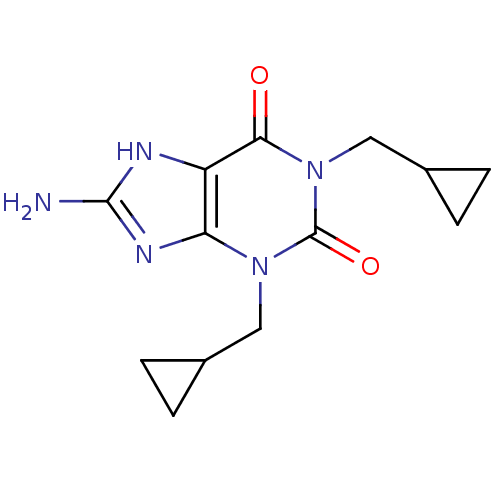
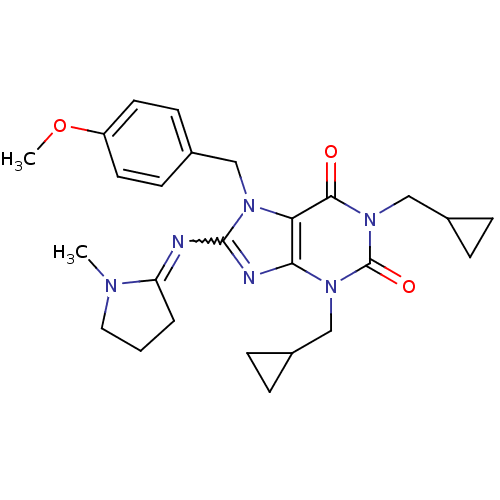
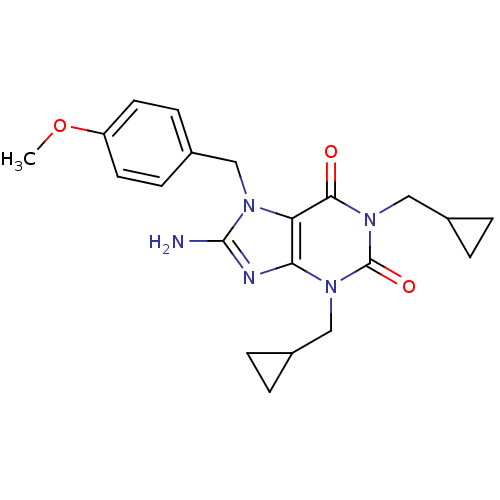
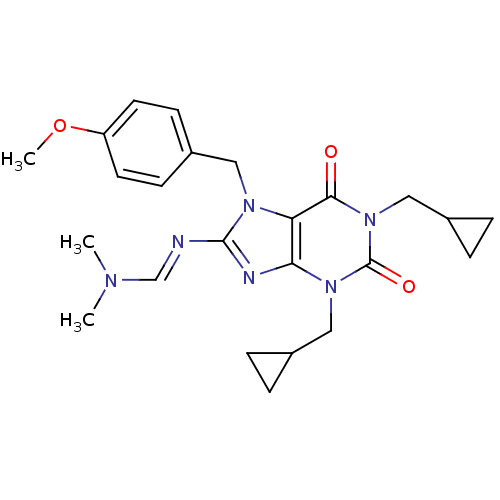
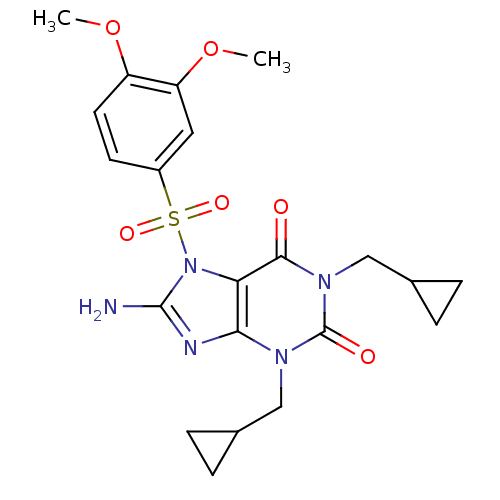
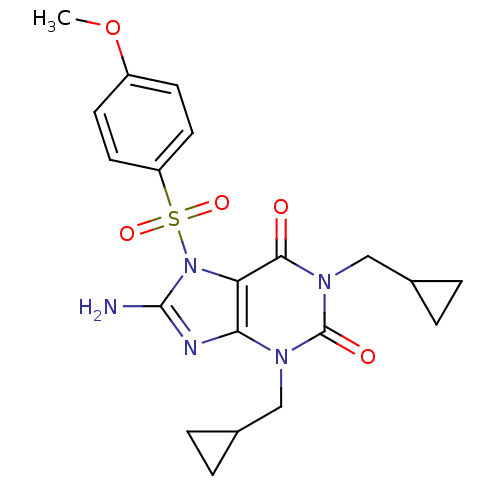
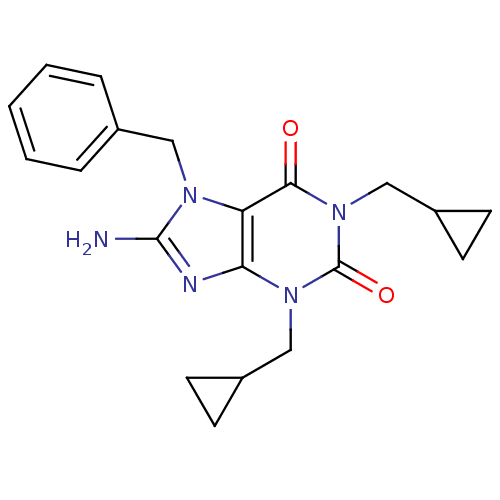
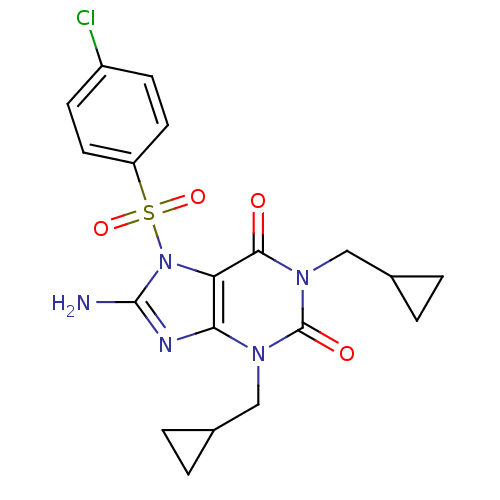
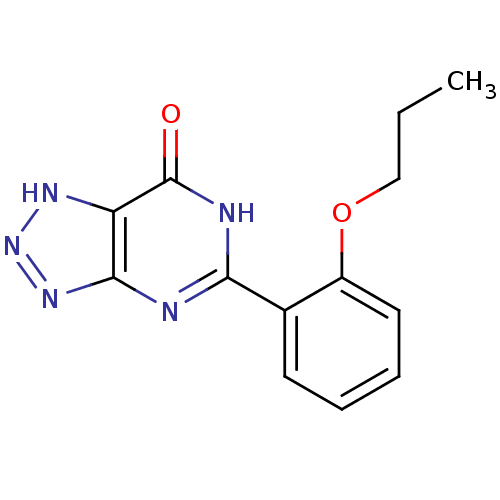
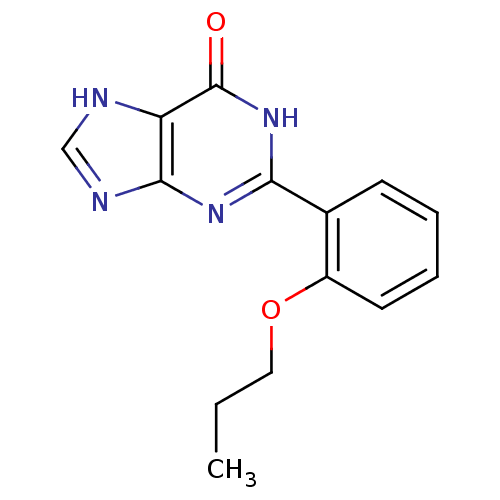
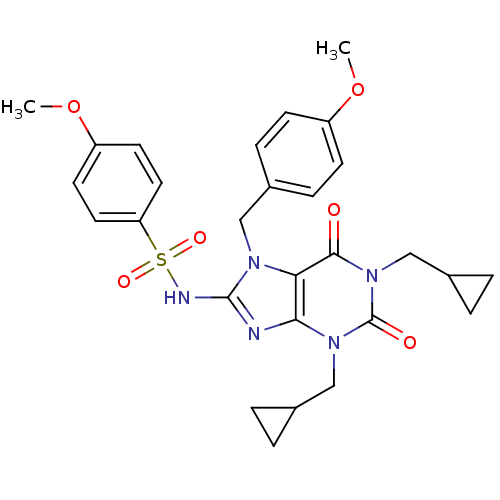
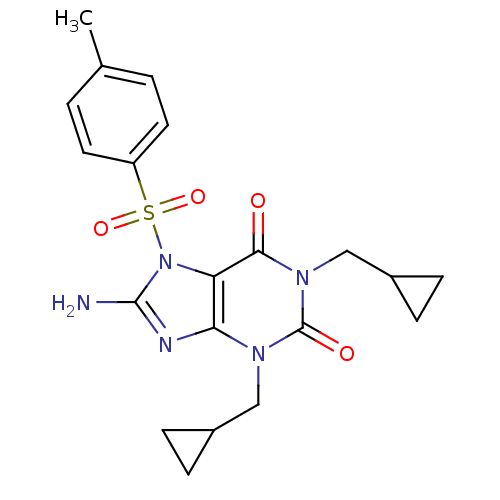
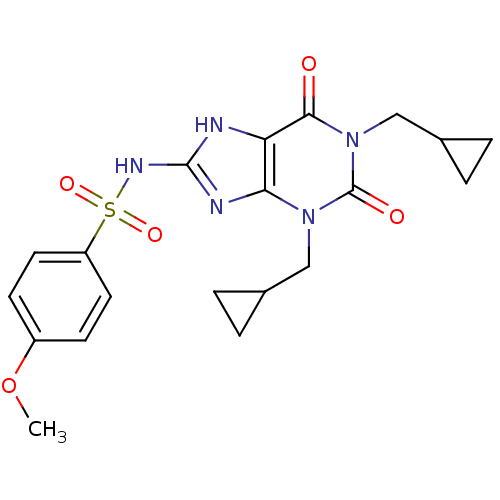
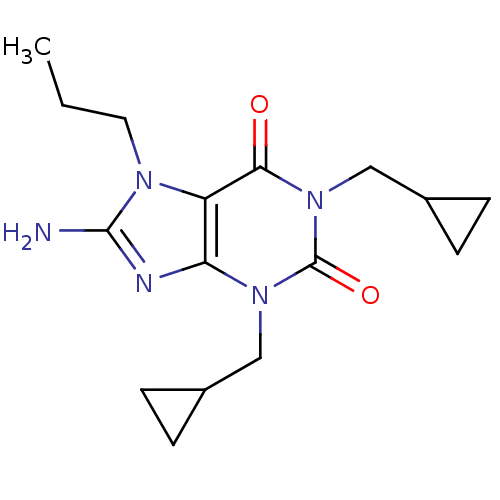
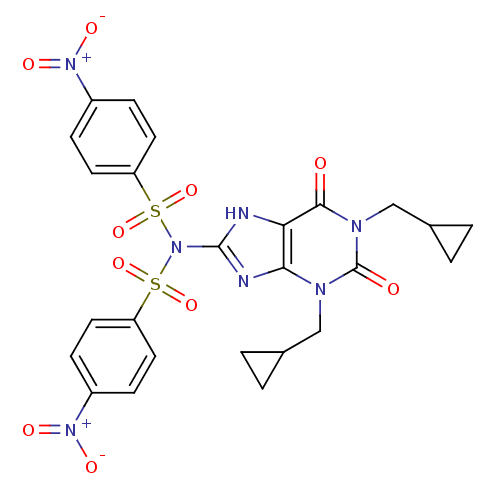
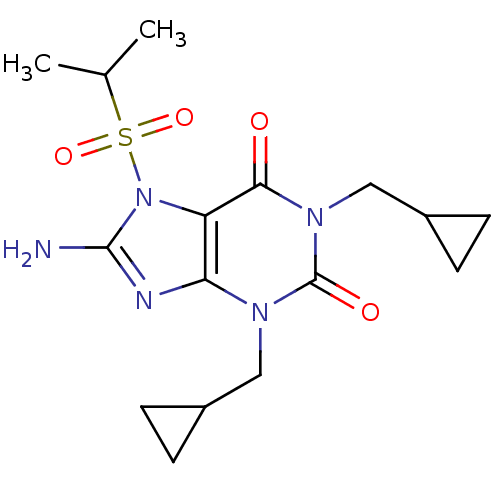
