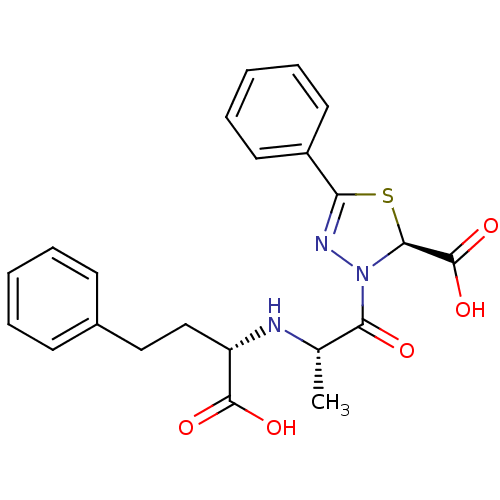
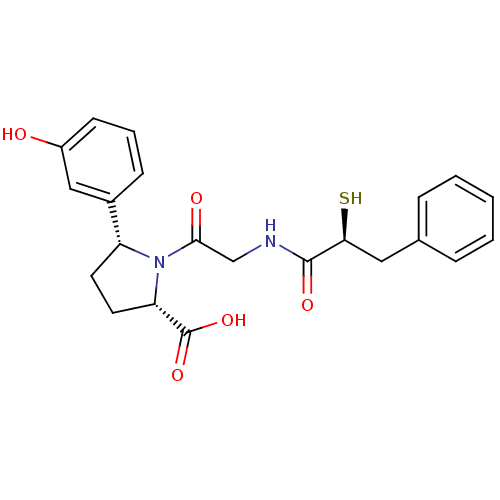
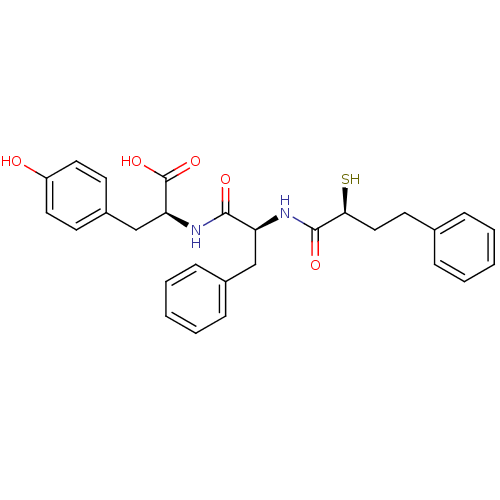
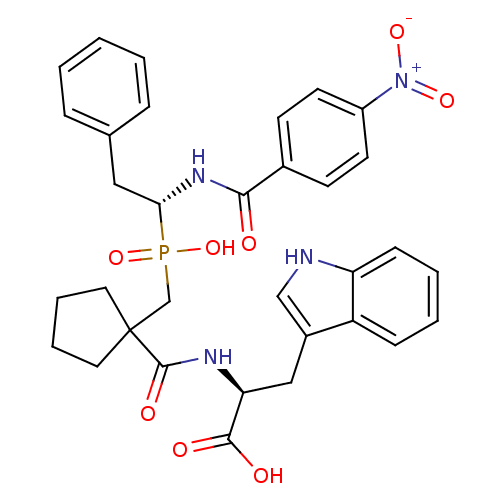
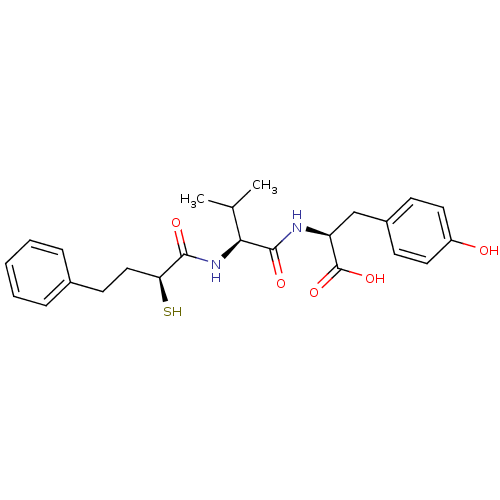
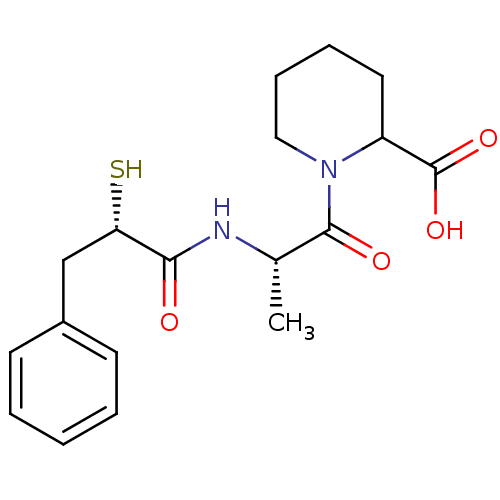
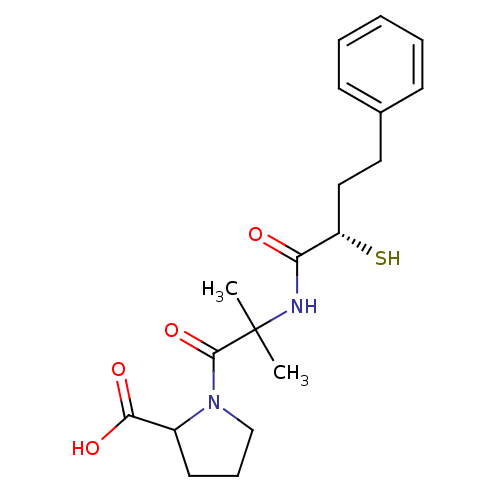
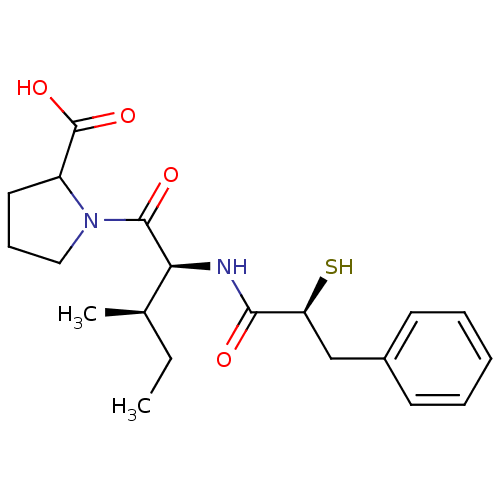
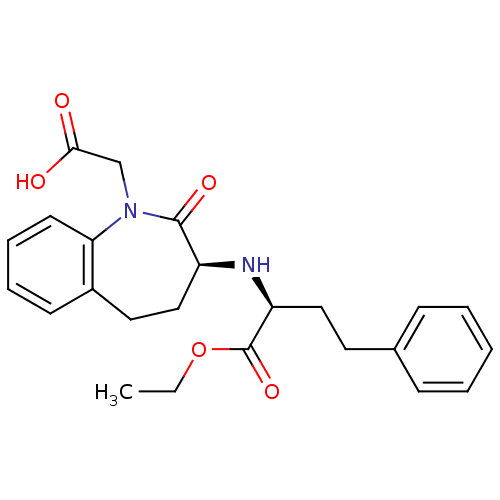
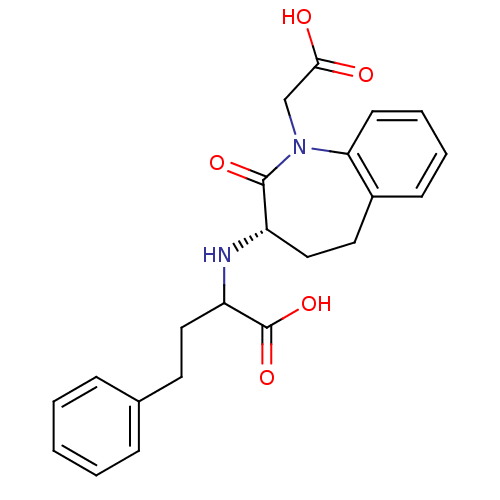
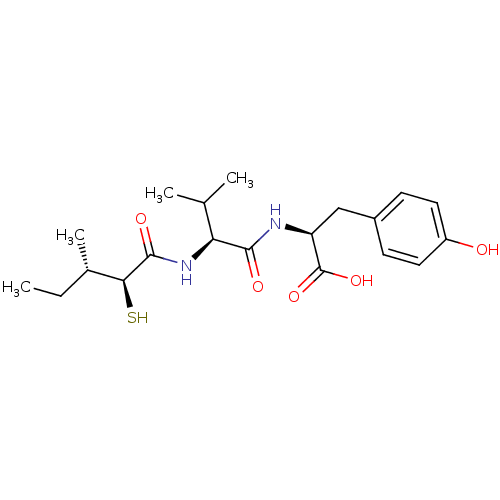
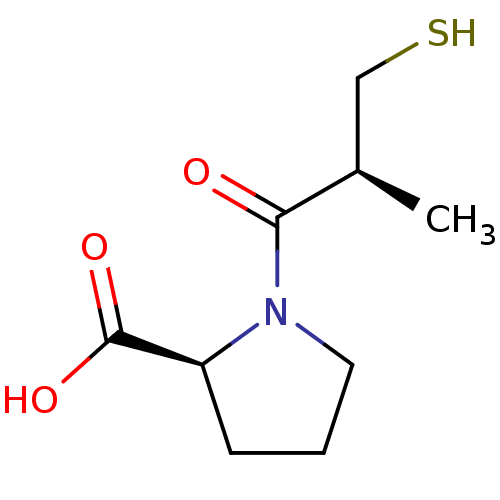
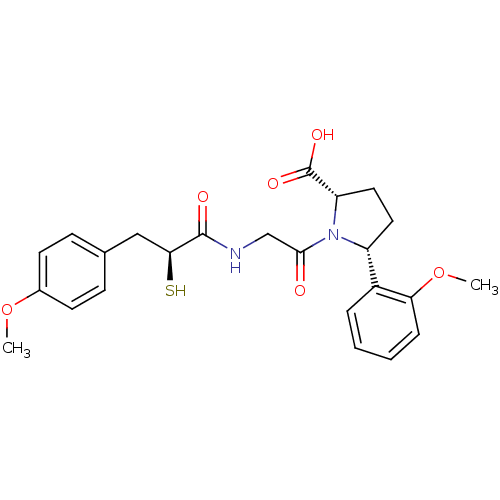
