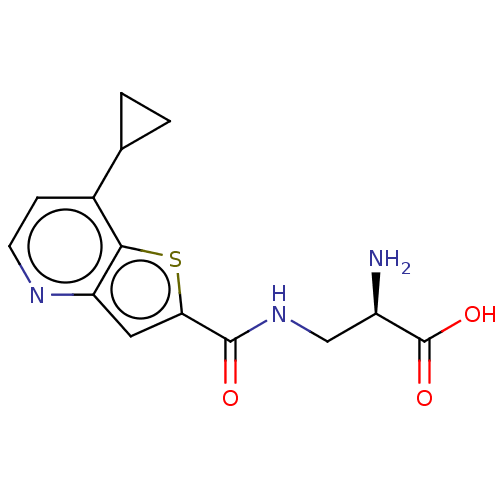
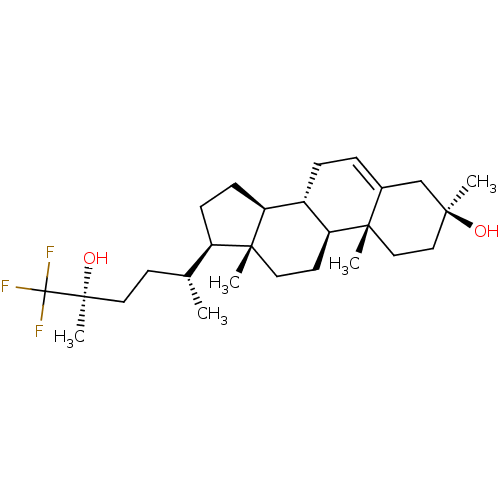
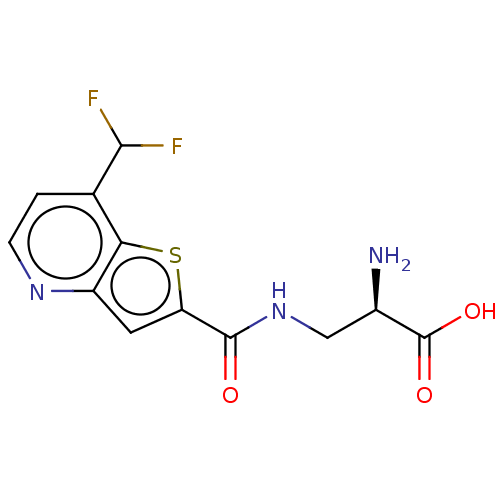
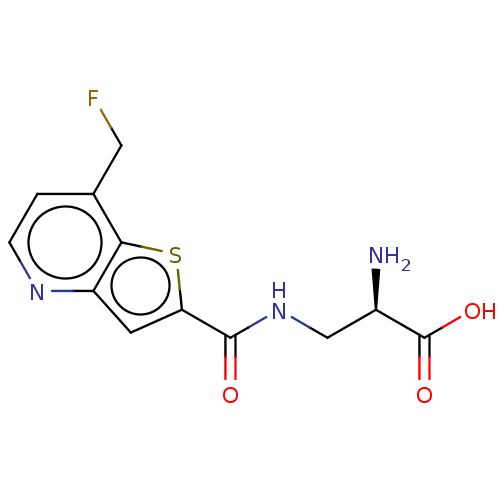
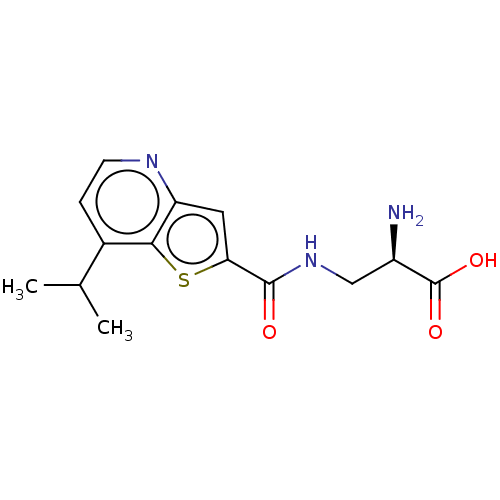
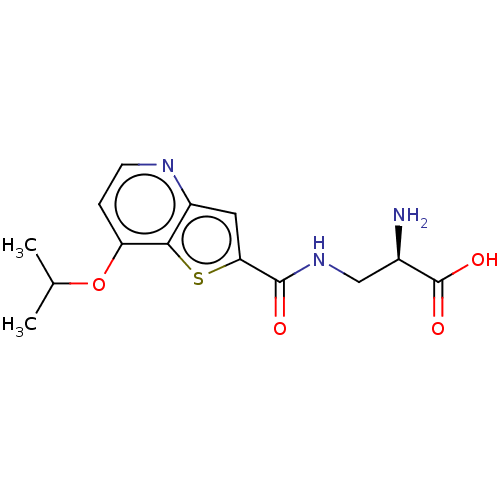
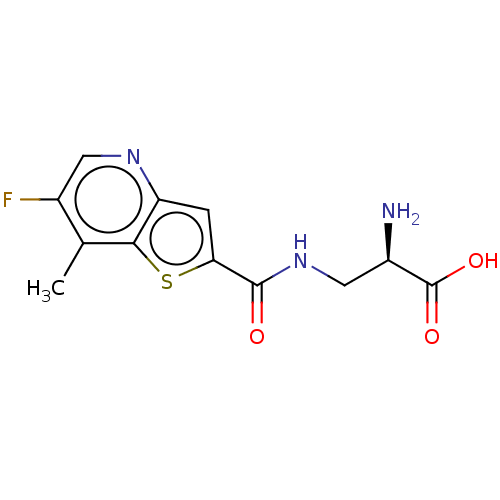
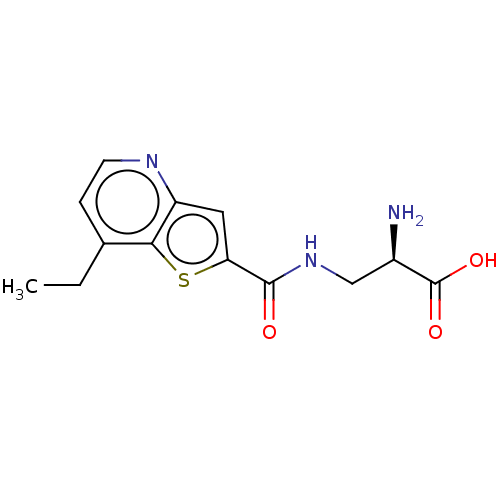
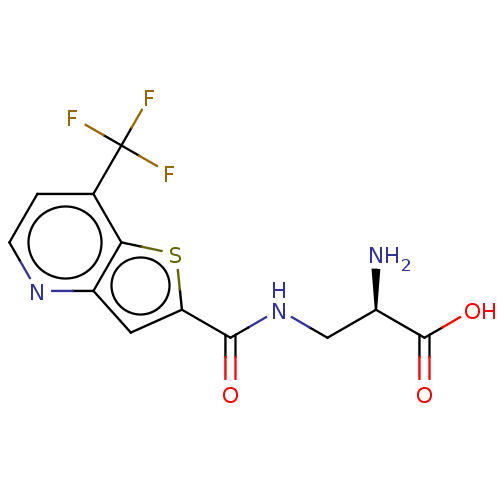
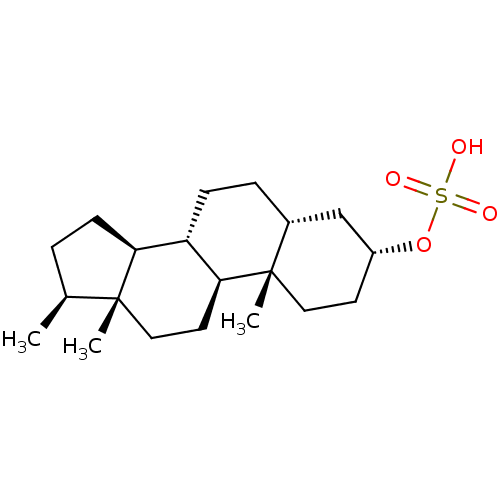
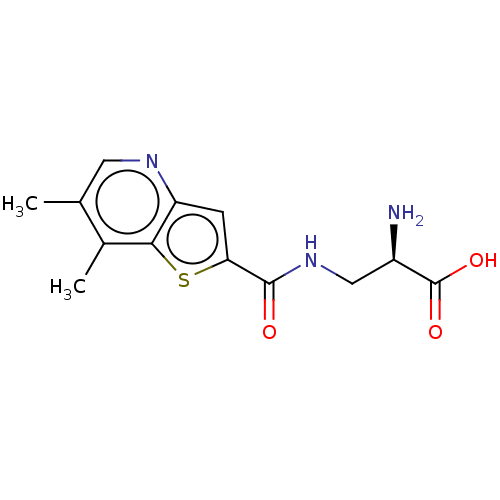
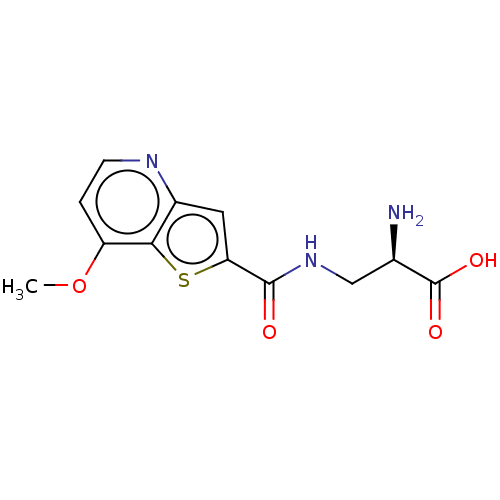
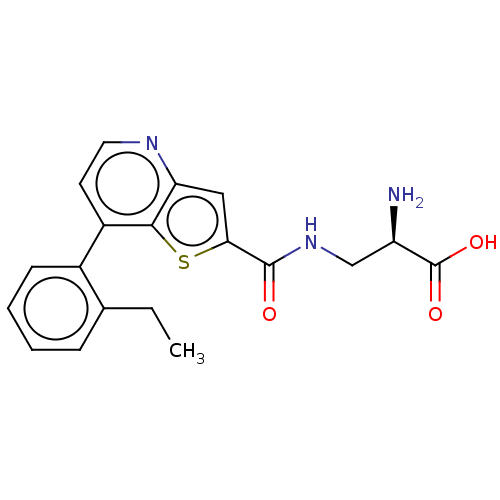
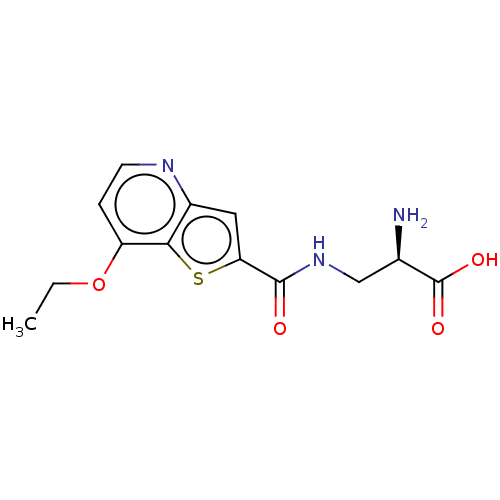
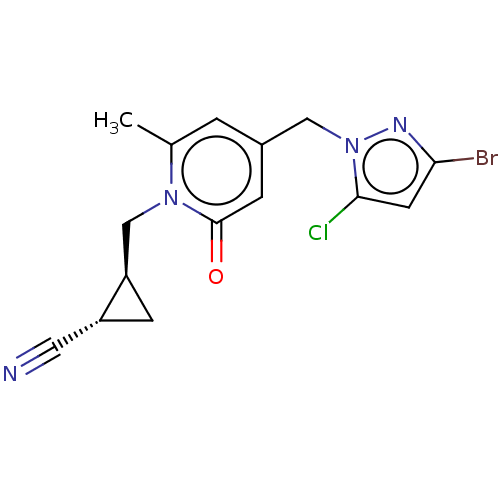
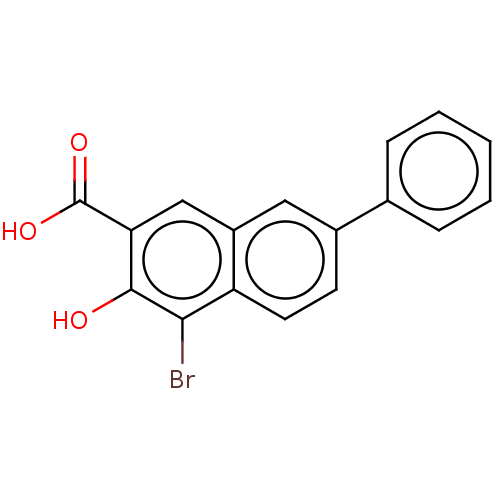
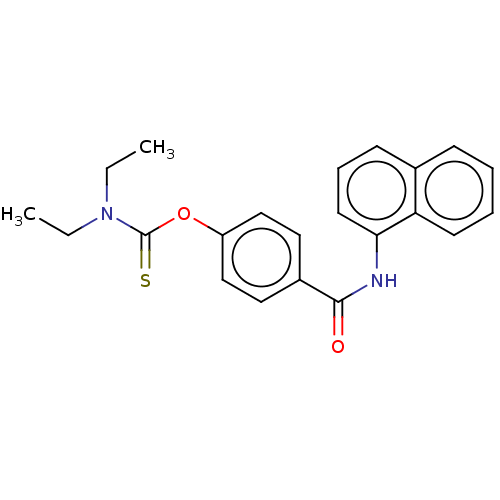
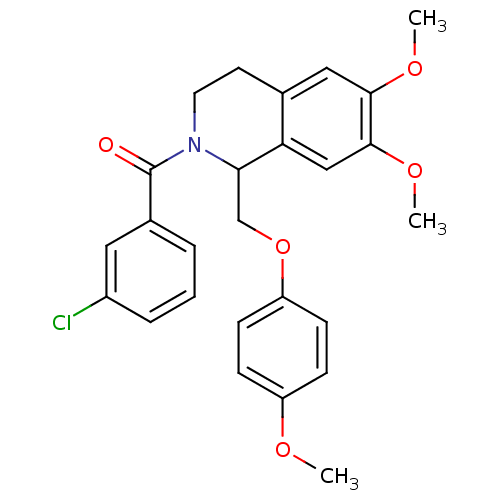
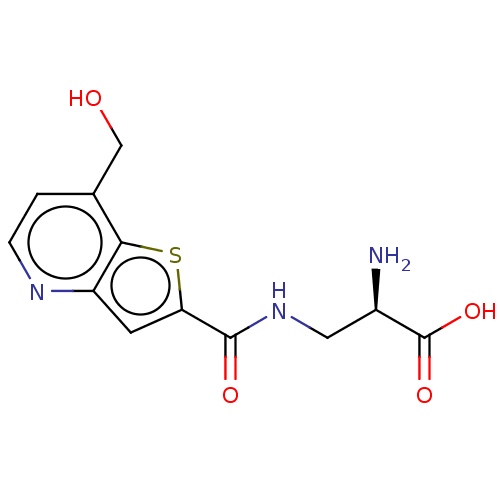
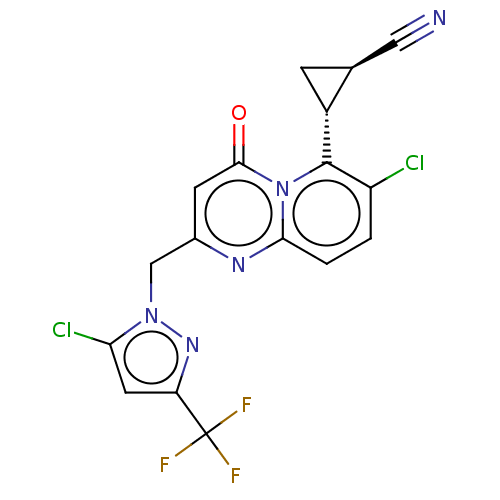
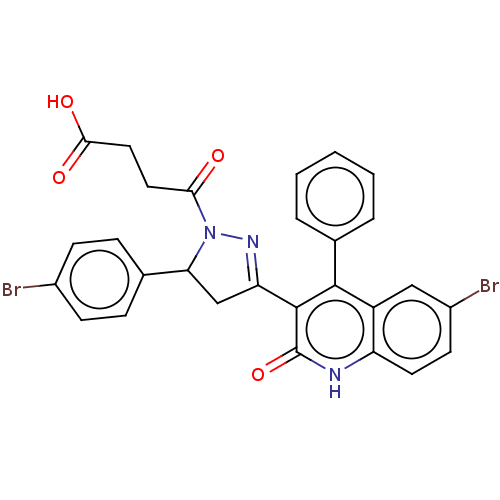
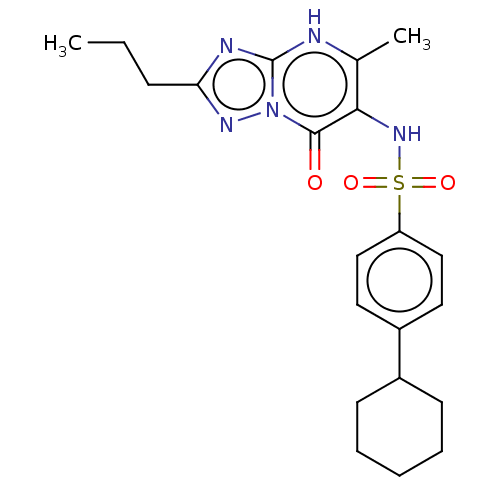
