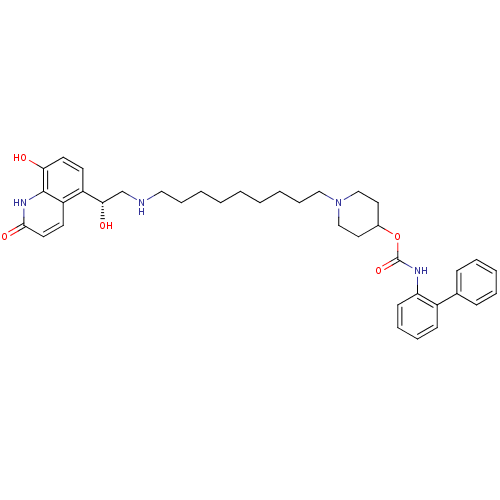
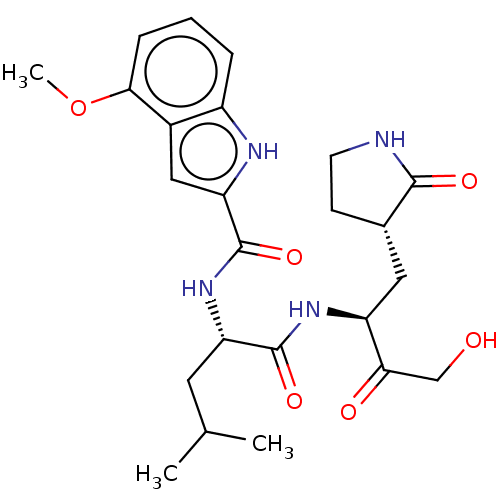
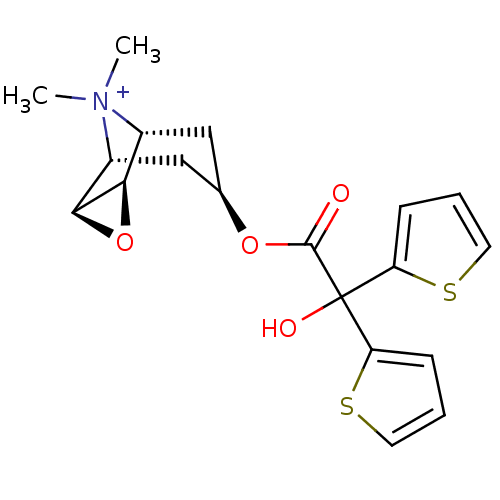
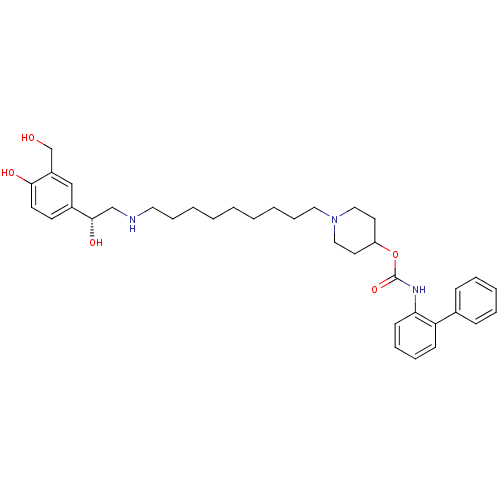
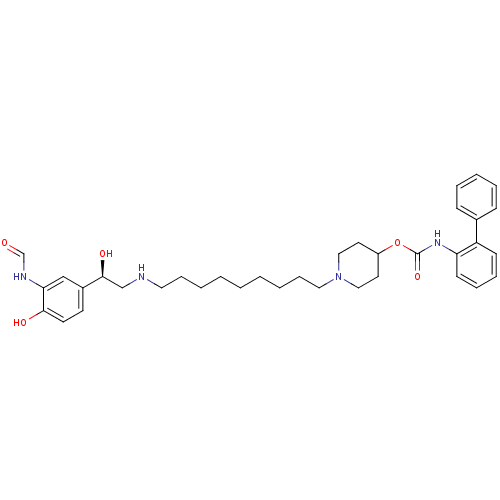
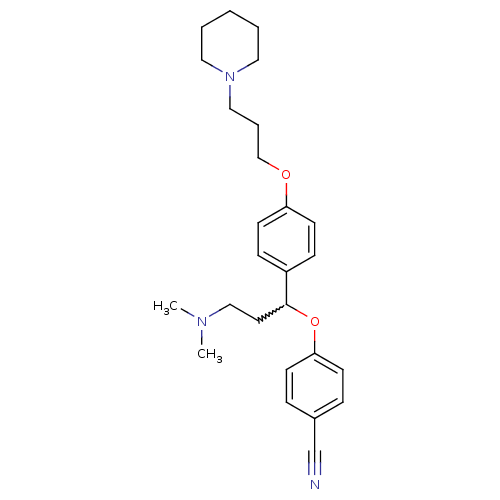
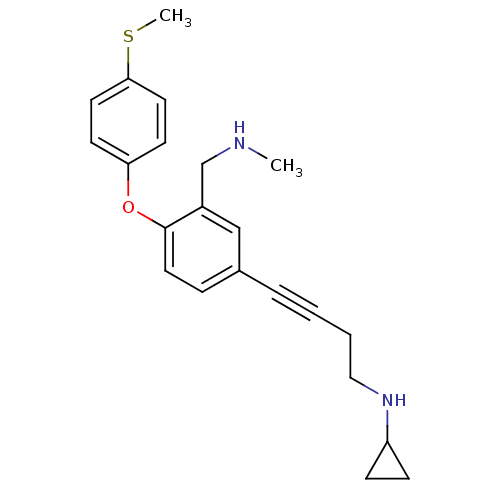
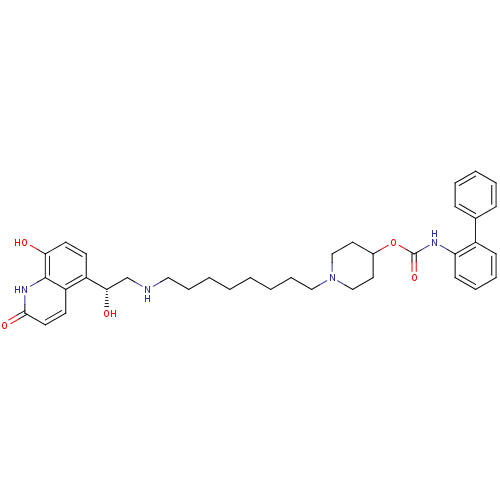
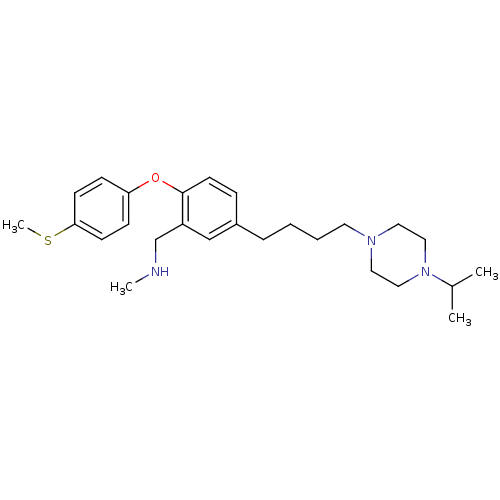
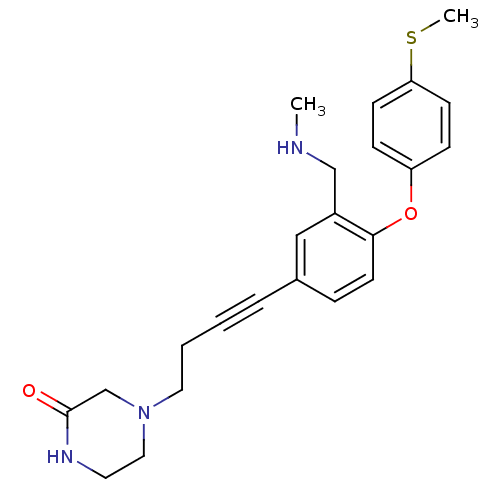
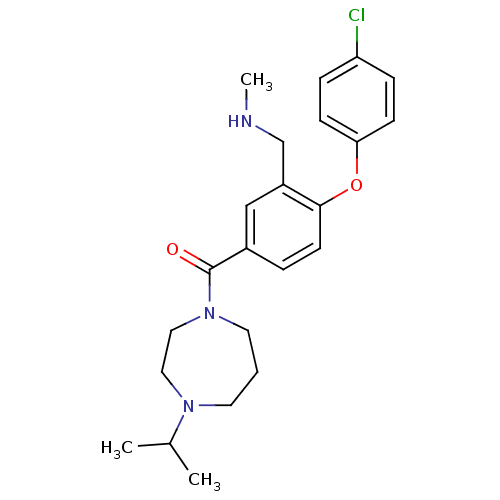
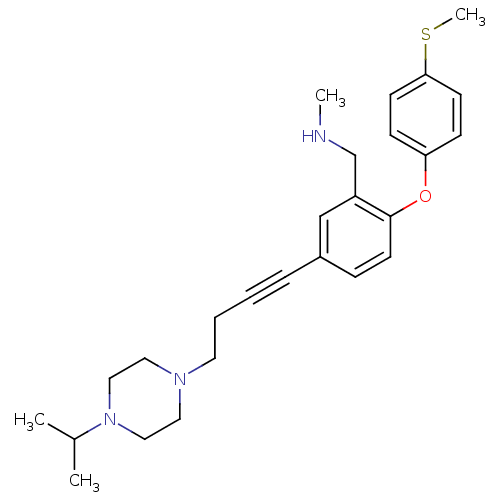
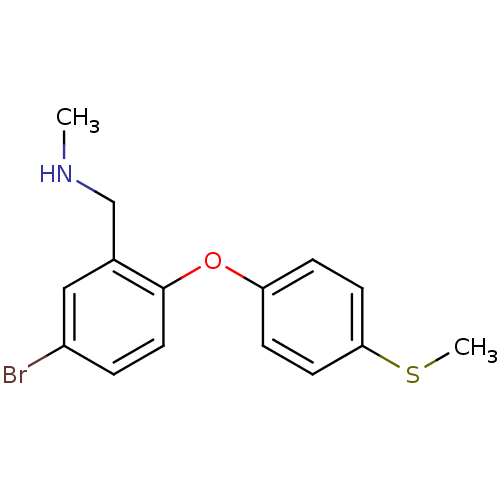