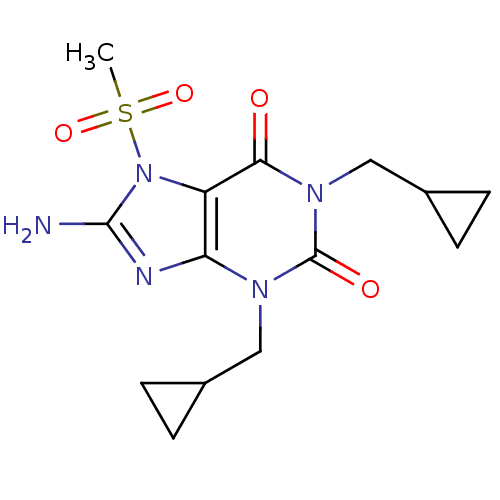
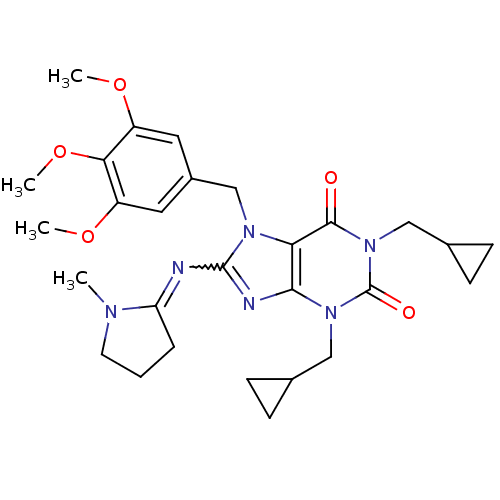
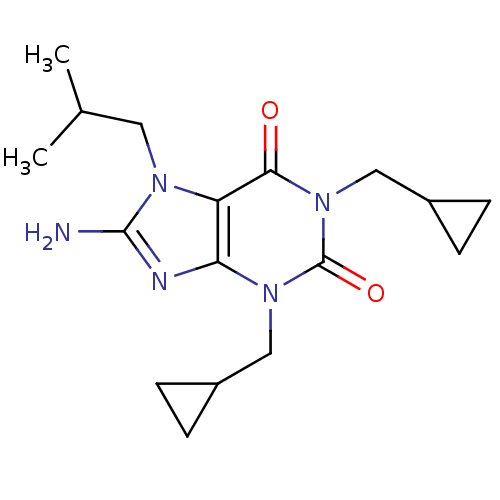
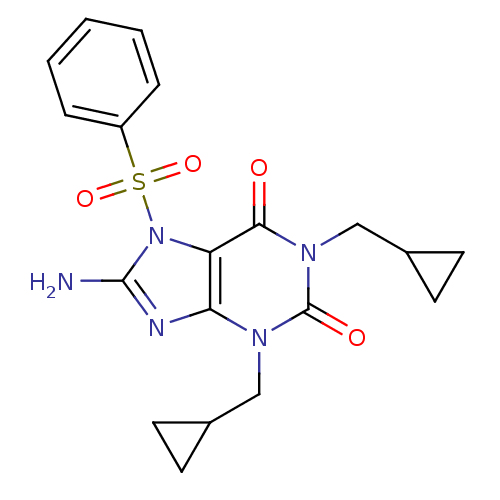
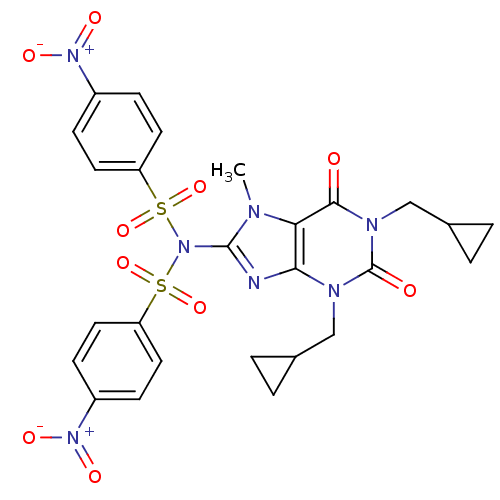
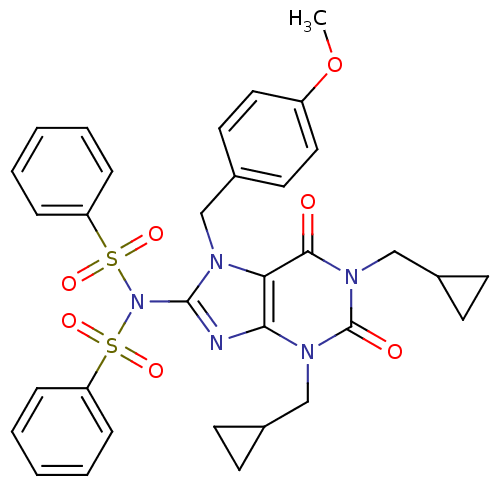
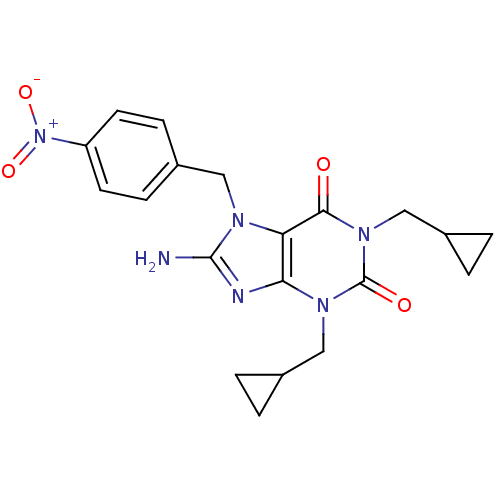
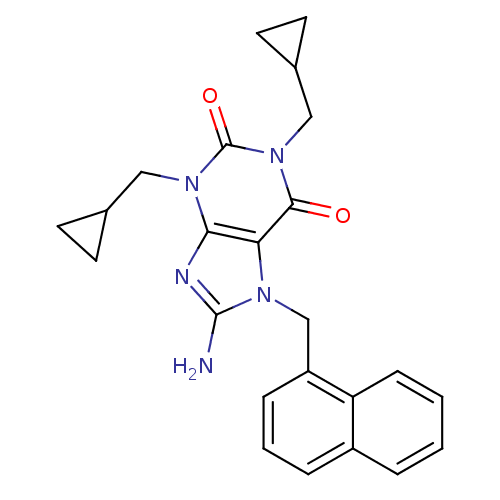
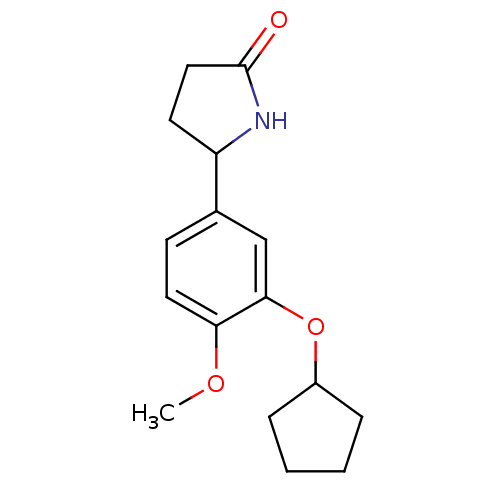
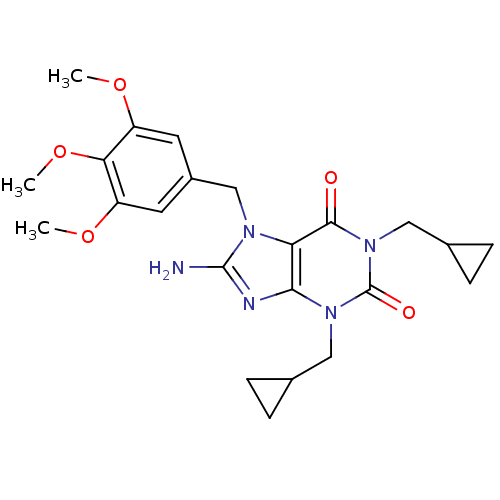
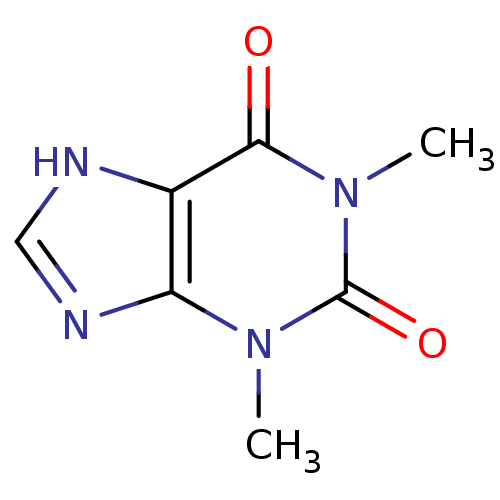
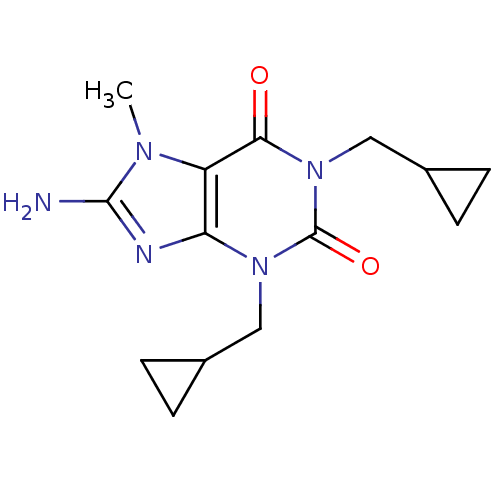
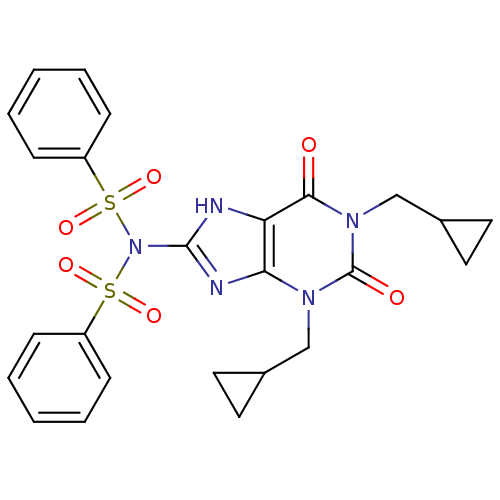
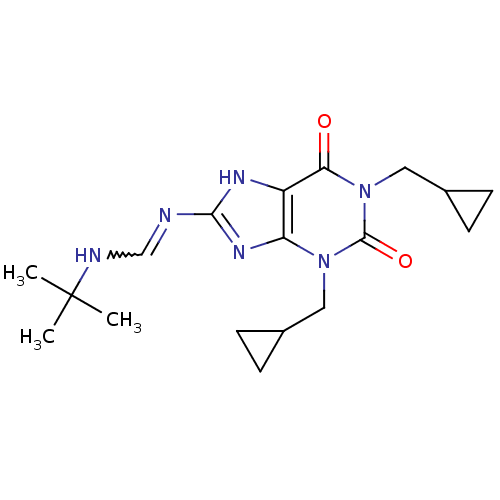
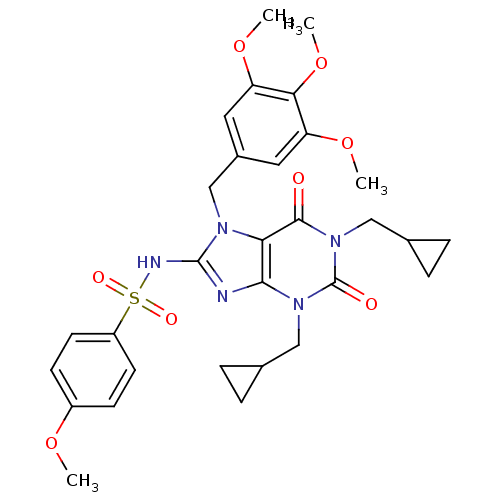
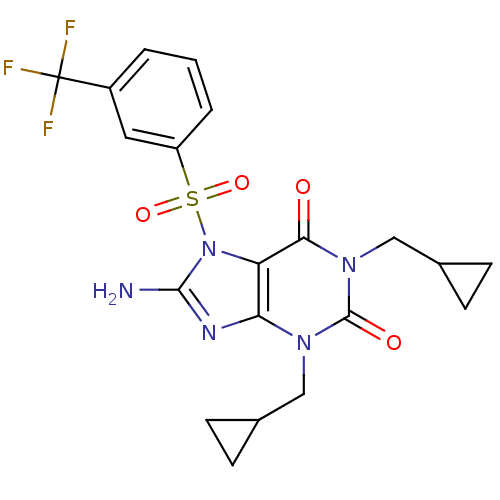
