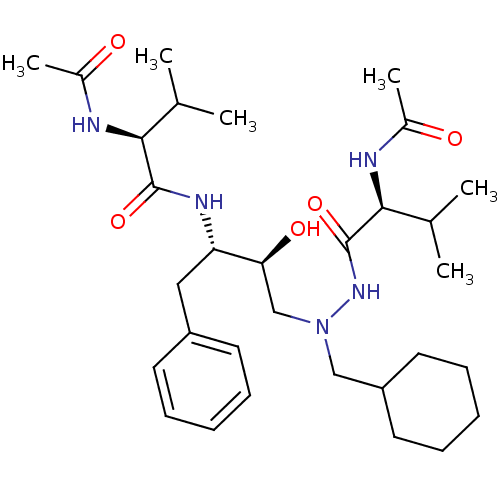
BDBM201 (2S)-N-[(2S,3S)-4-[(2S)-N'-(cyclohexylmethyl)-2-acetamido-3-methylbutanehydrazido]-3-hydroxy-1-phenylbutan-2-yl]-2-acetamido-3-methylbutanamide::CGP 53820
SMILES CC(C)[C@H](NC(C)=O)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)CN(CC1CCCCC1)NC(=O)[C@@H](NC(C)=O)C(C)C
InChI Key InChIKey=JNBVLGDICHLLTN-DZUOILHNSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 201
Found 7 hits for monomerid = 201
Affinity DataIC50: 8.5nMpH: 6.0 T: 2°CAssay Description:A peptide cleavage assay was performed using the icosapeptide H-Arg-Arg-Ser-Asn-Gln-Val-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Asn-Ile-Gln-Gly-Arg-Arg-OH, a...More data for this Ligand-Target Pair
Affinity DataIC50: 7.50E+3nMAssay Description:The compound was tested for its inhibitory activity against human cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: >5.80E+4nMAssay Description:The compound was tested for its inhibitory activity against human reninMore data for this Ligand-Target Pair
Affinity DataIC50: 9nMpH: 6.0Assay Description:In vitro inhibition of HIV-1 protease at pH 6 using 164 microM peptide substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:The compound was tested for its inhibitory activity against human gastricsinMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:The compound was tested for its inhibitory activity against human cathepsin EMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:The compound was tested for its inhibitory activity against human pepsinMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)