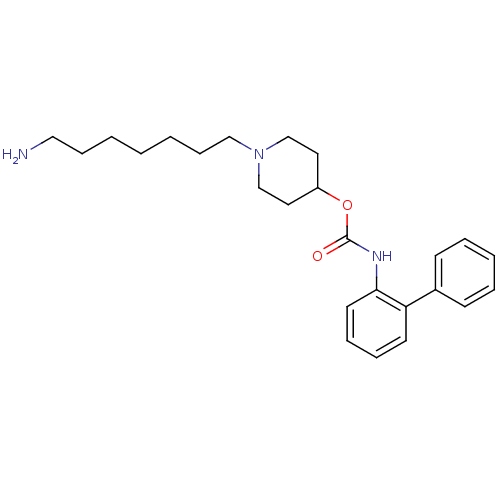
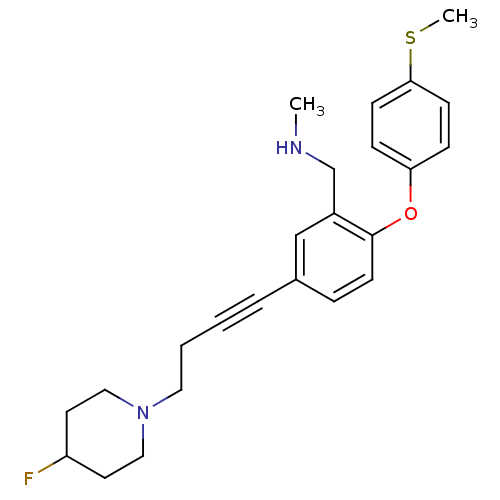
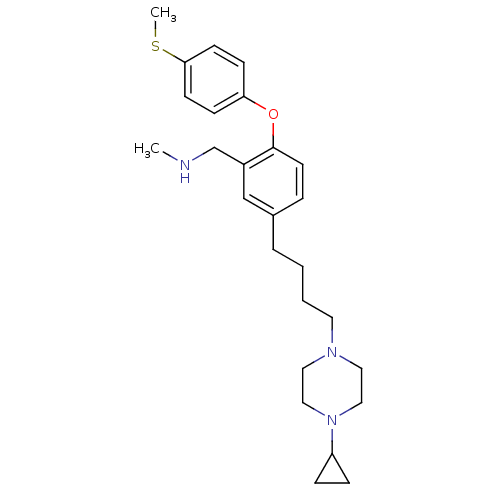
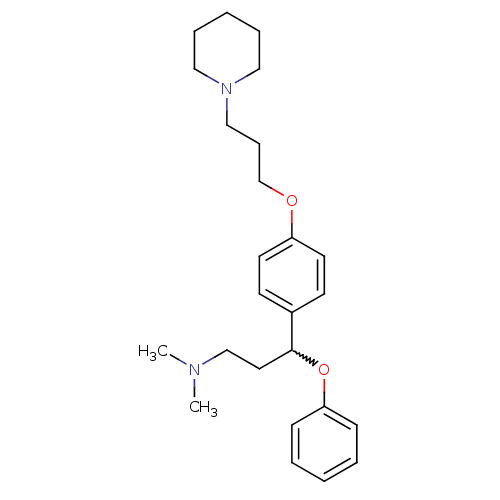
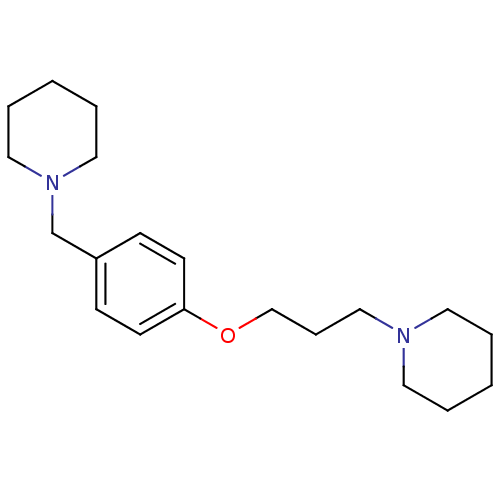
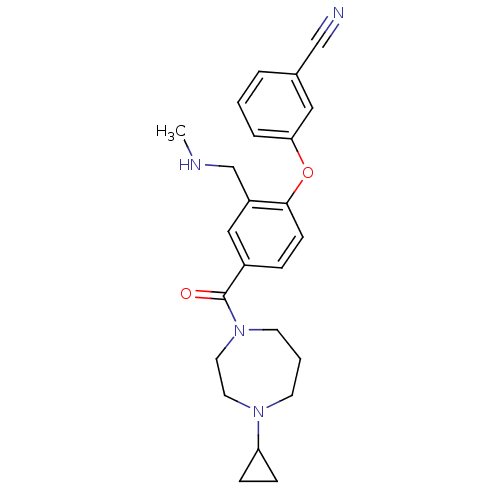
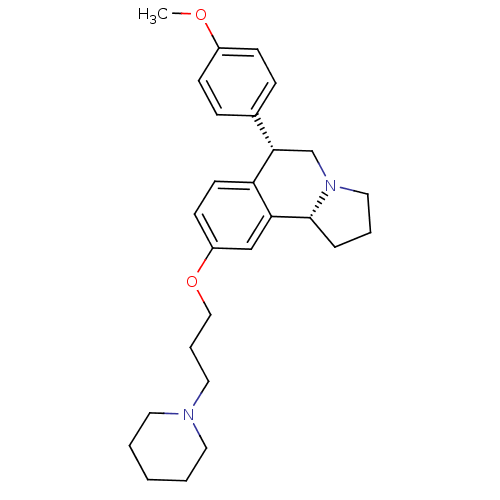
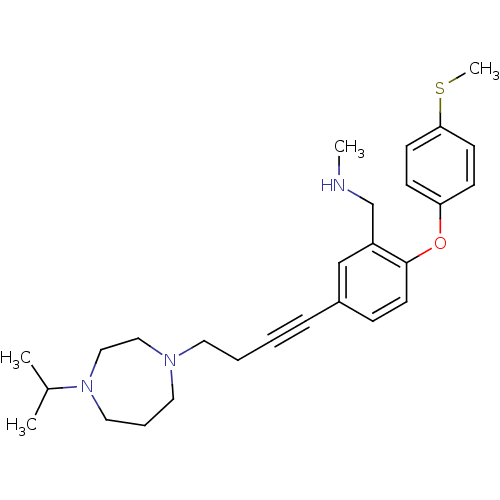
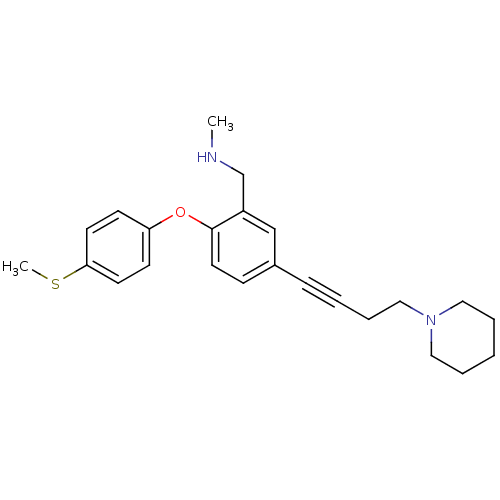
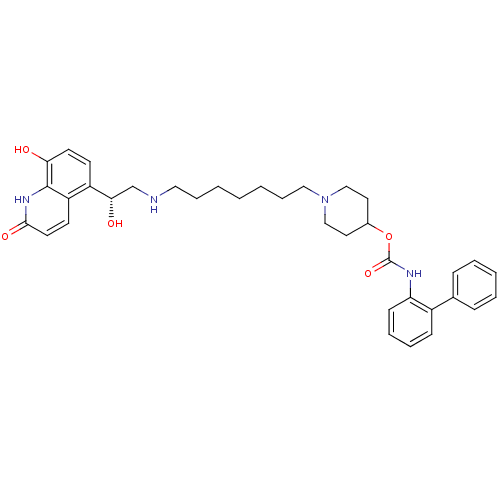
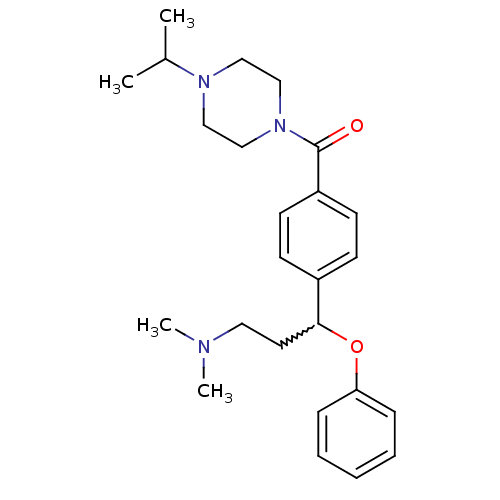
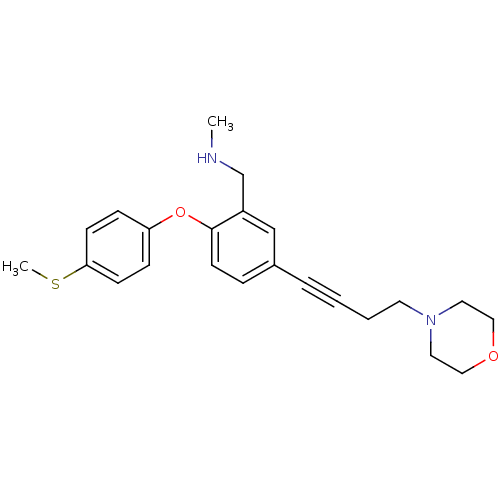
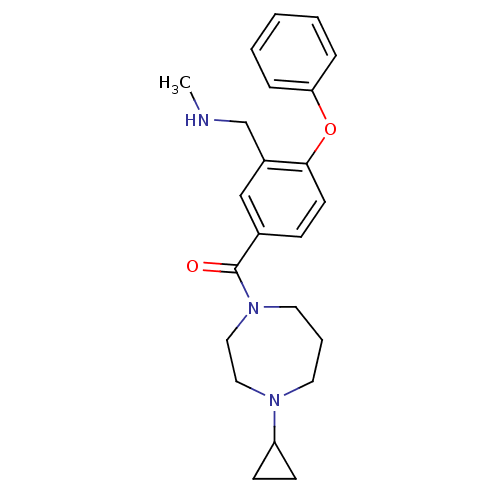
