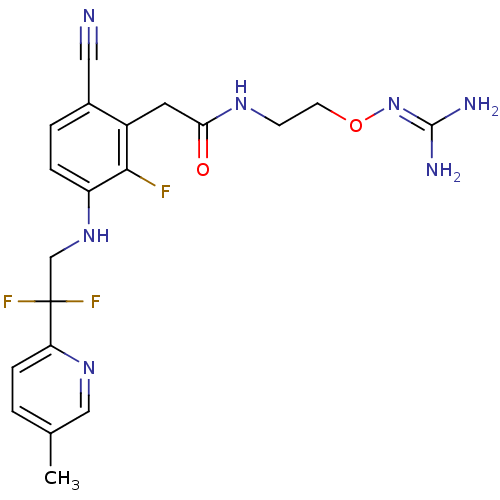
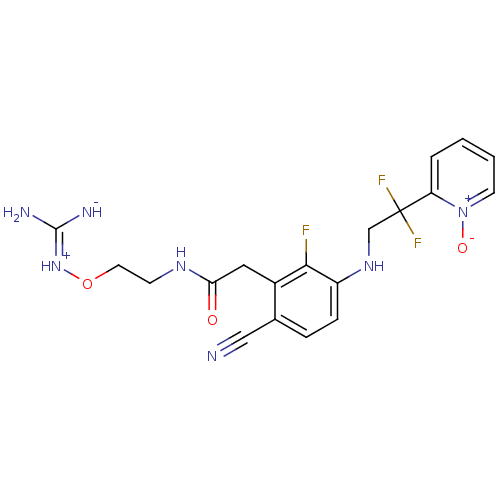
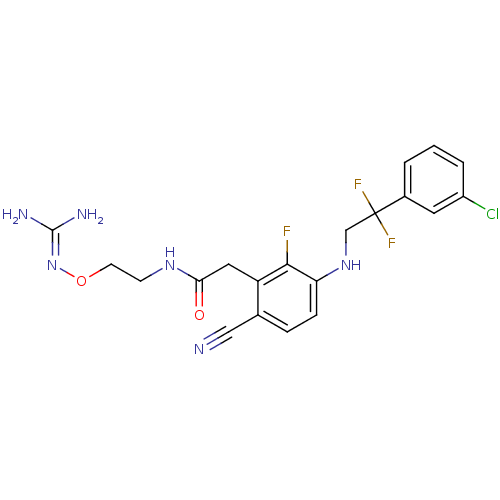
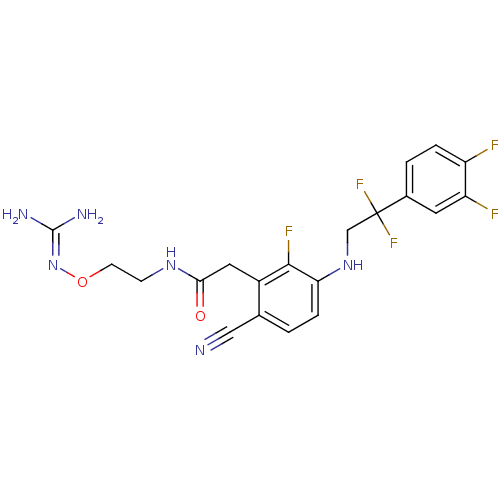
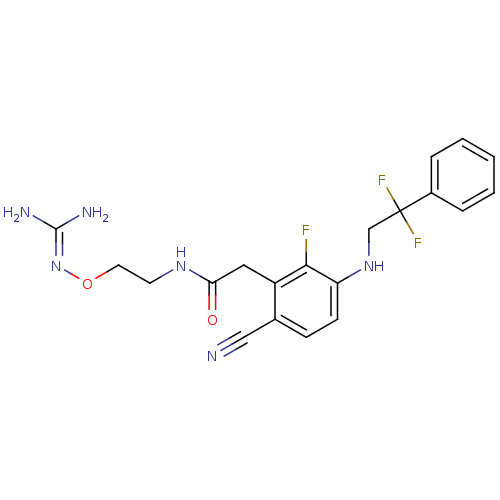
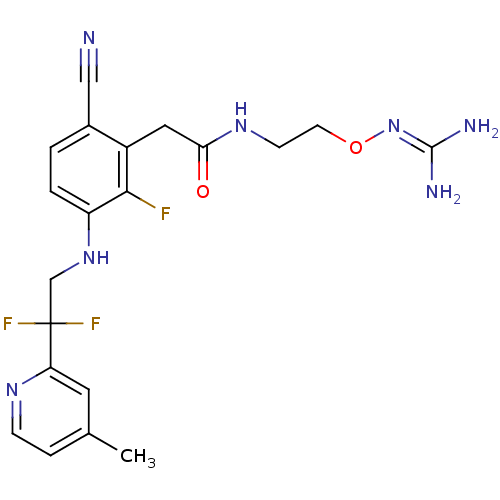
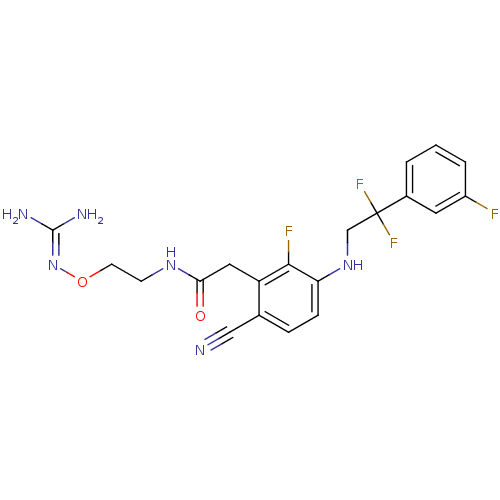
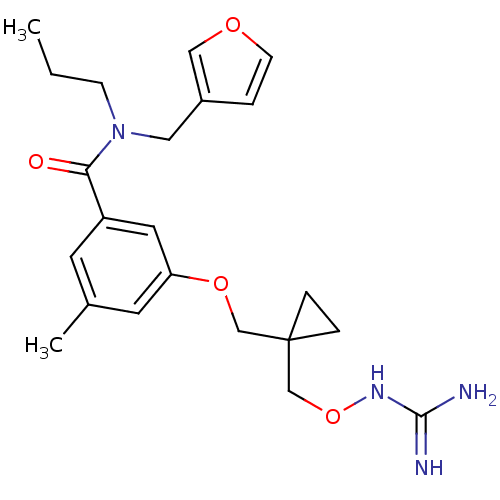
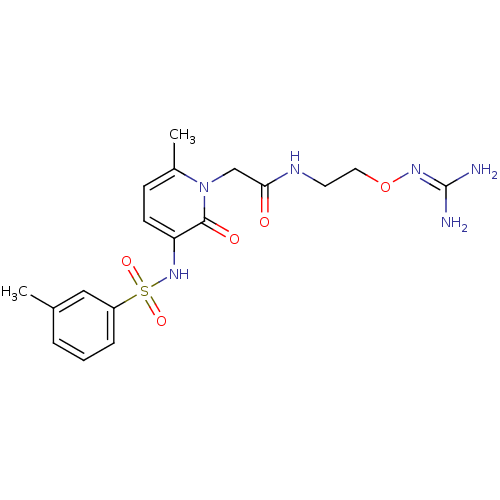
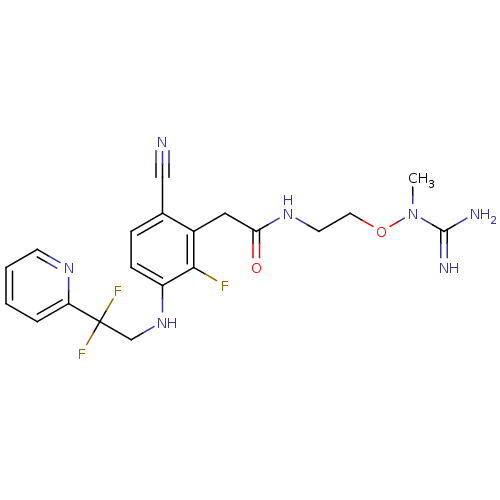
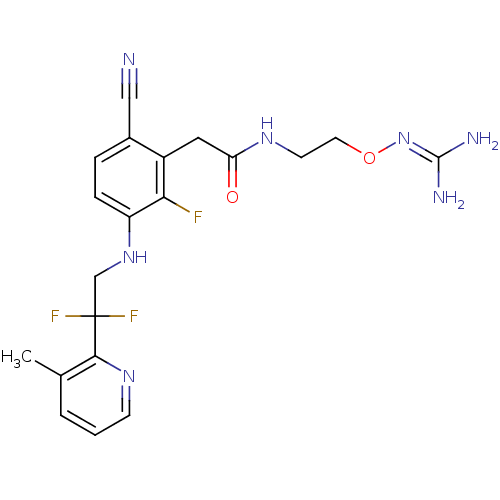
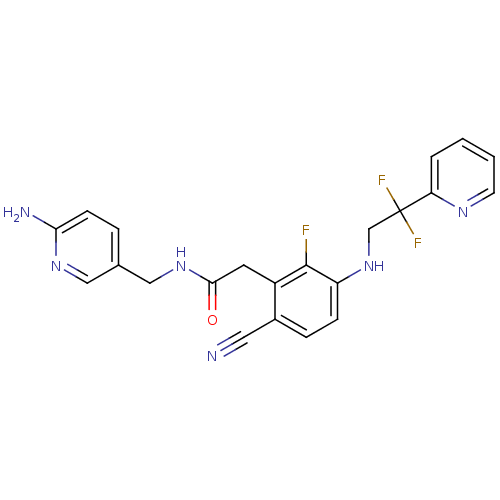
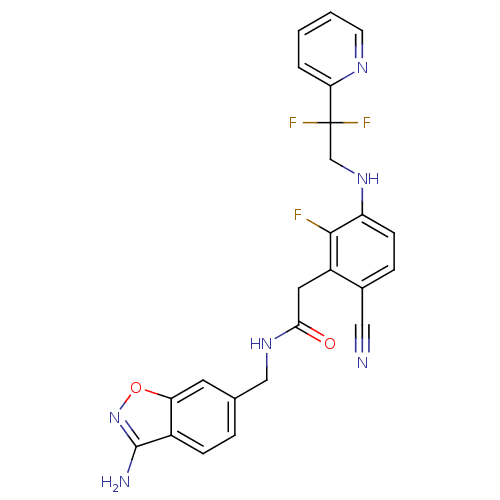
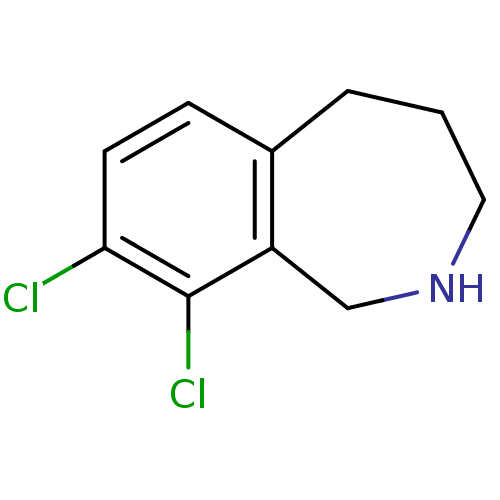
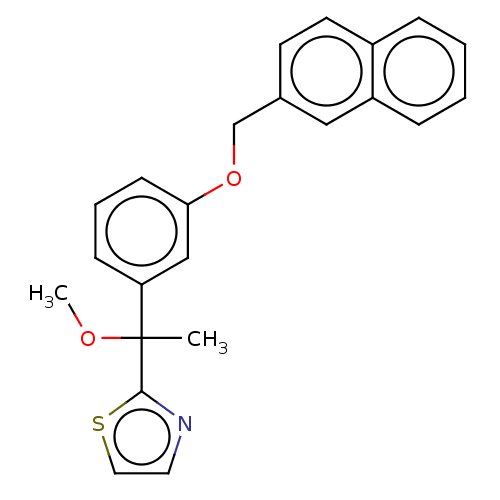
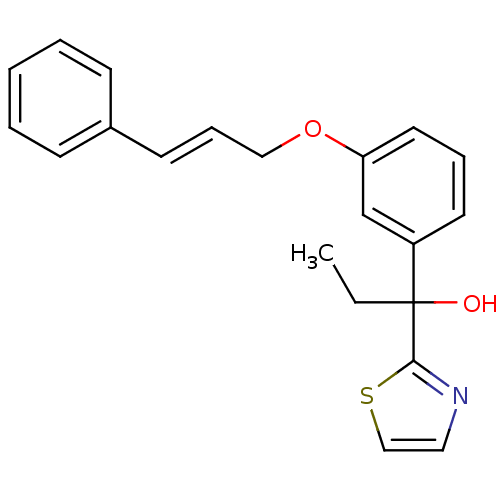
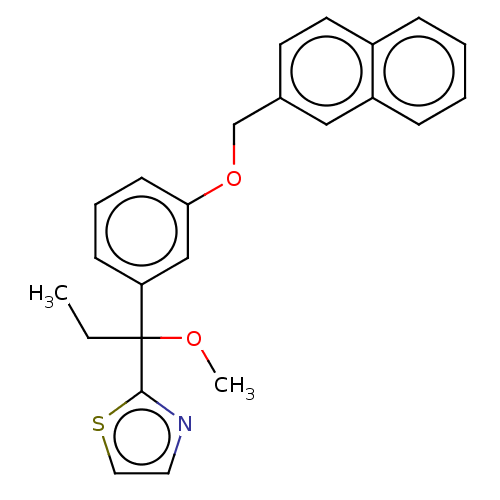