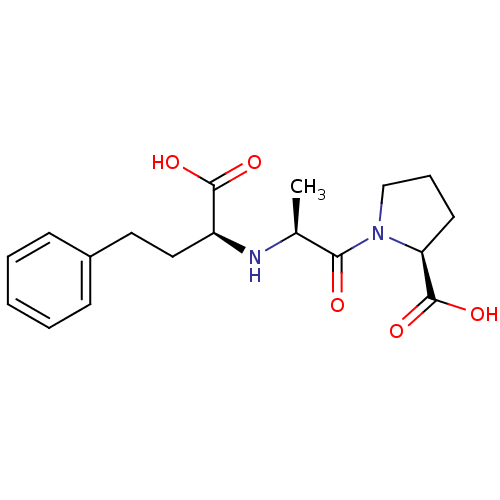
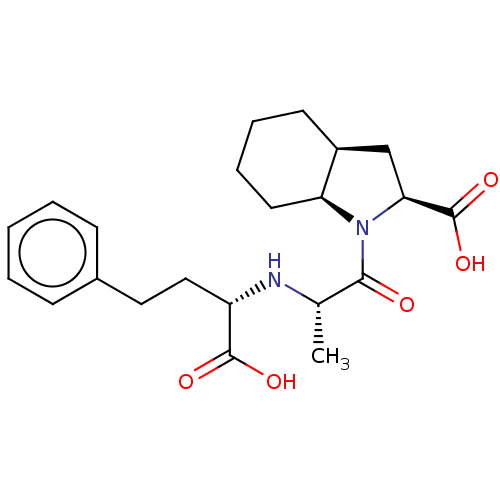
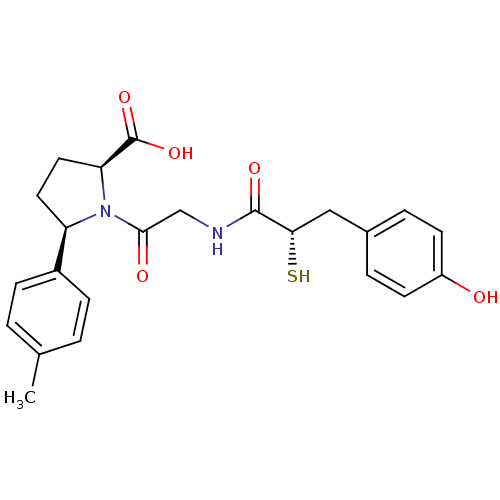
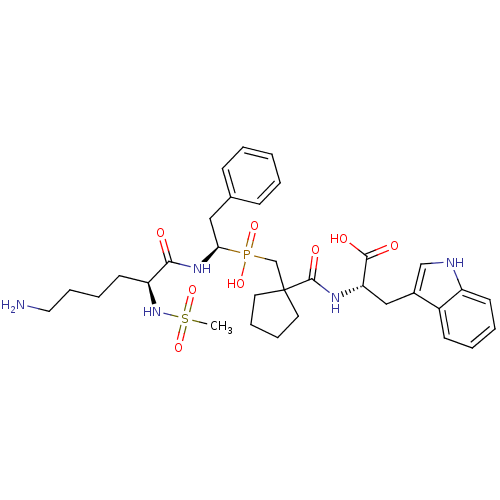
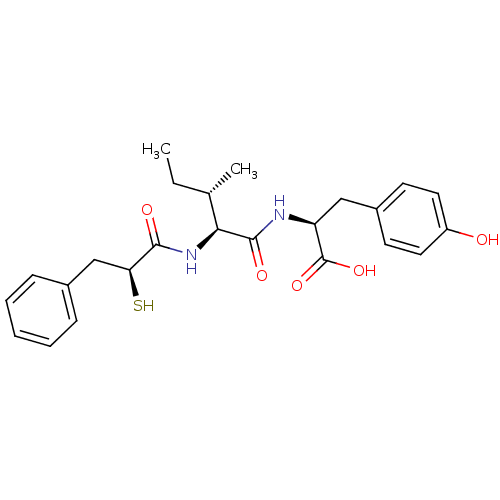
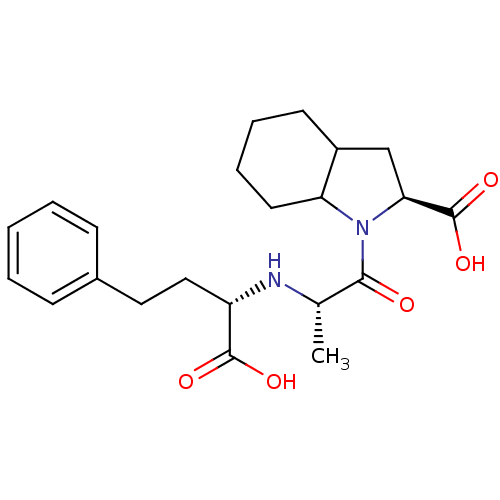
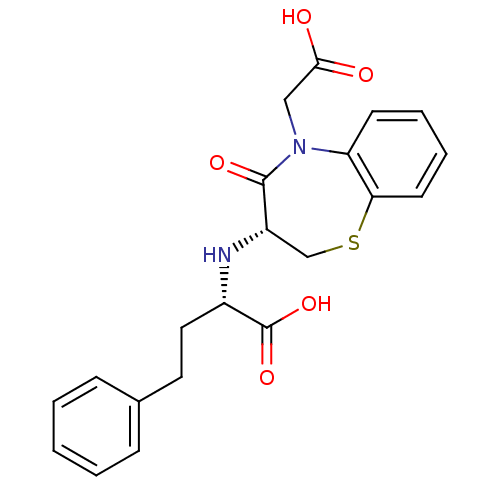
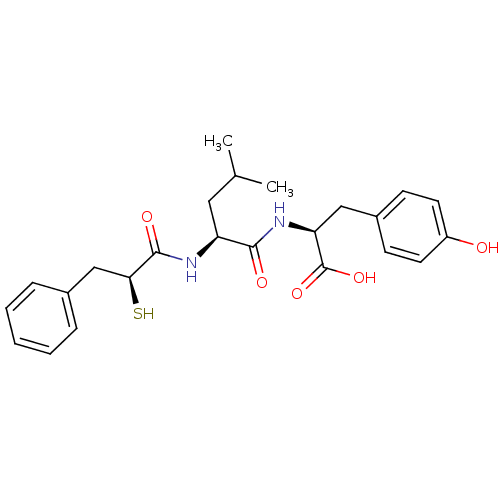
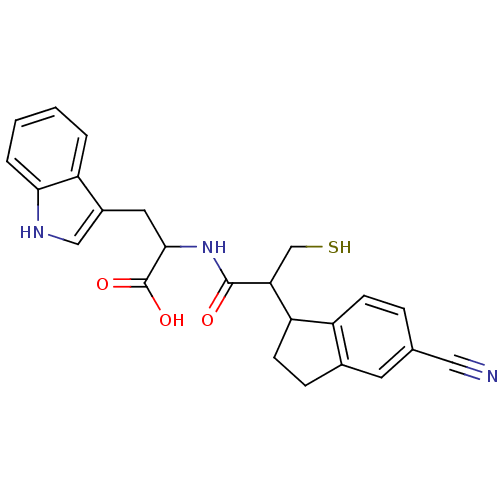
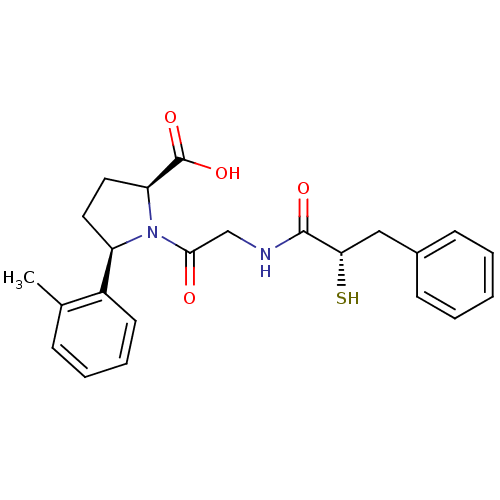
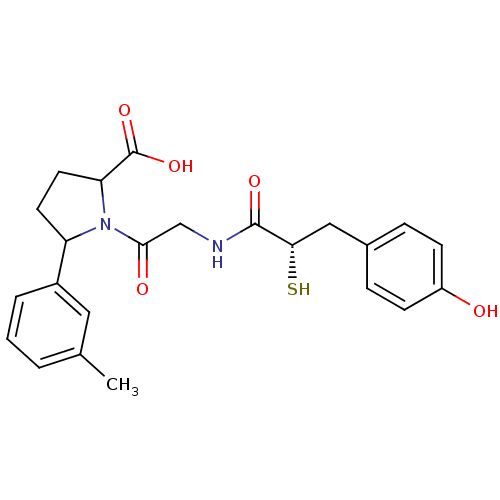
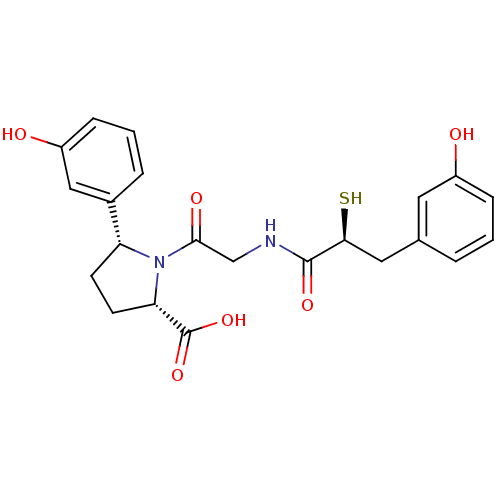
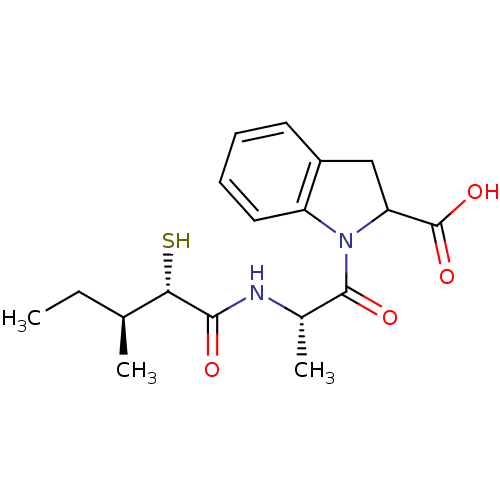