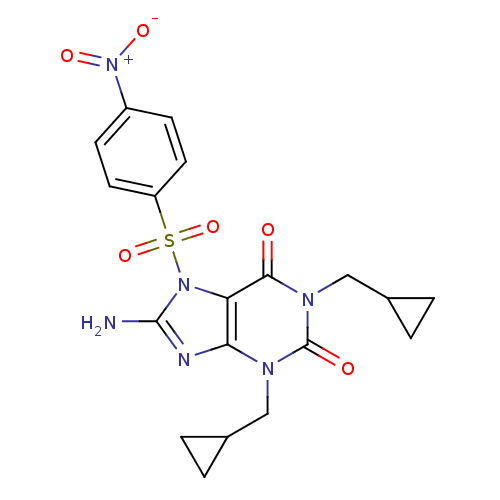
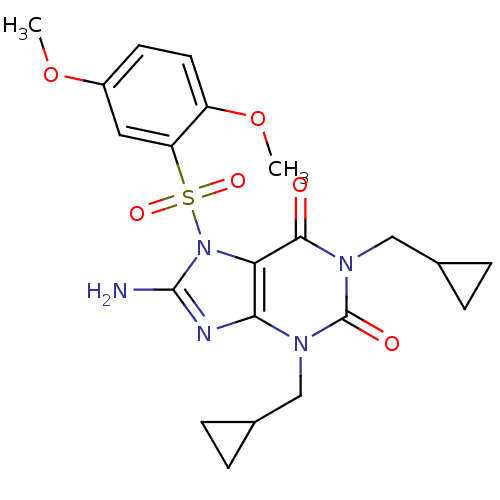
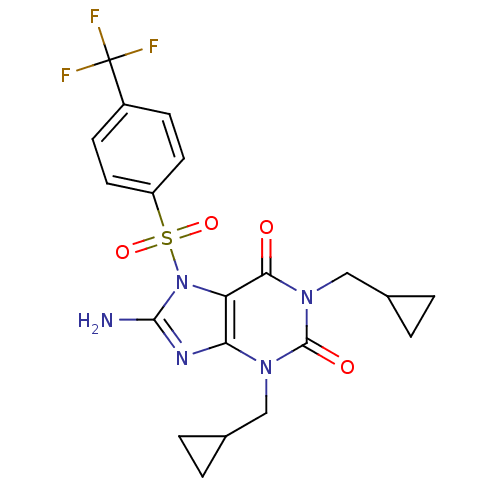
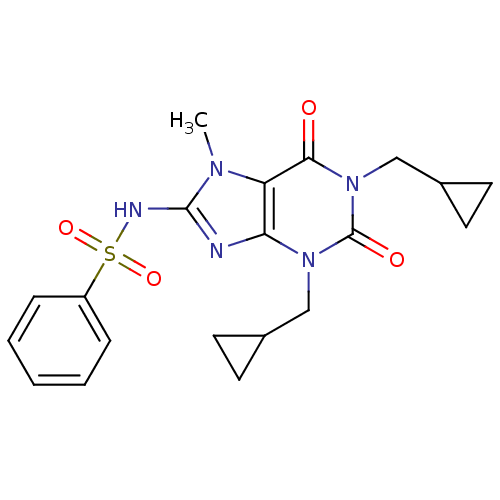
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 7.50E+3nMAssay Description:Inhibition of cGMP hydrolysis by PDE 5AMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 140nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 300nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 440nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 500nMAssay Description:Inhibition of phosphodiesterase 5AMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 610nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 840nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 900nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 900nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of phosphodiesterase 5AMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.20E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.20E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.20E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.40E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.70E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.80E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.00E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.50E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.60E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.80E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 4.10E+3nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-dependent 3',5'-cyclic phosphodiesterase(Bos taurus)
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 5.40E+3nMAssay Description:Inhibition of phosphodiesterase 2 at 100 uMMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 6.50E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 8.30E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 9.80E+3nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetDual specificity calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1C(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibition of phosphodiesterase 1C at 100 uMMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.30E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.40E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of adenosine binding to A1 receptorof rat brain homogenatesMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.00E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.00E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.20E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 2.40E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.20E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.40E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.70E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.70E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.80E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 3.80E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 4.90E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 5.30E+4nMAssay Description:Concentration required to inhibit 50% activity of phosphodiesterase VA isoenzyme at 100 microM.More data for this Ligand-Target Pair