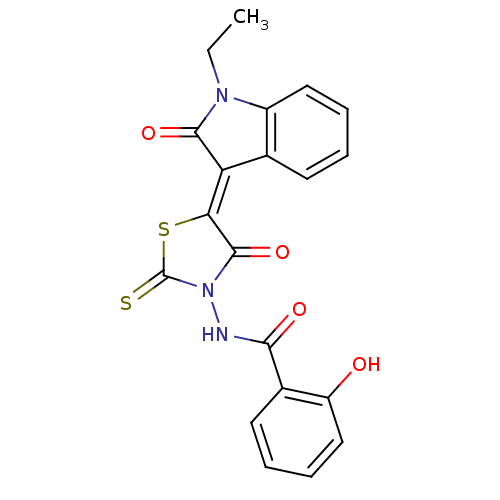
BDBM52773 MLS000777231::N-[(5Z)-5-(1-ethyl-2-keto-indolin-3-ylidene)-4-keto-2-thioxo-thiazolidin-3-yl]-2-hydroxy-benzamide::N-[(5Z)-5-(1-ethyl-2-oxidanylidene-indol-3-ylidene)-4-oxidanylidene-2-sulfanylidene-1,3-thiazolidin-3-yl]-2-oxidanyl-benzamide::N-[(5Z)-5-(1-ethyl-2-oxo-3-indolylidene)-4-oxo-2-sulfanylidene-3-thiazolidinyl]-2-hydroxybenzamide::N-[(5Z)-5-(1-ethyl-2-oxoindol-3-ylidene)-4-oxo-2-sulfanylidene-1,3-thiazolidin-3-yl]-2-hydroxybenzamide::N-{5-[1-Ethyl-2-oxo-1,2-dihydro-indol-(3Z)-ylidene]-4-oxo-2-thioxo-thiazolidin-3-yl}-2-hydroxy-benzamid e::SMR000413626::cid_5522479
SMILES CCN1C(=O)\C(=C2/SC(=S)N(NC(=O)c3ccccc3O)C2=O)c2ccccc12
InChI Key InChIKey=GCHDUTBVIFYTPO-NXVVXOECSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 52773
Found 8 hits for monomerid = 52773
Affinity DataEC50: 5.31E+3nMAssay Description:Broad Institute: MLPCN maternal gene expression Project ID: 2024 Keywords: Zinc finger, C. elegans, maternal gene expression, RNA-protein interac...More data for this Ligand-Target Pair
Affinity DataEC50: 7.47E+3nMAssay Description:Broad Institute: MLPCN maternal gene expression Project ID: 2024 Keywords: Zinc finger, C. elegans, maternal gene expression, RNA-protein interac...More data for this Ligand-Target Pair
TargetM1 family aminopeptidase(Plasmodium falciparum (isolate FcB1 / Columbia))
Srmlsc
Curated by PubChem BioAssay
Srmlsc
Curated by PubChem BioAssay
Affinity DataIC50: 9.43E+3nMAssay Description:Southern Research Molecular Libraries Screening Center (SRMLSC) Southern Research Institute (Birmingham, Alabama) NIH Molecular Libraries Screening...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase 1(Rattus norvegicus (Rat))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 5.35E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetIntestinal-type alkaline phosphatase(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetAlkaline phosphatase, germ cell type(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetAlkaline phosphatase, tissue-nonspecific isozyme(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetProcathepsin L(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataIC50: >5.96E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center Affiliation: The Scripps Research Institute, TSRI Assay Provide...More data for this Ligand-Target Pair