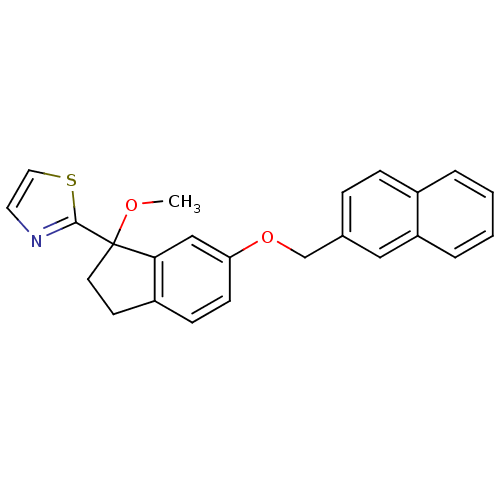
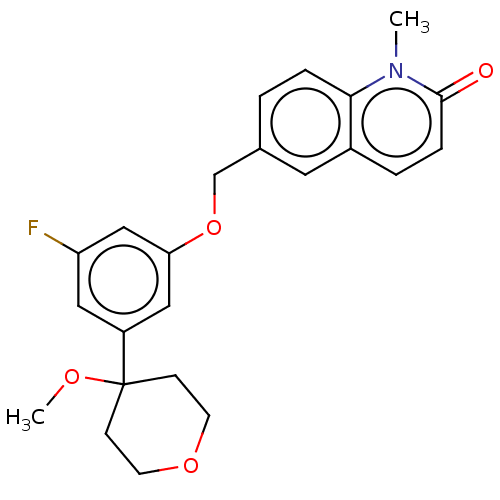
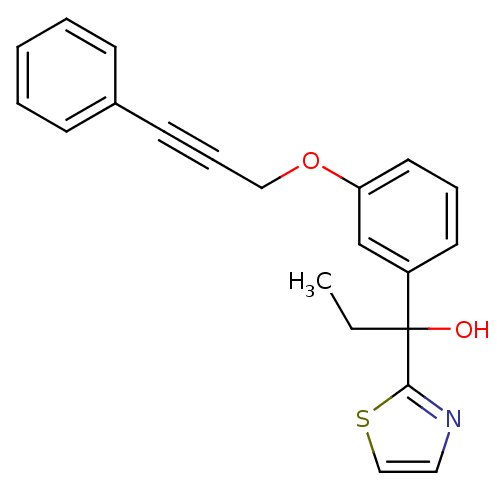
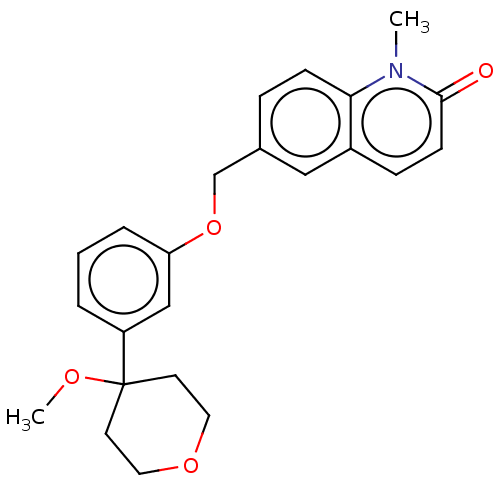
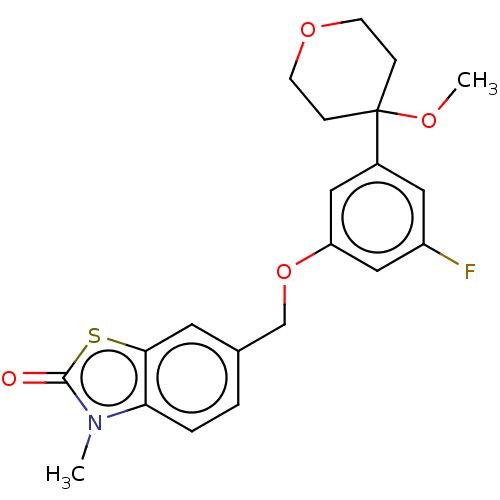
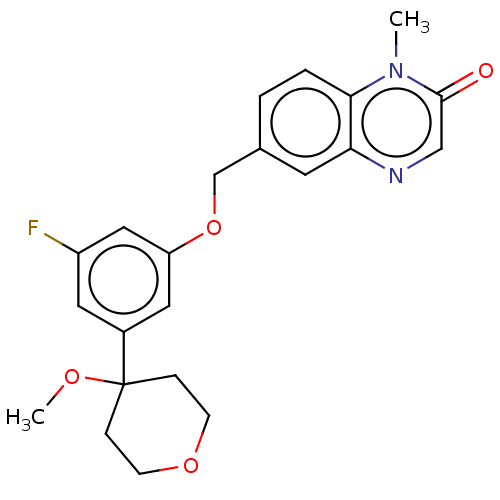
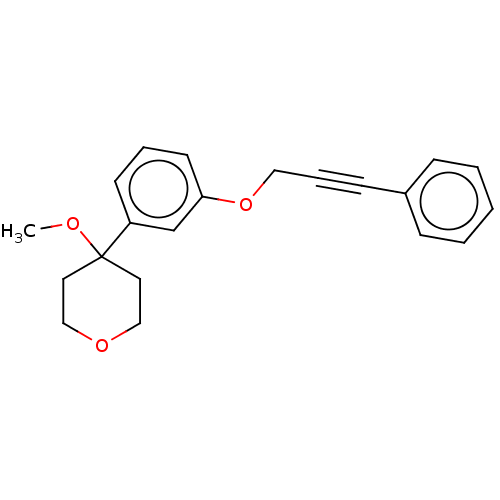
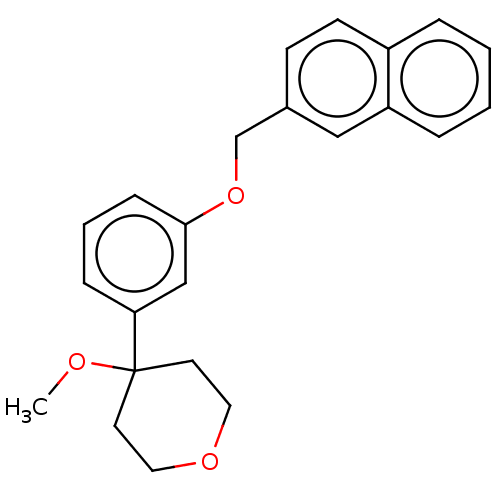
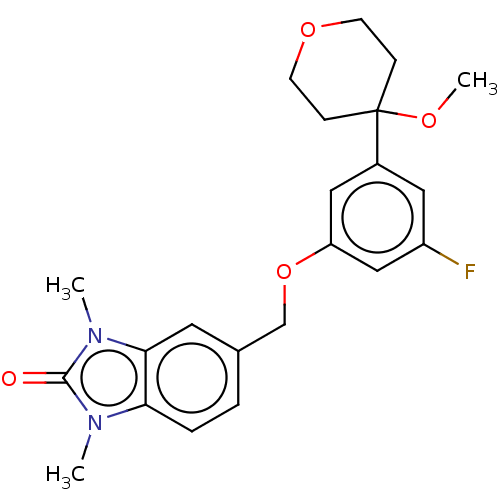
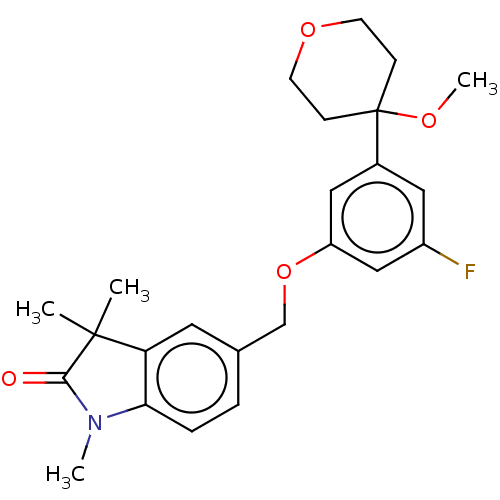
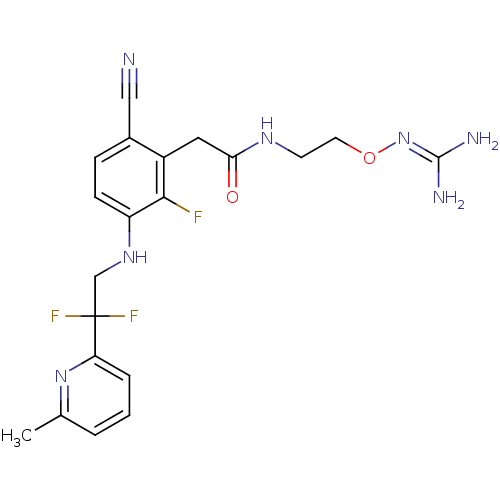
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
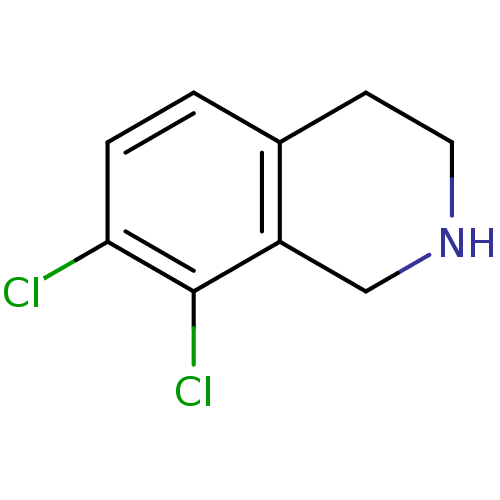
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.380nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
