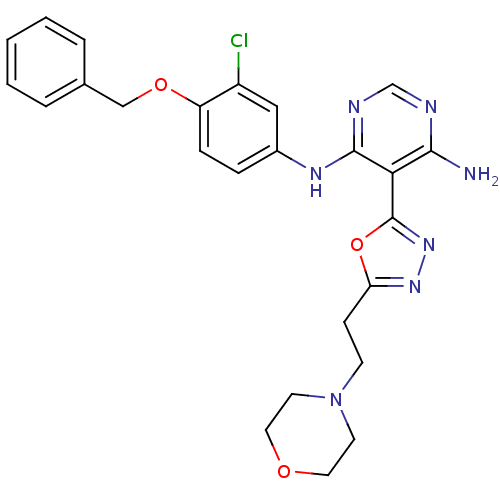
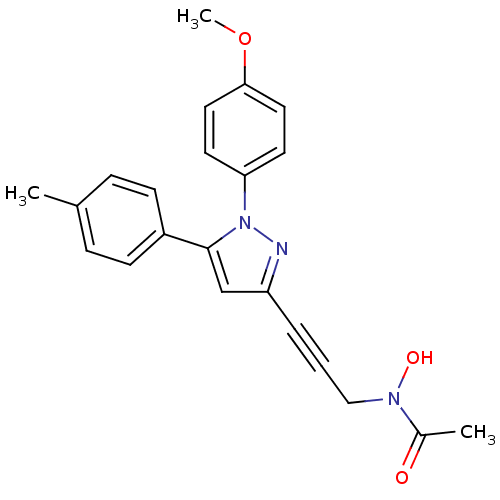
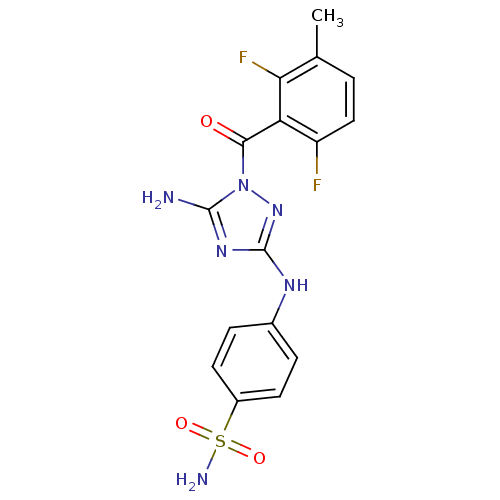
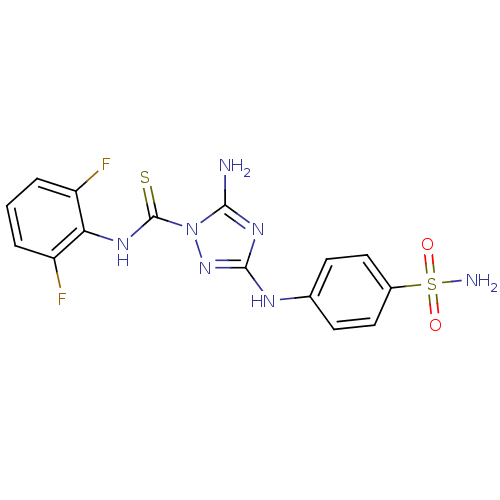
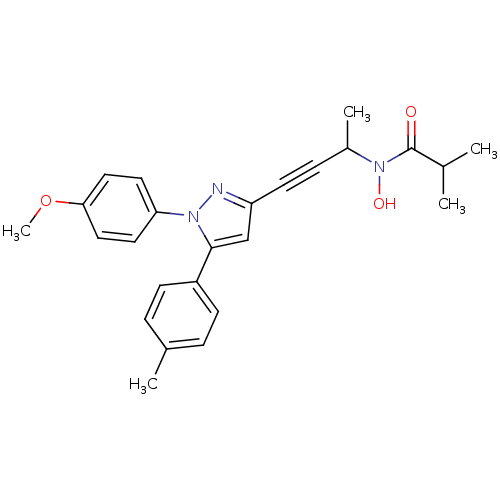
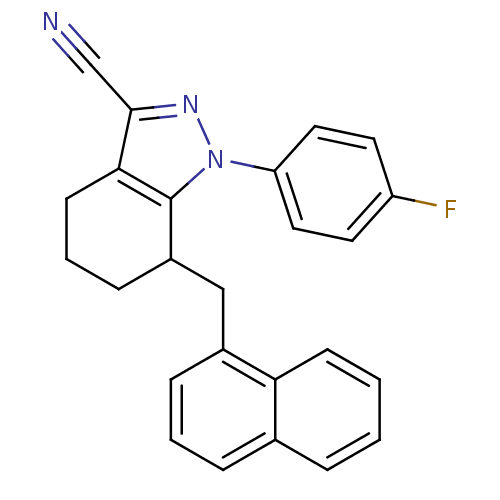
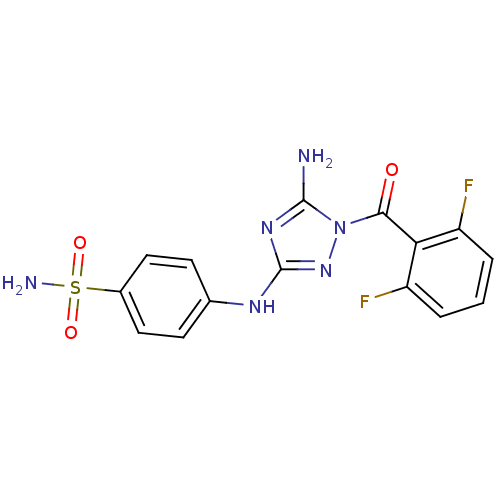
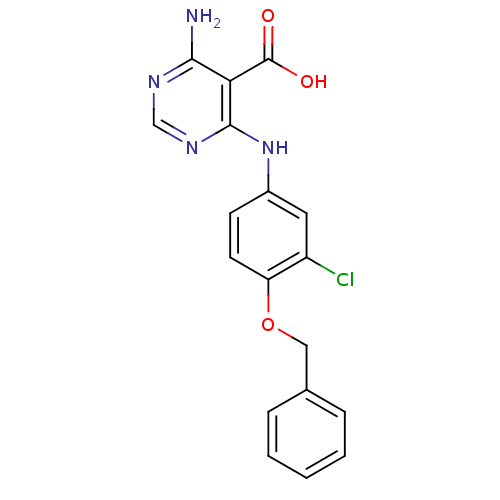
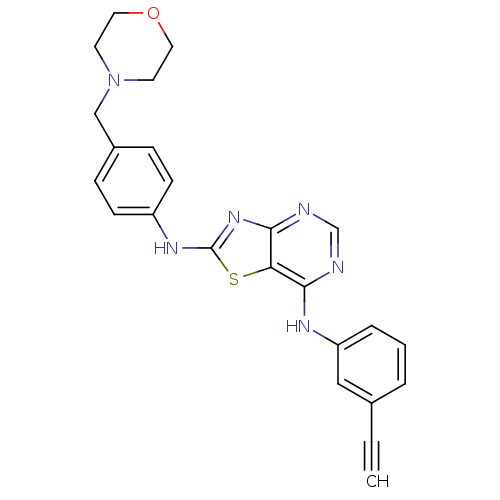
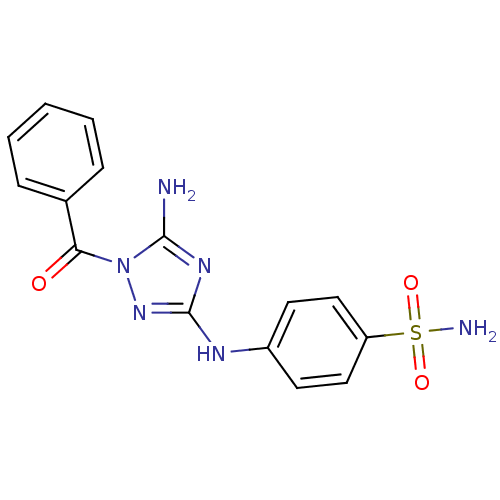
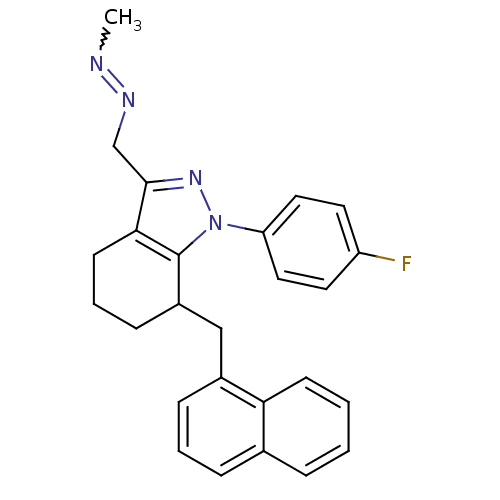
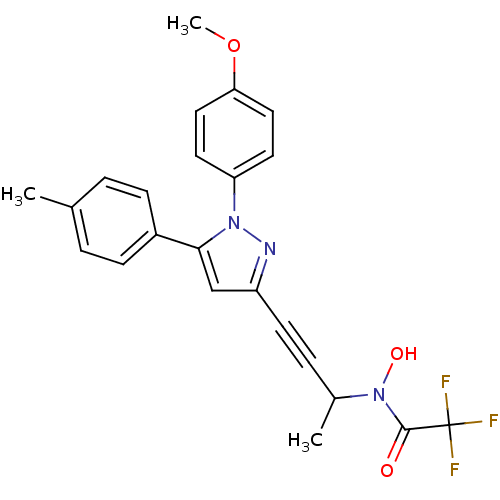
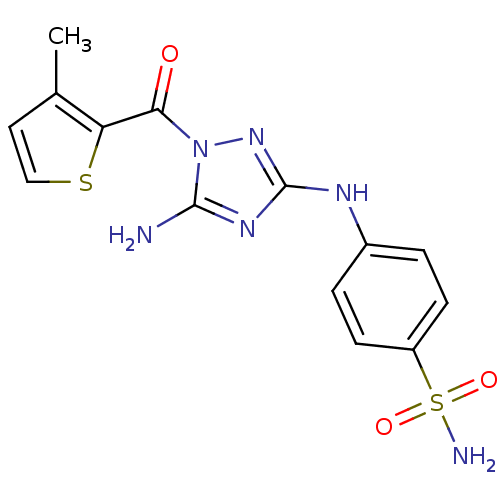
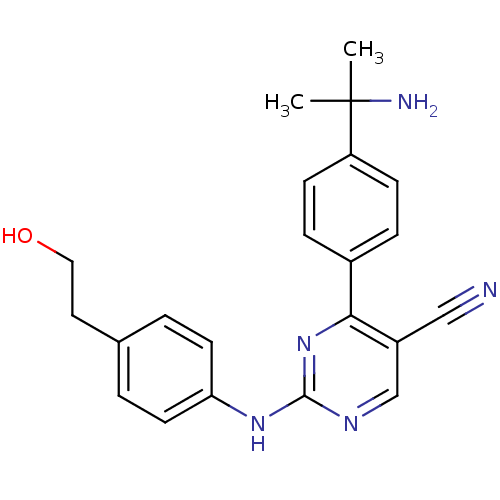
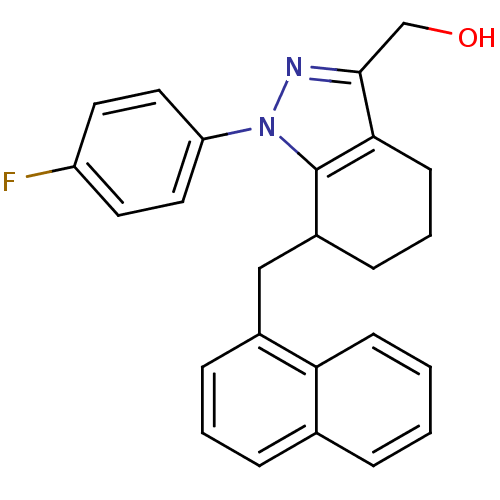
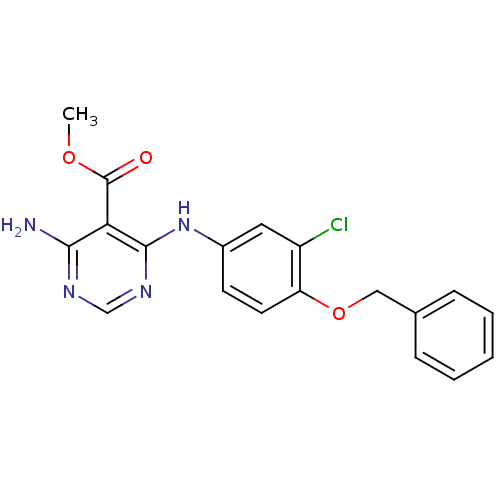
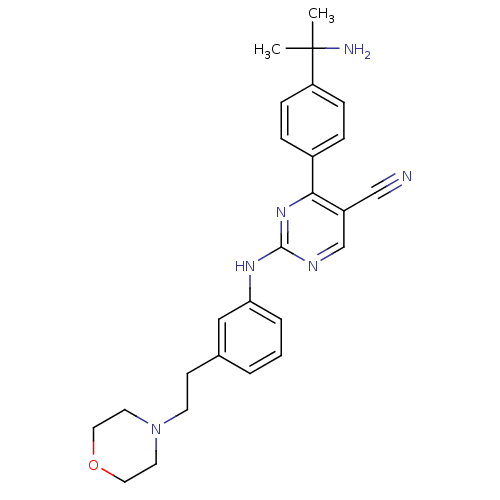
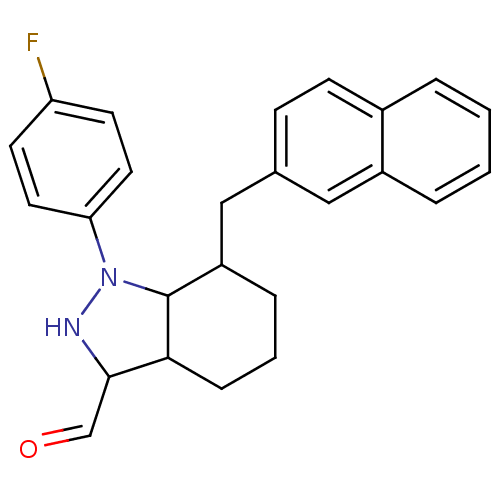
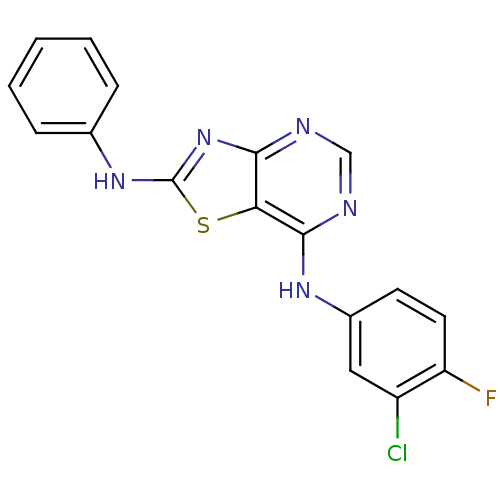