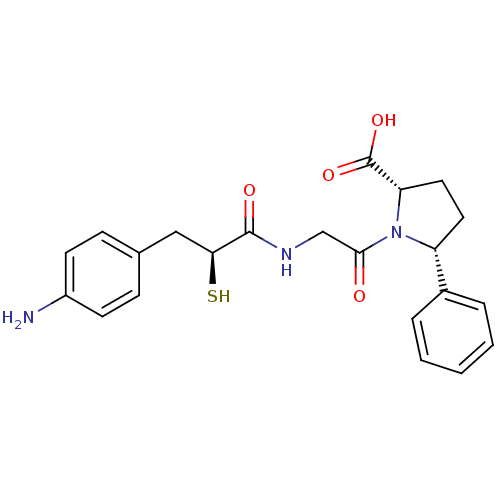
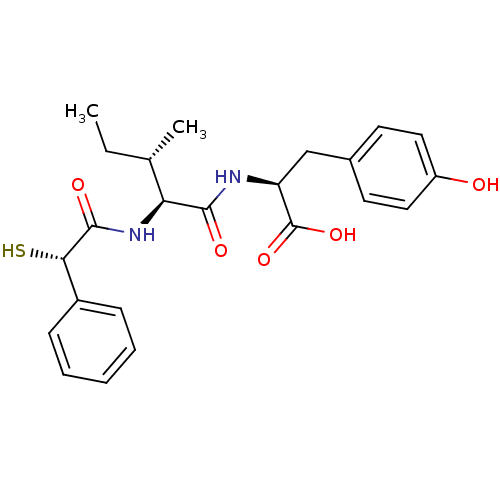
Affinity DataIC50: 0.260nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.420nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.460nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.510nMAssay Description:Compound was tested for inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.580nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Compound was tested for inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Compound was tested for inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.850nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:Compound tested in vivo for inhibition of Angiotensin I converting enzyme in ratMore data for this Ligand-Target Pair
Affinity DataIC50: 0.950nMAssay Description:Compound was tested for inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Compound was tested for inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Compound was measured for the inhibition of Angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:In vitro inhibitory activity against Angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:Inhibition of Angiotensin I converting enzyme in ratMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:NEP was preincubated in black 96-well microplates with or without increasing concentrations of inhibitors. DGPA was added, and the reaction was stopp...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate.More data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMAssay Description:Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate.More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Compound was measured for the inhibition of Angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate.More data for this Ligand-Target Pair
Affinity DataIC50: 2.90nMAssay Description:Inhibition of Angiotensin I converting enzyme in ratMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:In vitro inhibitory activity against rat serum angiotensin I converting enzyme using hippuryl-glycyl-glycine as substrateMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:In vitro inhibition of Angiotensin I converting enzyme.More data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate.More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Inhibition of Wistar rat plasma angiotensin 1-converting enzyme using H-hippuryl-His-Leu-OH as substrate after 20 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:In vitro inhibition of rat angiotensin I converting enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:Inhibitory activity against angiotensin I converting enzymeMore data for this Ligand-Target Pair
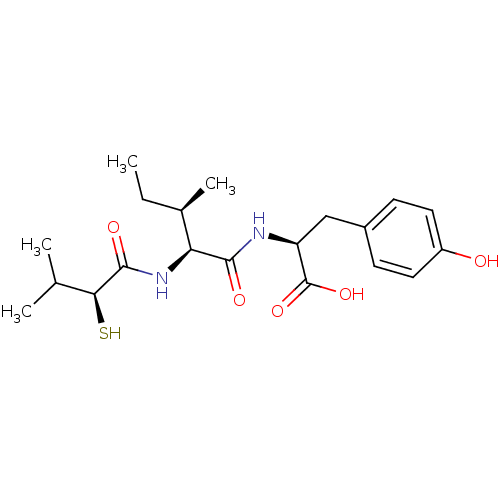
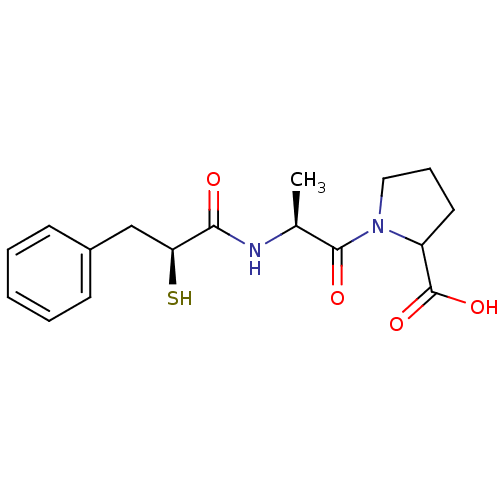
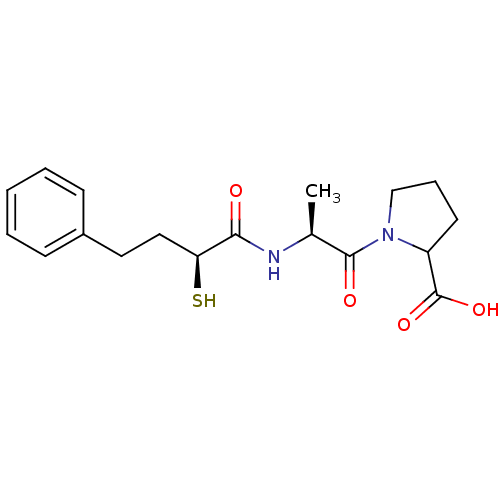
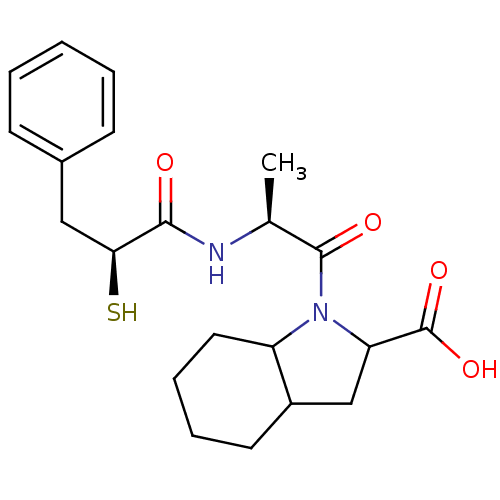
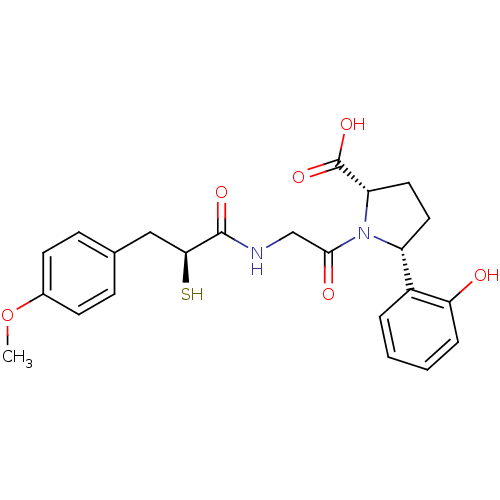

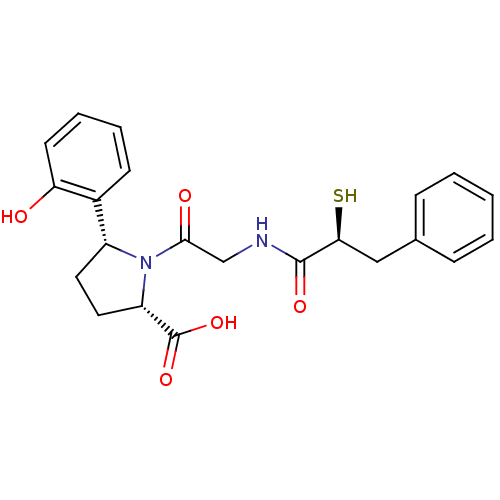
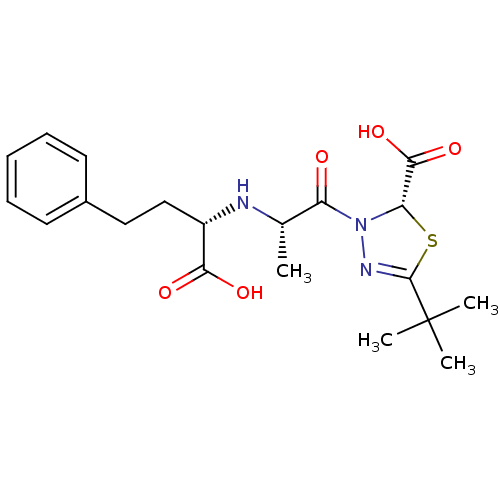
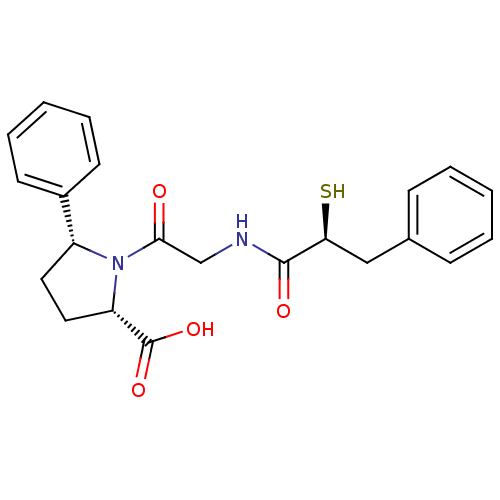
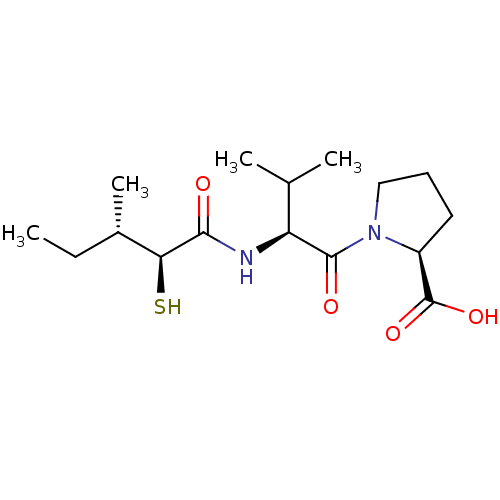
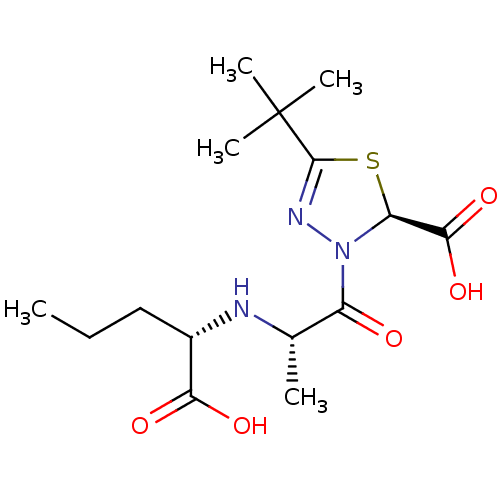
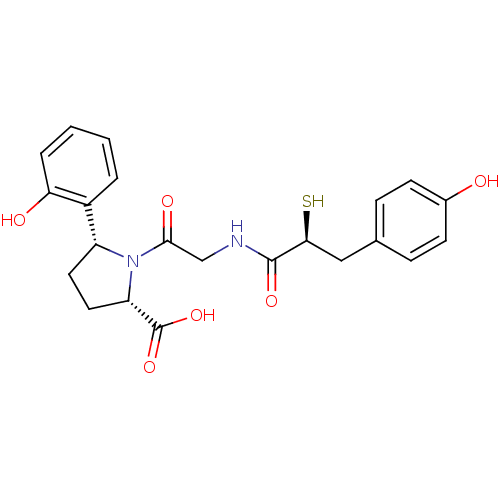
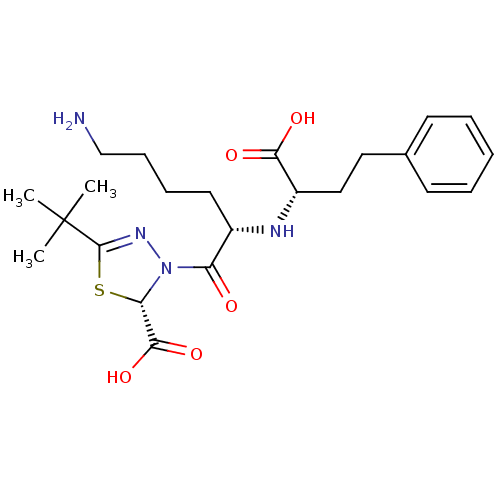
 3D Structure (crystal)
3D Structure (crystal)