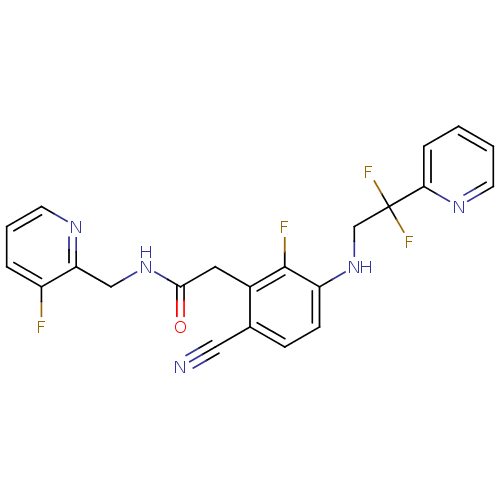
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.570nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
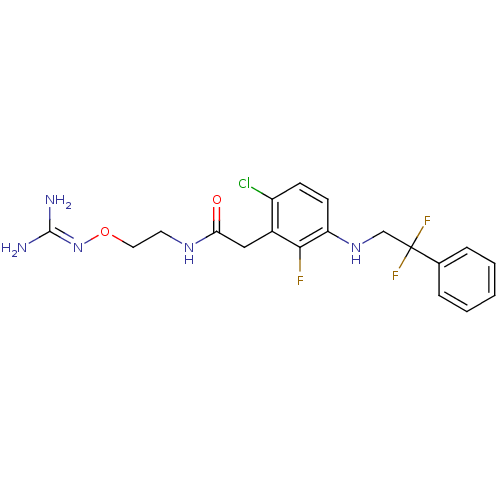
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.650nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.660nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.770nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.790nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.70nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.90nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 3.20nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 4nMAssay Description:Binding