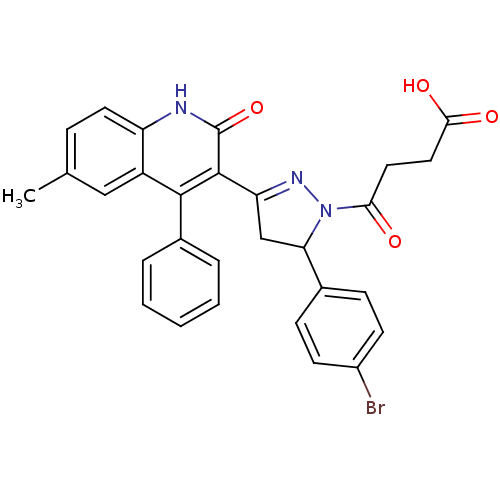
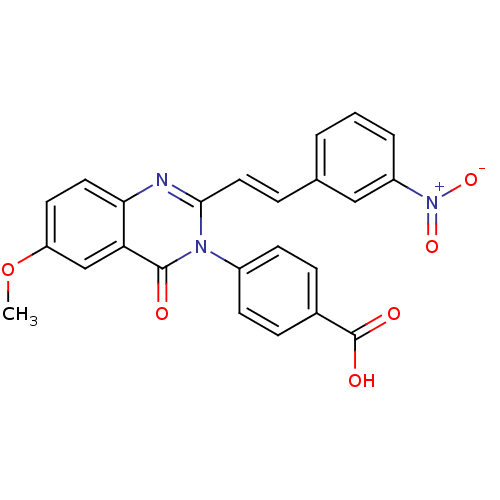
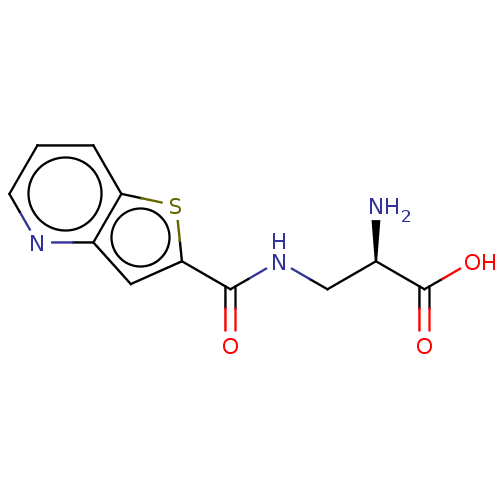
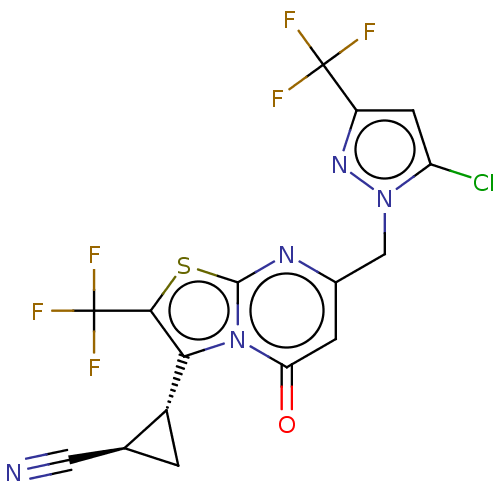
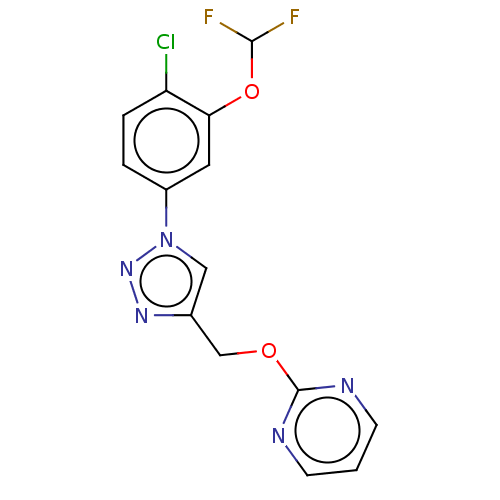
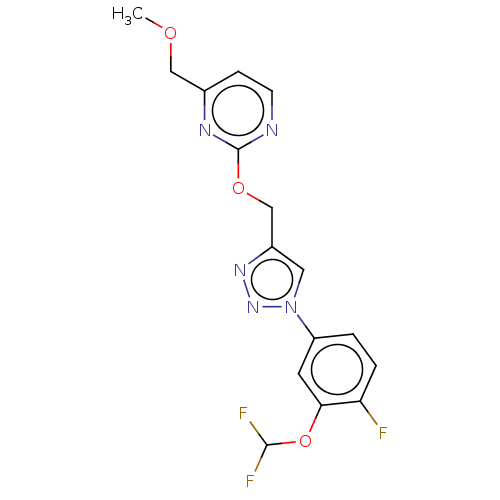
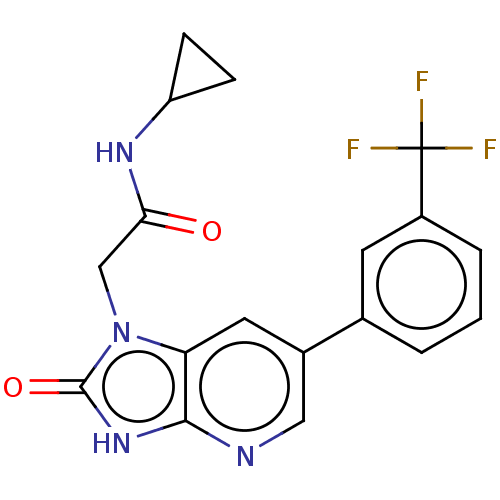
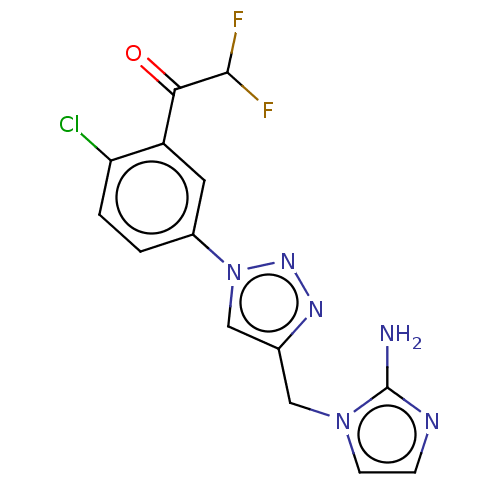
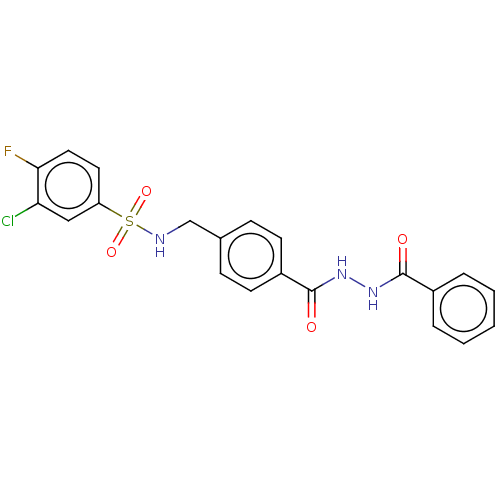
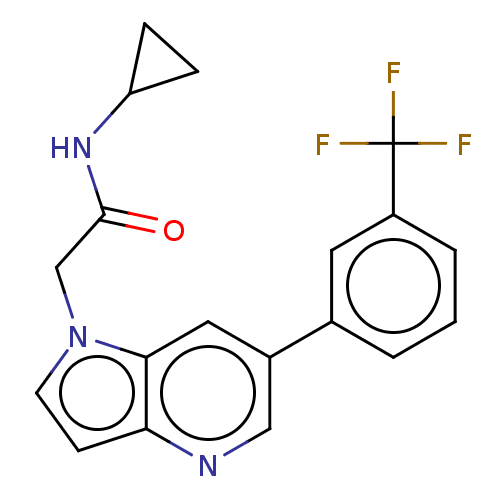
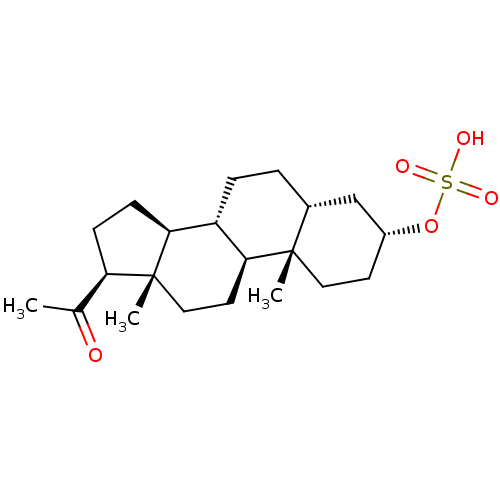
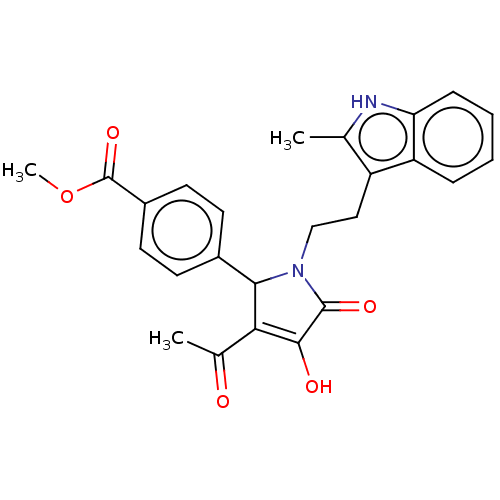
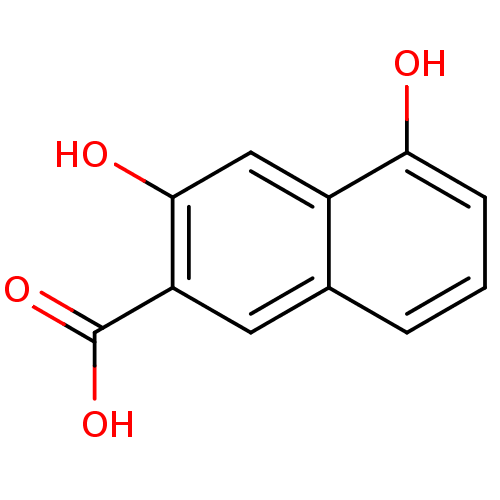
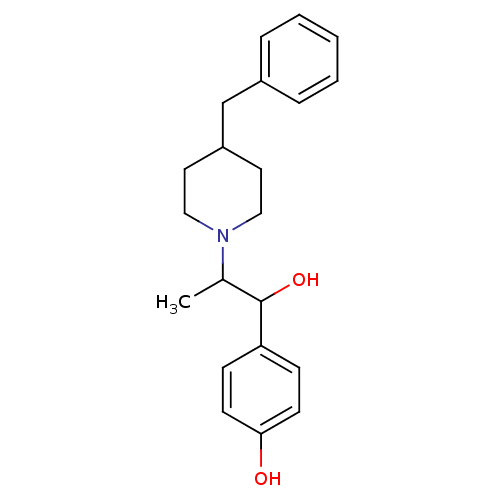
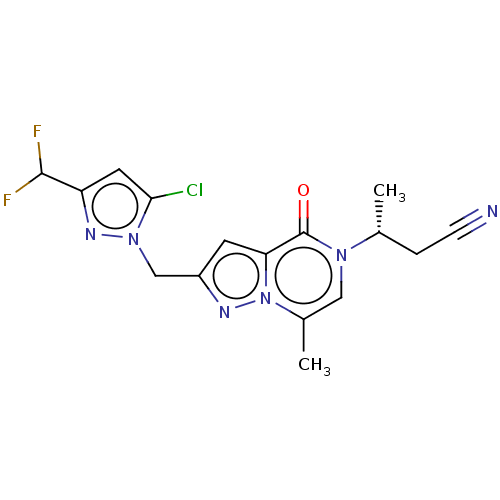
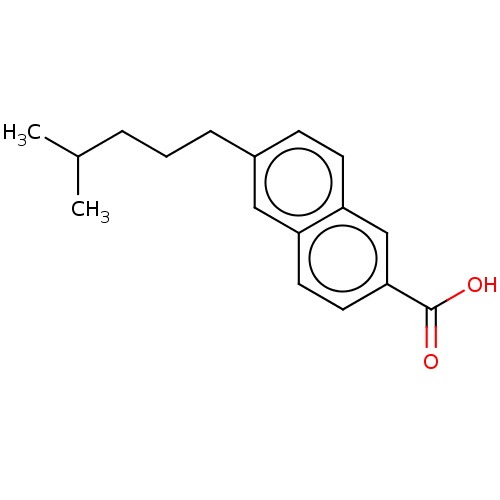
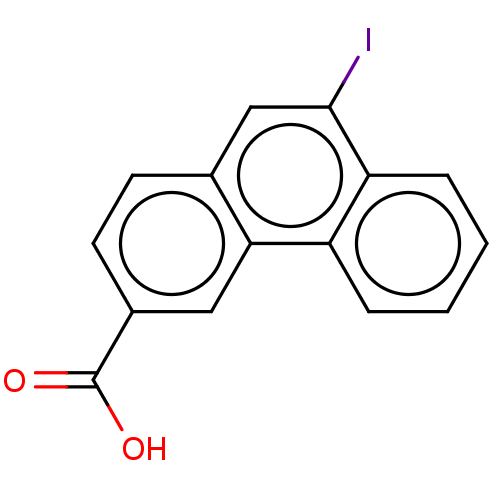
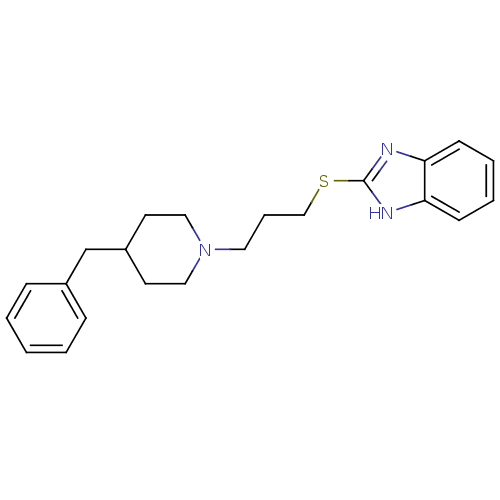
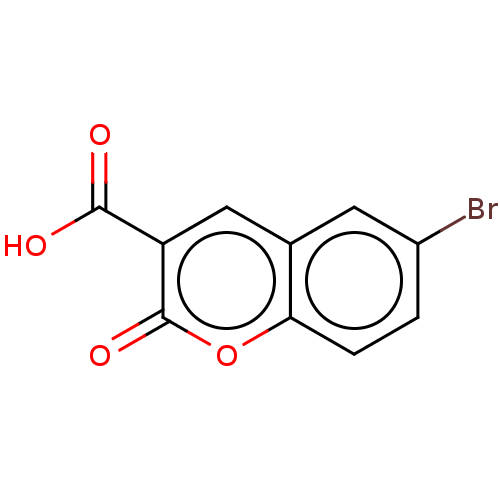
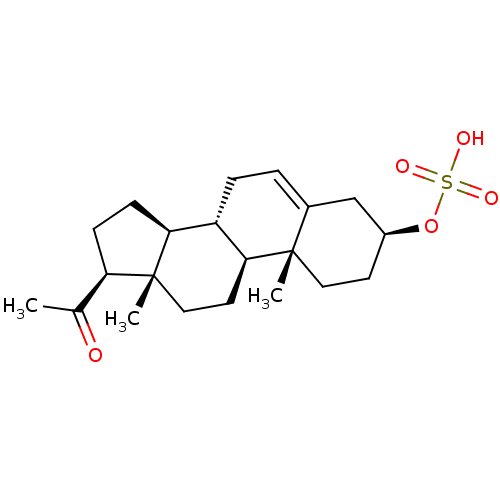
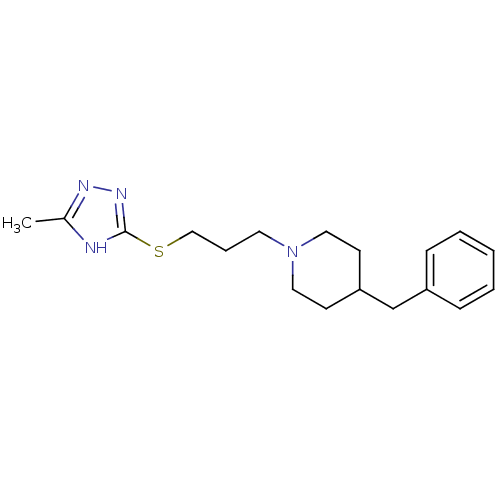
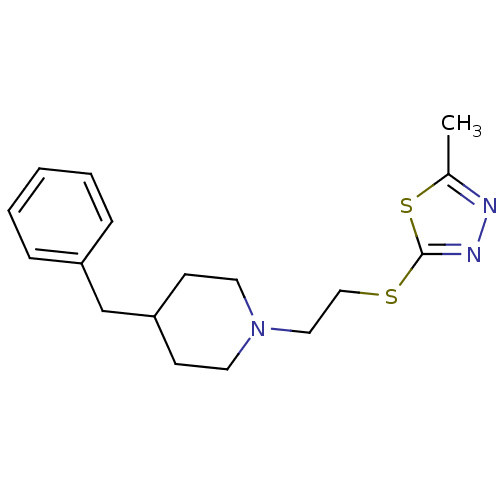
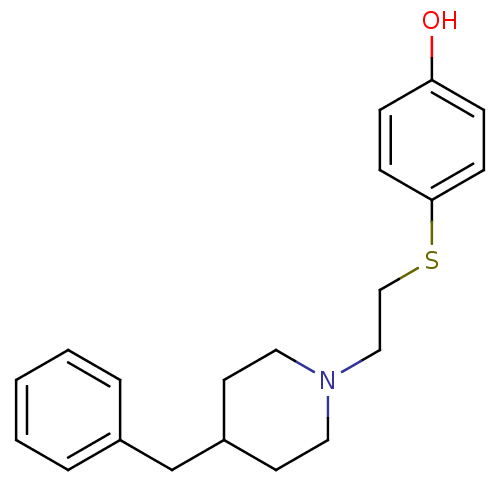
