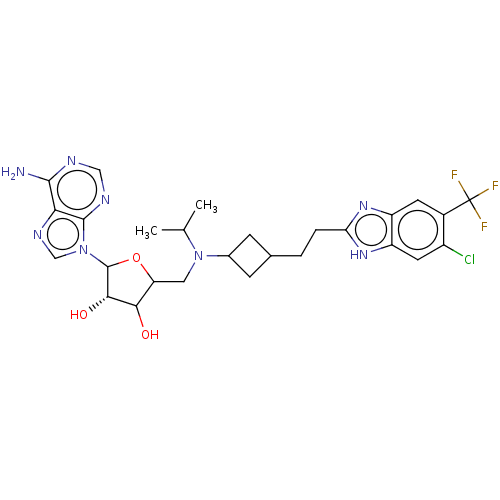
(2R,3R,4S,5R)-2-(6-amino-9H-purin-9-yl)-5-((((1r,3S)-3-(2-(5-chloro-6-(trifluoromethyl)-1H-benzo[d]imidazol-2-yl)ethyl)cyclobutyl)(isopropyl)amino)methyl)tetrahydrofuran-3,4-diol | US10112968, Compound A5
Type:
Small organic molecule
Emp. Form.:
C27H32ClF3N8O3
Mol. Mass.:
609.043
SMILES:
CC(C)N(CC1OC([C@@H](O)C1O)n1cnc2c(N)ncnc12)C1CC(CCc2nc3cc(c(Cl)cc3[nH]2)C(F)(F)F)C1 |r,wD:8.8,(-11.05,-1.3,;-9.51,-1.3,;-8.74,.03,;-8.74,-2.64,;-9.51,-3.97,;-11.05,-3.97,;-11.96,-2.72,;-13.42,-3.2,;-13.42,-4.74,;-14.67,-5.65,;-11.96,-5.22,;-11.48,-6.68,;-14.67,-2.3,;-14.67,-.76,;-16.13,-.28,;-17.04,-1.53,;-18.57,-1.69,;-19.47,-.44,;-19.19,-3.09,;-18.29,-4.34,;-16.76,-4.18,;-16.13,-2.77,;-7.2,-2.64,;-6.11,-1.55,;-5.02,-2.64,;-3.48,-2.64,;-2.71,-1.3,;-1.17,-1.3,;-.27,-2.55,;1.2,-2.07,;2.53,-2.84,;3.86,-2.07,;3.86,-.53,;5.2,.24,;2.53,.24,;1.2,-.53,;-.27,-.06,;5.2,-2.84,;5.2,-4.38,;6.53,-2.07,;6.53,-3.61,;-6.11,-3.73,)|