affinity to thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 9nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 11nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 19nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 21nM ΔG°: -43.8kJ/molepH: 7.7 T: 2°CAssay Description:The binding affinity of compound was evaluated using [3H] Clonidine as ligand in competition binding experiments. Nonspecific binding was determined ...More data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 33nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 220nM ΔG°: -38.0kJ/molepH: 7.7 T: 2°CAssay Description:The binding affinity of compound was evaluated using [3H] Clonidine as ligand in competition binding experiments. Nonspecific binding was determined ...More data for this Ligand-Target Pair
Affinity DataKi: 220nM ΔG°: -39.5kJ/molepH: 8.0 T: 2°CAssay Description:Enzyme activity is determined by measuring the amount of 3H incorporated into the substrate during the reaction. AdoMet/[methyl-3H]AdoMet serves as a...More data for this Ligand-Target Pair
Affinity DataKi: 260nM ΔG°: -39.1kJ/molepH: 8.0 T: 2°CAssay Description:Enzyme activity is determined by measuring the amount of 3H incorporated into the substrate during the reaction. AdoMet/[methyl-3H]AdoMet serves as a...More data for this Ligand-Target Pair
Affinity DataKi: 370nM ΔG°: -38.2kJ/molepH: 8.0 T: 2°CAssay Description:Enzyme activity is determined by measuring the amount of 3H incorporated into the substrate during the reaction. AdoMet/[methyl-3H]AdoMet serves as a...More data for this Ligand-Target Pair
Affinity DataKi: 560nM ΔG°: -37.1kJ/molepH: 8.0 T: 2°CAssay Description:Enzyme activity is determined by measuring the amount of 3H incorporated into the substrate during the reaction. AdoMet/[methyl-3H]AdoMet serves as a...More data for this Ligand-Target Pair
Affinity DataKi: 4.50E+3nM ΔG°: -30.5kJ/molepH: 7.7 T: 2°CAssay Description:The binding affinity of compound was evaluated using [3H] Clonidine as ligand in competition binding experiments. Nonspecific binding was determined ...