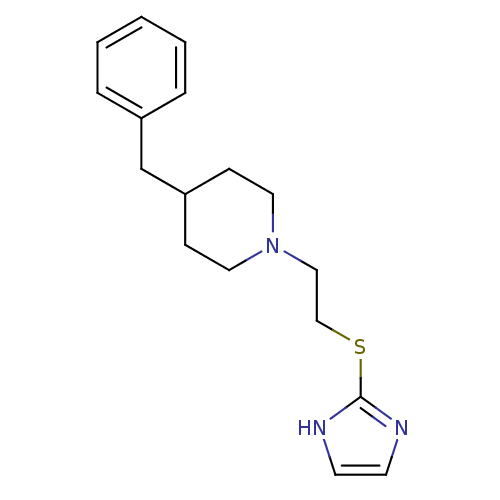
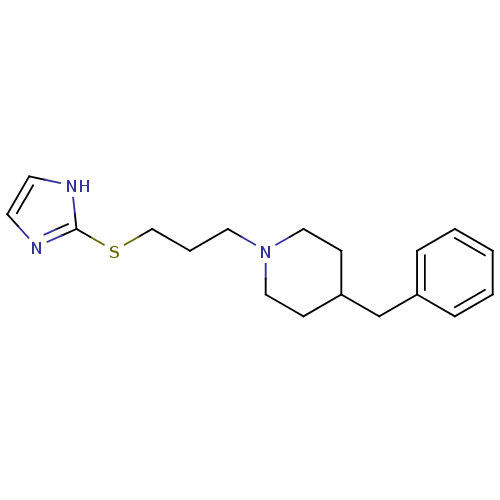
Affinity DataKi: 63nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 96nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataEC50: 150nMAssay Description:Positive allosteric modulation of recombinant human GluN1/GluN2C receptor stably expressed in HEK293 cells assessed as increase in glycine/L-glutamat...More data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 180nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 220nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 220nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 260nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 270nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 360nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibition of recombinant GluN1/GluN2C receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of glutamate-evoked current by whol...More data for this Ligand-Target Pair
