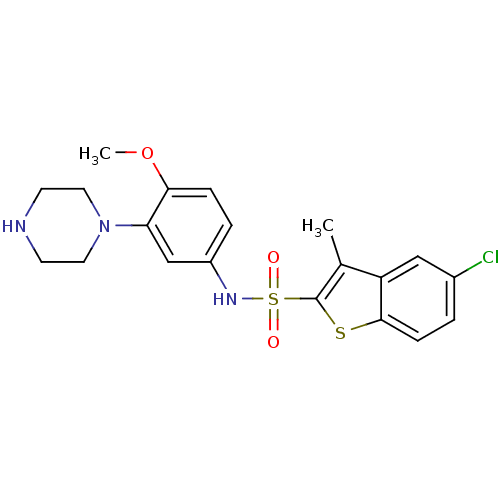
BDBM28583 5-chloro-N-[4-methoxy-3-(piperazin-1-yl)phenyl]-3-methyl-1-benzothiophene-2-sulfonamide hydrochloride::CHEMBL431298::SB 271046::SB-271046
SMILES COc1ccc(NS(=O)(=O)c2sc3ccc(Cl)cc3c2C)cc1N1CCNCC1
InChI Key InChIKey=LOCQRDBFWSXQQI-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 48 hits for monomerid = 28583
Found 48 hits for monomerid = 28583
Affinity DataKi: 0.631nMAssay Description:Displacement of [3H]-LSD from human 5-HT6R expressed in human HeLa cells after 1 hr by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 0.660nM ΔG°: -12.4kcal/molepH: 7.5 T: 2°CAssay Description:Competition experiments were performed in the presence radioligand with membrane protein (obtained from cells expressing the receptor) and test compo...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Inhibition of [3H]-LSD binding to human 5-hydroxytryptamine 6 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Binding affinity against 5 Hydroxy tryptamine 6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Binding affinity towards human 5-hydroxytryptamine 6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Displacement of [3H]-LSD from human cloned 5HT6 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Binding affinity for human 5-hydroxytryptamine 6 receptorChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Antagonist activity at human recombinant 5HT6 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]-LSD from human 5-hydroxytryptamine 6 receptor expressed in HEK 293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]LSD from human 5-hydroxytryptamine 6 receptor expressed in HeLa cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Binding affinity against human 5-hydroxytryptamine 6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 6 receptor in HeLa cells, using [3H]LSD as radioligand; n=3More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]LSD from human 5HT6 receptor expressed in HeLa cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:Displacement of [3H]LSD from human 5-HT6 receptor expressed in HEK293 cell membranes incubated for 60 mins by radioligand binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Displacement of [3H]LSD from human 5-HT6 receptor expressed in hamster BHK cells after 60 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Displacement of [3H]-LSD from human 5HT6 receptor expressed in BHK cell membrane measured after 60 mins by scintillation counter methodMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]LSD from human 5HT6 receptor expressed in HEK293 cells by liquid scintillation countingMore data for this Ligand-Target Pair
TargetD(1A) dopamine receptor(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 120nMAssay Description:Displacement of [3H]SCH-23390 from human cloned dopamine D1 receptorMore data for this Ligand-Target Pair
TargetD(4) dopamine receptor(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 130nMAssay Description:Displacement of [3H]spiperone from human cloned dopamine D4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 251nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 1D receptor in CHO cells, using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 260nMAssay Description:Displacement of [3H]-LSD from human cloned 5HT7 receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 1A(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 340nMAssay Description:Displacement of [3H]-8-OH-DPAT from human cloned 5HT1A receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 1A(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 398nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 1A receptor in HEK293 cells, using [3H]8-OH-DPAT as radioli...More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 460nMAssay Description:Displacement of [3H]spiperone from human cloned dopamine D2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 501nMAssay Description:The compound was tested for the binding affinity towards human cloned Dopamine receptor D3 in CHO cells, using [125I]iodosulpride as radioligandMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 530nMAssay Description:Displacement of [3H]mesulergine from human cloned 5HT2C receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 770nMAssay Description:Displacement of [3H]5-HT from 5-HT2C receptor (unknown origin) expressed in hamster AV12 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 794nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 1B receptor in CHO cells, using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: <1.00E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 1F receptor in CHO cells, using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 1.33E+3nMAssay Description:Displacement of [3H]spiperone from human cloned dopamine D3 receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 1.74E+3nMAssay Description:Displacement of [3H]ketanserin from human cloned 5HT2A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:The compound was tested for the binding affinity towards Alpha-1B adrenergic receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 2C receptor in HEK293 cells, using [3H]5-mesulergine as rad...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: <2.51E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 2A receptor in HEK293 cells, using [3H]ketanserin as radiol...More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Korea Research Institute of Chemical Technology
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned Dopamine receptor D2 in CHO cells, using [125I]iodosulpride as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 3.98E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 7 receptor in HEK293 cells, using [3H]5-CT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 3.98E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 4 receptor in HeLa cells, using [3H]LSD as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: <3.98E+3nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 2B receptor in HEK293 cells, using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: <1.00E+4nMAssay Description:The compound was tested for the binding affinity towards human cloned 5-hydroxytryptamine 1E receptor in CHO cells, using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Affinity DataIC50: 6.60E+4nMAssay Description:Inhibition of human cytochrome P450 1A2 activity as caffeine N3 demethylation (500 uM)More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+4nMAssay Description:Inhibition of human cytochrome P450 2C19 as S-mephenytoin-4-hydroxylation (100 uM)More data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of human cytochrome P450 3A4 as total cyclosporin oxidation (1 uM)More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of human recombinant 5HT6 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+4nMAssay Description:Inhibition of human cytochrome P450 2D6 as bufuralol 1''-hydroxylation (10 uM)More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory potential of human cytochrome P450 2C9 as tolbutamide methylhydroxylation (100 uM)More data for this Ligand-Target Pair