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+5nM ΔG°: -22.8kJ/molepH: 7.7 T: 2°CAssay Description:The binding affinity of compound was evaluated using [3H] Clonidine as ligand in competition binding experiments. Nonspecific binding was determined ...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 6nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:Compound was tested for the inhibition of 5-lipoxygenase (5-LPO) in rat basophilic leukemia (RBL) cells.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: <30nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Mus musculus)
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of 5-lipoxygenase in mouse macrophages.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Ici Pharmaceuticals Group
Curated by ChEMBL
Ici Pharmaceuticals Group
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:In vitro inhibition of leukotriene B4 synthesis in human whole blood by inhibiting 5-lipoxygenaseMore data for this Ligand-Target Pair

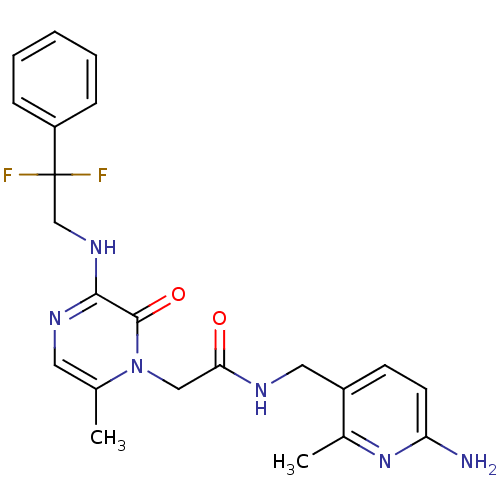
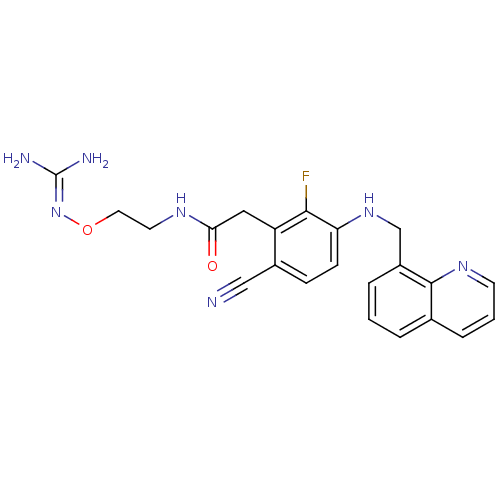
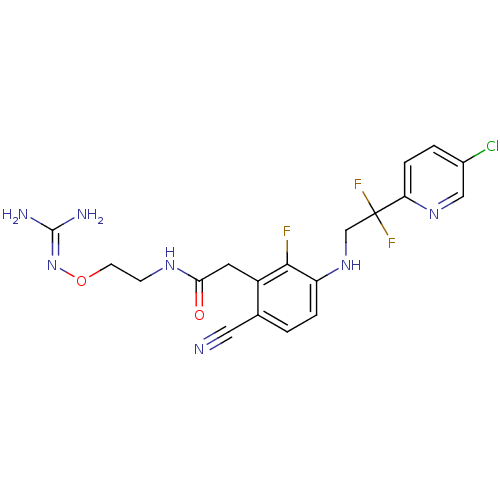
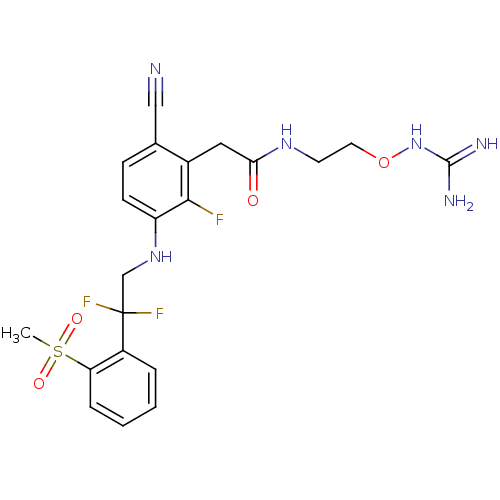
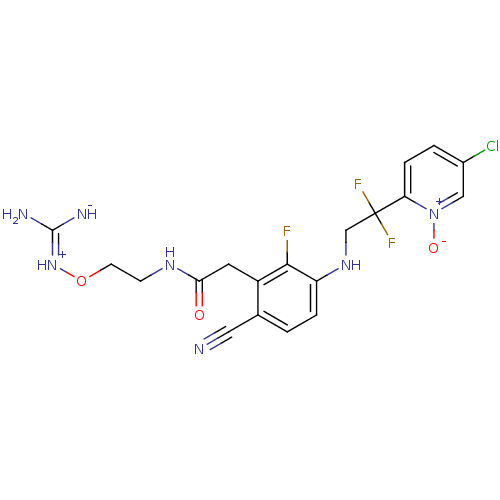
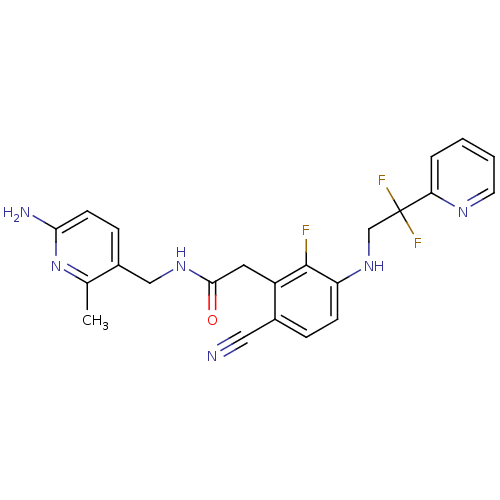

 3D Structure (crystal)
3D Structure (crystal)