Affinity DataKi: 490nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 690nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 860nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataKi: 870nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataEC50: 1.90E+3nMAssay Description:Positive allosteric modulator activity at GluN2C NMDA receptor (unknown origin) expressed in CHO cells co-expressing GluN1a in the presence of L-glut...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataEC50: 2.70E+3nMAssay Description:Positive allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as increase in glycine-induced channe...More data for this Ligand-Target Pair
Affinity DataKi: 3.90E+3nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataEC50: 4.70E+3nMAssay Description:Positive allosteric modulation of GluN1/GluN2C receptor (unknown origin) assessed as increase in glutamate-induced calcium flux measured at time inte...More data for this Ligand-Target Pair
Affinity DataIC50: 5.40E+3nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataEC50: 6.60E+3nMAssay Description:Positive allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as increase in glycine-induced channe...More data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataIC50: 7.10E+3nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataKi: 7.40E+3nMAssay Description:To determine the affinity of the compounds of the present invention a SPA is used. The assay is run in a 384-plate format (OptiPlate-384) where each ...More data for this Ligand-Target Pair
Affinity DataEC50: 7.40E+3nMAssay Description:Positive allosteric modulation of GluN1/GluN2C NMDAR (unknown origin) expressed in Dox-inducible cells by BD calcium indicator dye based-fluorescence...