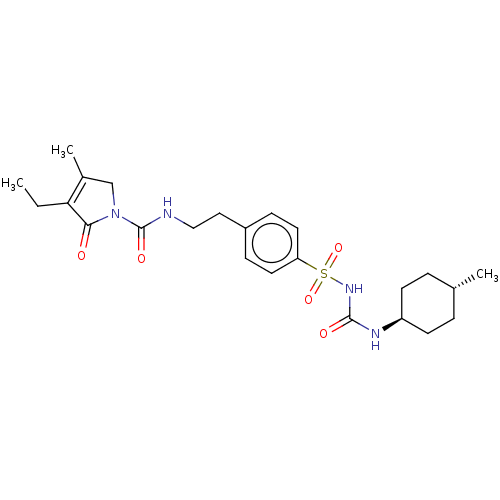
BDBM50237590 Amaryl::CHEBI:5383::Glimepiride::HOE-490::Niddaryl
SMILES CCC1=C(C)CN(C(=O)NCCc2ccc(cc2)S(=O)(=O)NC(=O)N[C@H]2CC[C@H](C)CC2)C1=O
InChI Key InChIKey=WIGIZIANZCJQQY-UHFFFAOYSA-N
Data 9 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 50237590
Found 9 hits for monomerid = 50237590
Affinity DataIC50: 850nMAssay Description:Inhibition of N-terminal His-tagged human AKR1C3 expressed in Escherichia coli BL21(Condon Plus) competent cells using 9,10-phenanthrenequinone as su...More data for this Ligand-Target Pair
Affinity DataIC50: 6.39E+3nMAssay Description:Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.86E+3nMAssay Description:Inhibition of N-terminal His-tagged human AKR1C1 expressed in Escherichia coli BL21 (Condon Plus) competent cells using 9,10-phenanthrenequinone as s...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human BSEP expressed in baculovirus transfected fall armyworm Sf21 cell membranes vesicles assessed as reduction in ATP-dependent [3H]-...More data for this Ligand-Target Pair
Affinity DataIC50: 1.57E+4nMAssay Description:Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.57E+4nMAssay Description:Inhibition of human BSEP expressed in fall armyworm sf9 cell plasma membrane vesicles assessed as reduction in vesicle-associated [3H]-taurocholate t...More data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibition of recombinant human BSEP expressed in baculovirus infected sf9 cell membrane vesicles assessed as reduction in ATP or AMP-dependent [3H]-...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Inhibition of N-terminal His-tagged human AKR1C2 expressed in Escherichia coli BL21 (Condon Plus) competent cells using 9,10-phenanthrenequinone as s...More data for this Ligand-Target Pair
Affinity DataIC50: 6.40E+4nMAssay Description:Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and...More data for this Ligand-Target Pair