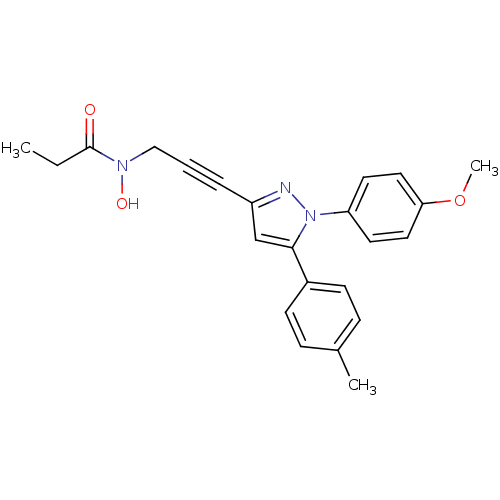
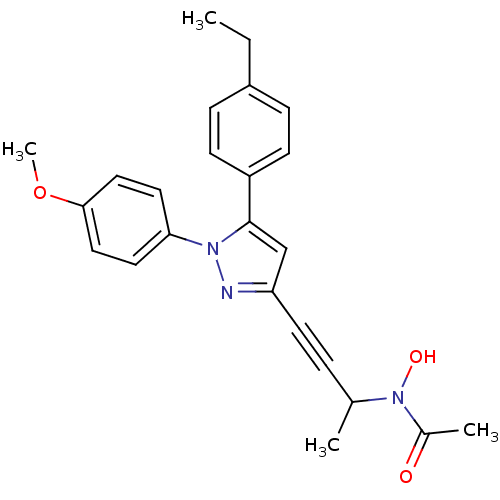
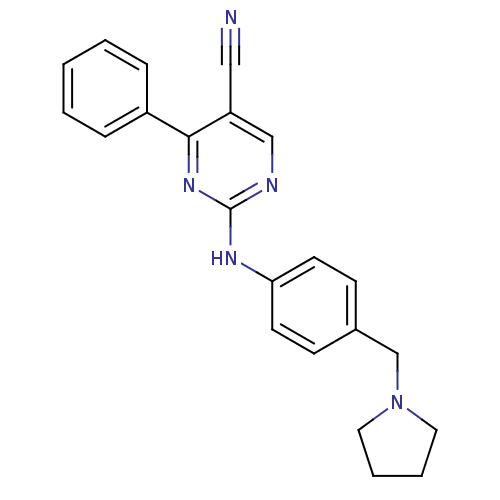
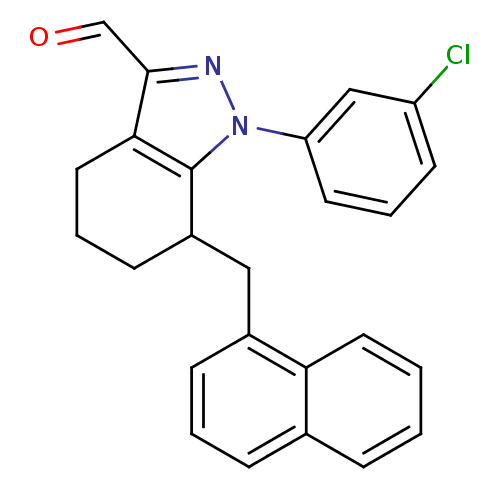
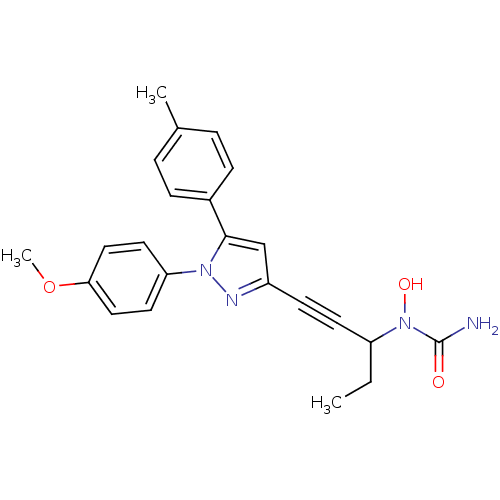
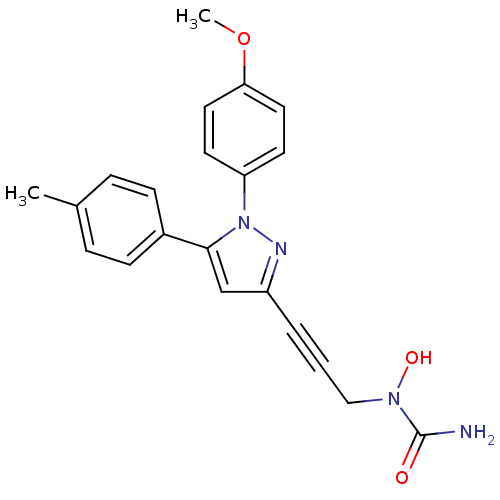
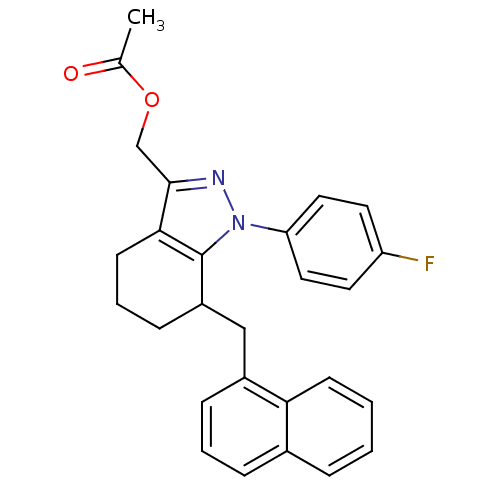
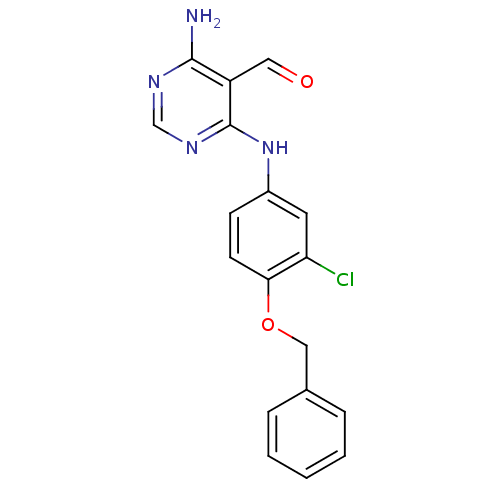
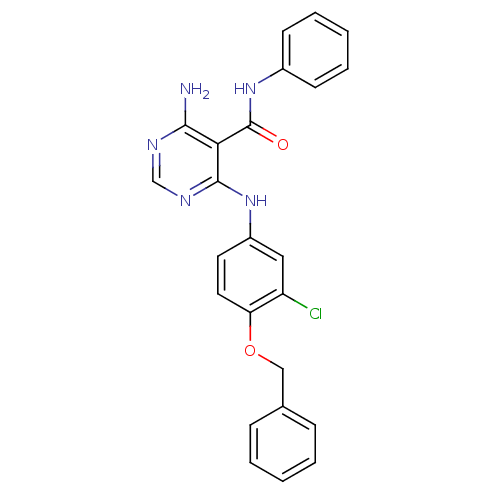
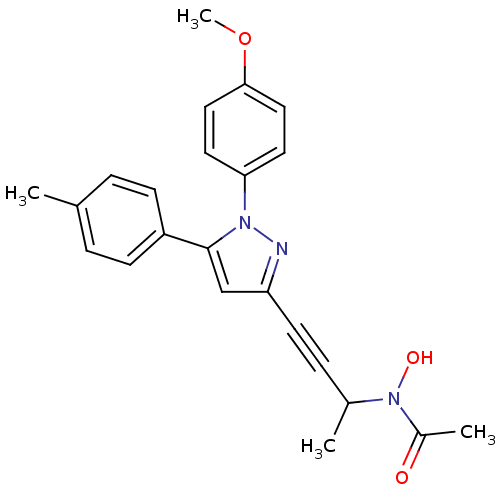
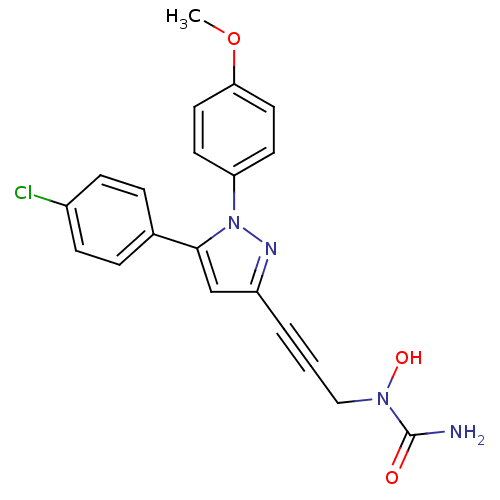
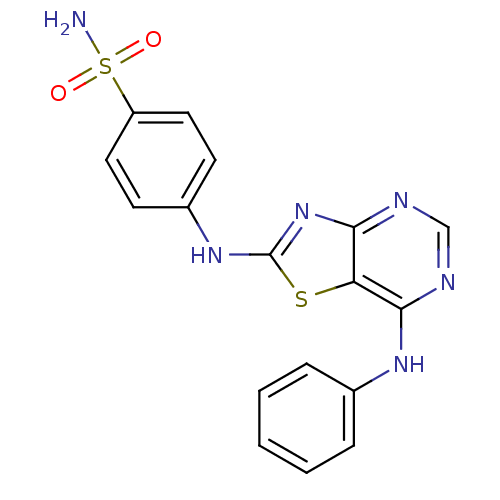
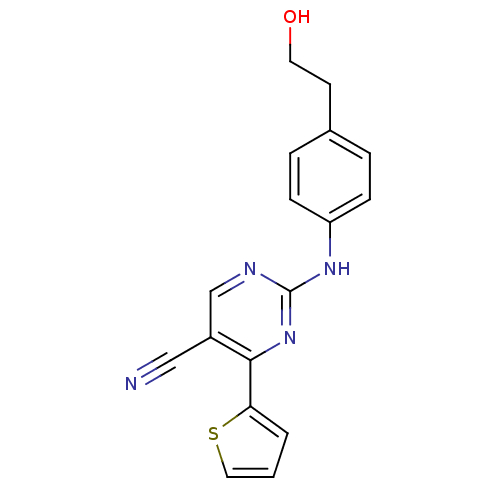
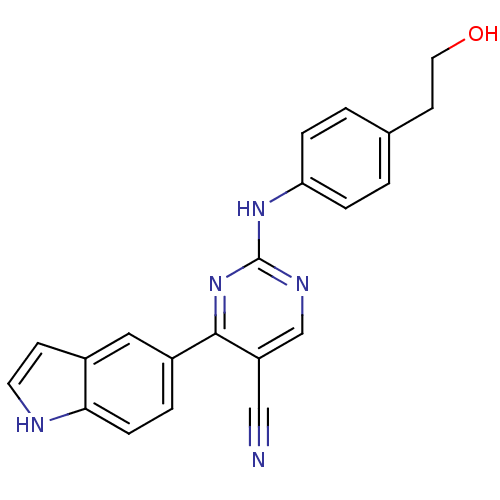
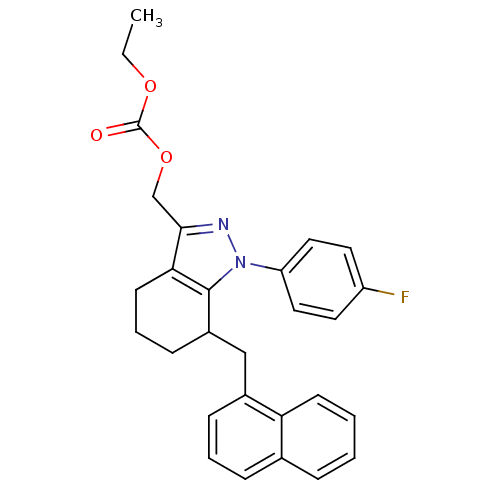
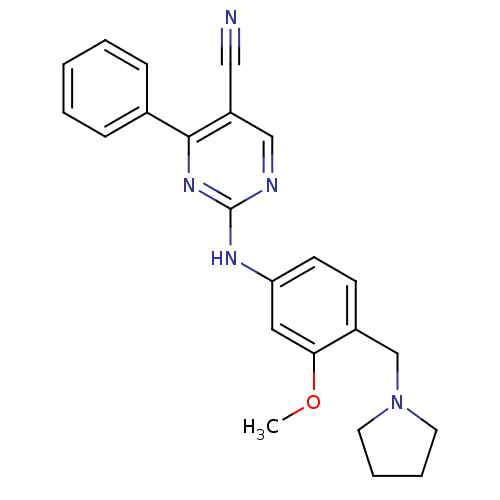
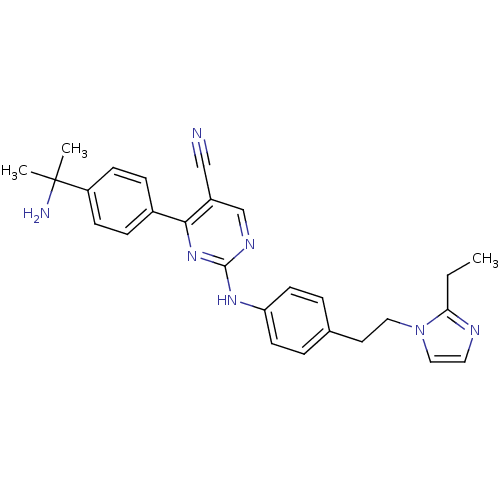
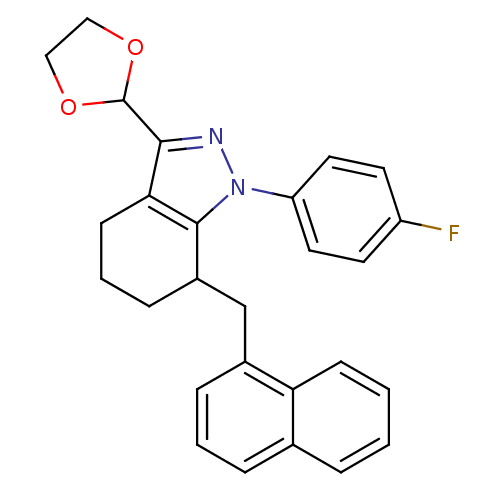
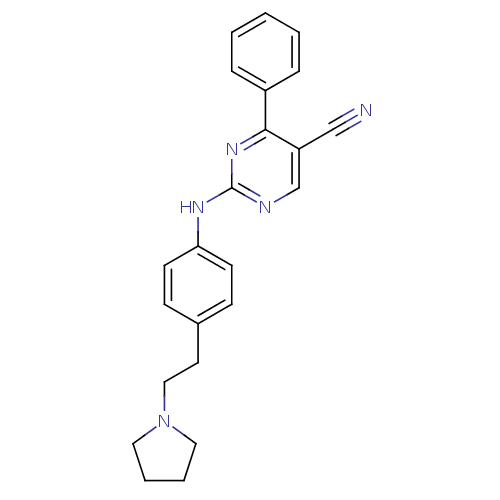