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human GluN2C receptor by FLIPR assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human GluN2C receptor by FLIPR assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Negative allosteric modulation of recombinant human GluN1a/GluN2C expressed in CHO-T-REx cells assessed as inhibition of glutamate/glycine-induced re...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human GluN2C receptor by FLIPR assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Negative allosteric modulation of human GluN2C receptor expressed in xenopus laevis oocytes assessed as reduction in 3 uM glycine-induced channel cur...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Negative allosteric modulation of recombinant human GluN1a/GluN2C expressed in CHO-T-REx cells assessed as inhibition of glutamate/glycine-induced re...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataEC50: 1.30E+4nMAssay Description:Positive allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as increase in glycine-induced channe...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataIC50: 2.91E+4nMAssay Description:Negative allosteric modulation of human GluN2C receptor expressed in xenopus laevis oocytes assessed as reduction in 3 uM glycine-induced channel cur...More data for this Ligand-Target Pair
Affinity DataEC50: >3.00E+4nMAssay Description:Positive allosteric modulator activity at GluN1a/GluN2C (unknown origin) expressed in CHO cells in presence of glutamate by Ca2+ influx assayMore data for this Ligand-Target Pair
Affinity DataEC50: 3.70E+4nMAssay Description:Positive allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as increase in glycine-induced channe...