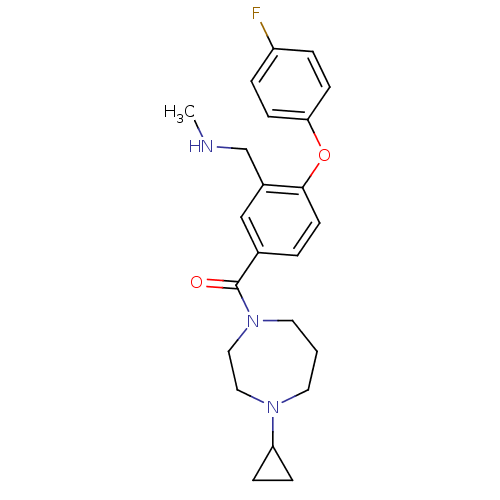
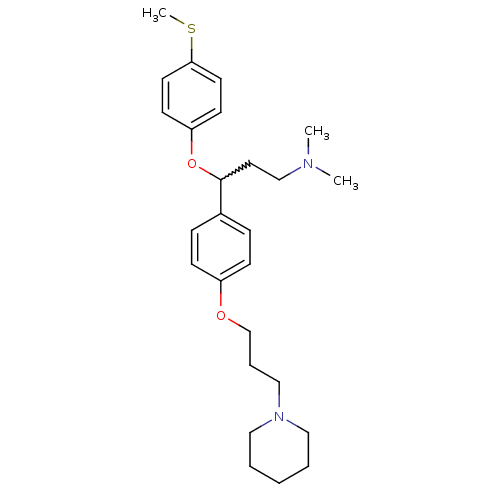
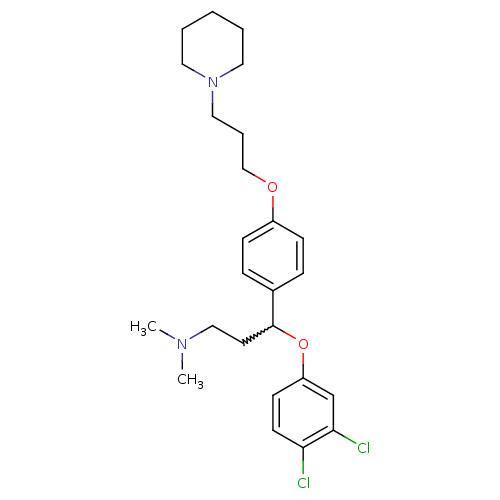
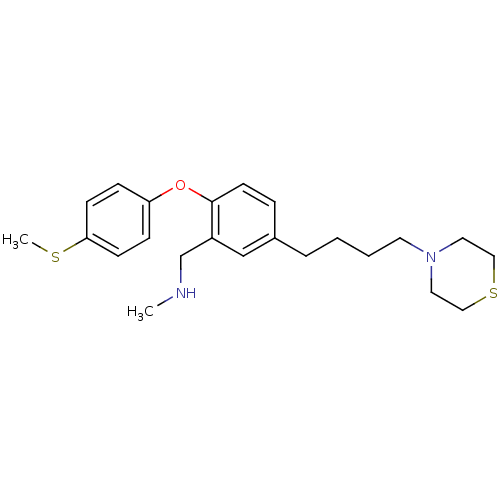
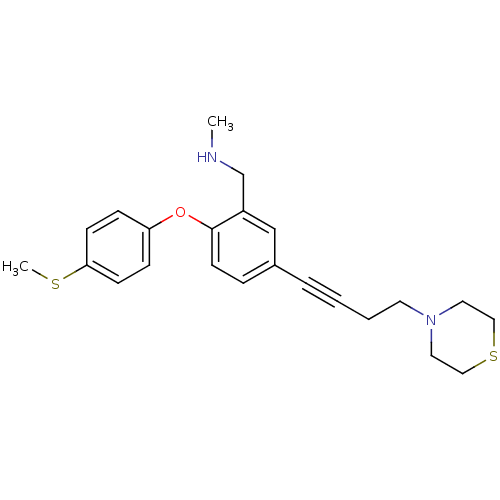
Report error Found 5048 with Last Name = 'king' and Initial = 'e'
Affinity DataKi: 0.0100nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 0.100nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
Affinity DataKi: 0.140nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 0.140nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 0.180nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.320nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.400nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.400nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
Affinity DataKi: 0.530nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Binding affinity to human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to human SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.700nMAssay Description:Binding affinity to human histamine H3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataKi: 0.75nMAssay Description:Displacement of [3H]-N-methyl-scopolamine from human muscarinic M3 receptor after 6 hrs by cell based assayChecked by AuthorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Binding affinity to human SERTMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Inhibition of human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Inhibition of human SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.900nMAssay Description:Proteolytic activity of SARS-CoV-2 Coronavirus 3CL protease is measured using a continuous fluorescence resonance energy transfer assay. The SARS-CoV...More data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity to human SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair
TargetHistamine H3 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity at human histamine H3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rat)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.900nMAssay Description:Binding affinity to rat SERTMore data for this Ligand-Target Pair