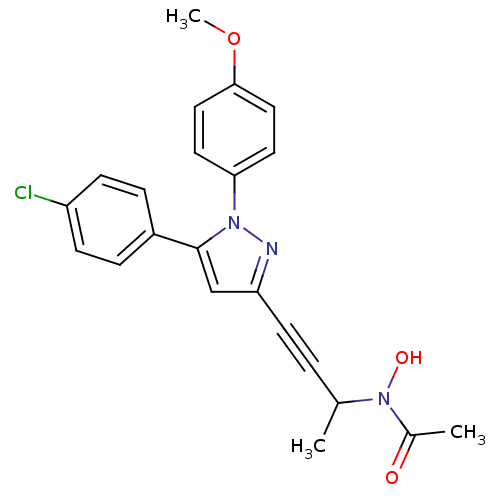
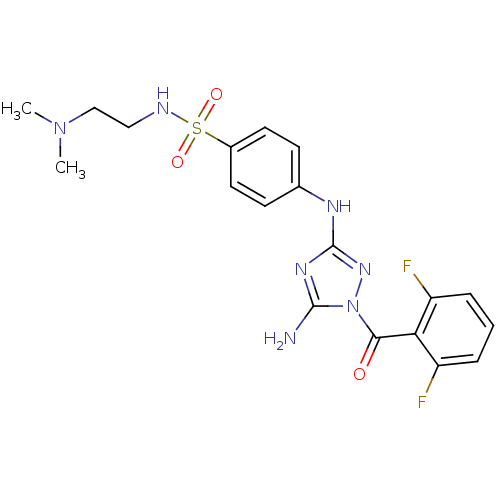
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta [7-25](Oryctolagus cuniculus (rabbit))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 31nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 32nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetGlycogen synthase kinase-3 beta [7-25](Oryctolagus cuniculus (rabbit))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 41nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 41nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 43nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetCyclin-dependent kinase/G2/mitotic-specific cyclin- 1(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 45nMpH: 