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataIC50: 5.10E+4nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 6.80E+4nMAssay Description:Negative allosteric modulation of GluN1a/GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glutamate/glyc...More data for this Ligand-Target Pair
Affinity DataIC50: 6.80E+4nMAssay Description:Negative allosteric modulation of GluN2C receptor (unknown origin) expressed in xenopus laevis oocytes assessed as reduction in glycine-induced chann...More data for this Ligand-Target Pair
Affinity DataEC50: 8.30E+4nMAssay Description:Positive allosteric modulation of EGFP-fused human GluN2C receptor expressed in HEK293T cells assessed as increase in glycine-induced channel current...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Antagonistic activity against NR1A/2C receptor in frog oocytesMore data for this Ligand-Target Pair
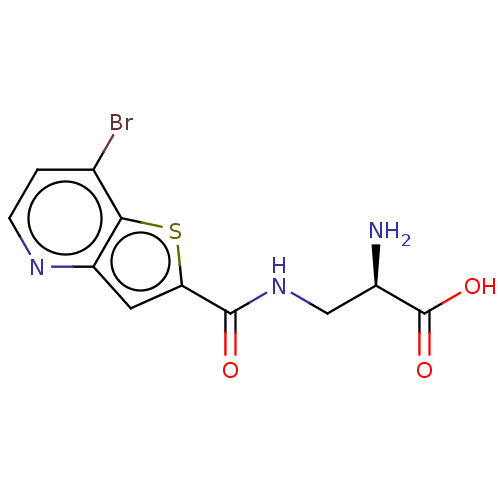
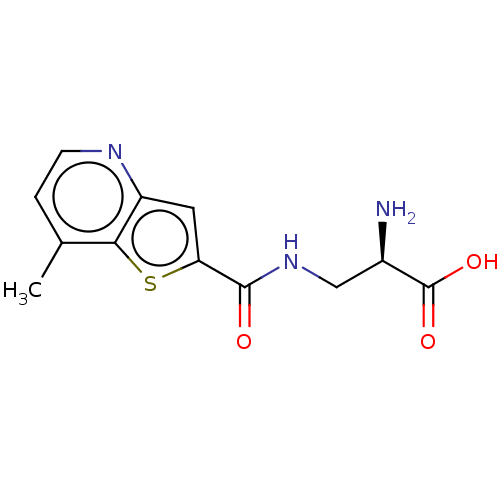
 3D Structure (crystal)
3D Structure (crystal)