8.0 T: 2°CAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated ...More data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibitory activity against Cyclooxygenase (COX) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 51nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 52nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
Affinity DataIC50: 52nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 54nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 55nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 59nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibitory activity against cyclooxygenase (COX) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibitory activity against Cyclooxygenase (COX) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 61nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
Affinity DataIC50: 69nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Inhibitory activity against 5-lipoxygenase(5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
Affinity DataIC50: 73nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 76nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 76nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibitory activity against 5-lipoxygenase(5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetReceptor tyrosine-protein kinase erbB-2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of HER2 (unknown origin)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataIC50: 82nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 82nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 90nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 90nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
Affinity DataIC50: 90nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 91nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 94nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
Affinity DataIC50: 97nMAssay Description:Binding affinity determined by ability to displace [3H]-R5020 radioligand using uterine progesterone receptor obtained from estrogen-primed rabbitMore data for this Ligand-Target Pair
TargetReceptor tyrosine-protein kinase erbB-2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of HER2 (unknown origin)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibitory activity against 5-lipoxygenase(5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataIC50: 110nMAssay Description:Inhibition of VEGFR2 activityMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1/2(RAT)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 110nMAssay Description:Inhibitory activity against cyclooxygenase (COX) in intact rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Rattus norvegicus)
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
The R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 110nMAssay Description:Inhibitory activity against 5-lipoxygenase (5-LO) using broken rat barophilic leukemia cells (RBL-1)More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 4/G1/S-specific cyclin-D1(Mus musculus (mouse))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 110nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 4/G1/S-specific cyclin-D1(Mus musculus (mouse))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 120nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Rattus norvegicus (rat))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 130nMAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated...More data for this Ligand-Target Pair
TargetCyclin-dependent kinase/G2/mitotic-specific cyclin- 1(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 130nMpH: 8.0 T: 2°CAssay Description:The enzyme was assayed with a biotinylated peptide substrate and test compounds in the presence of 10 uM ATP/[gamma-33P]ATP in a streptavidin coated ...More data for this Ligand-Target Pair