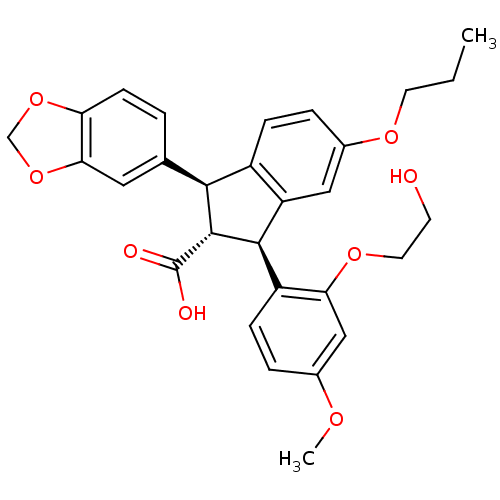
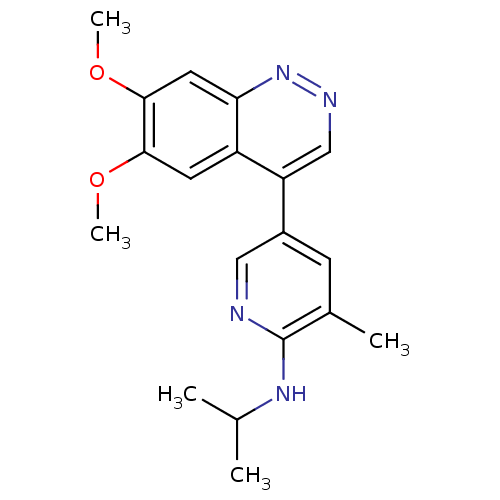
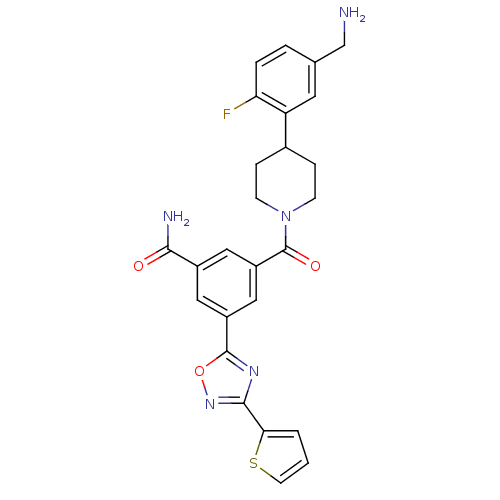
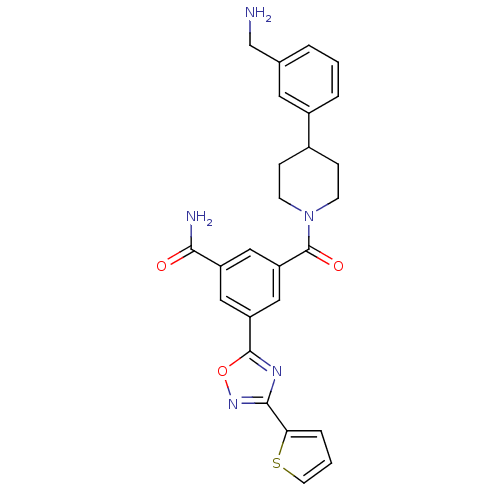
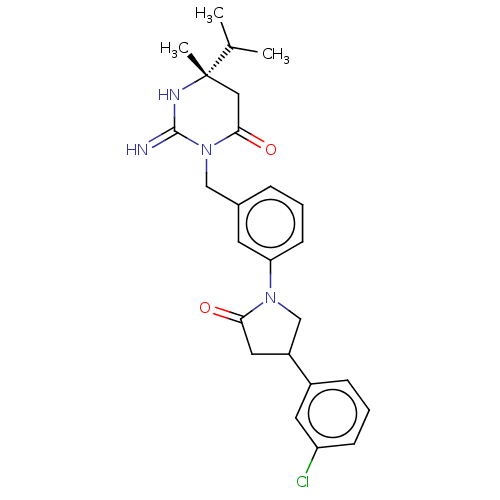
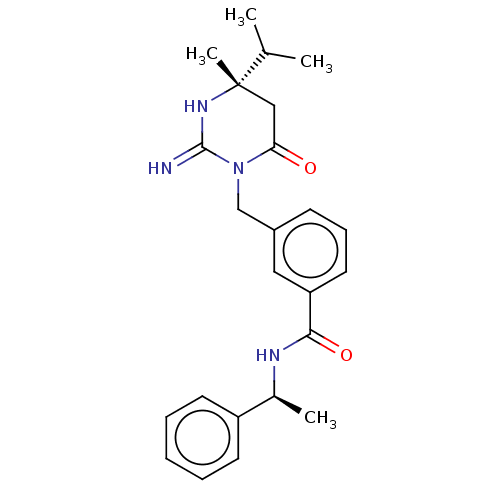
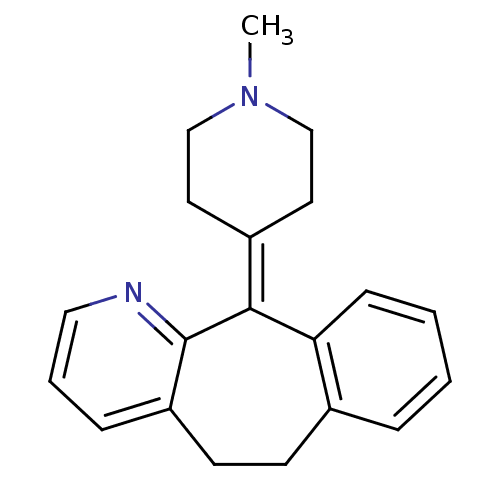
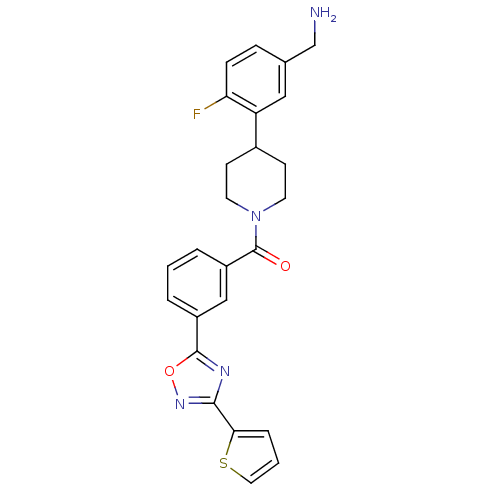
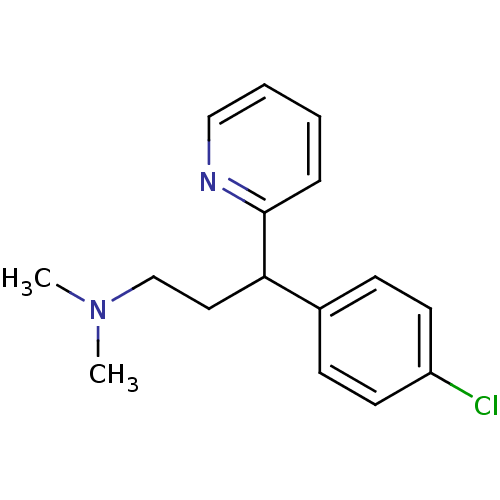
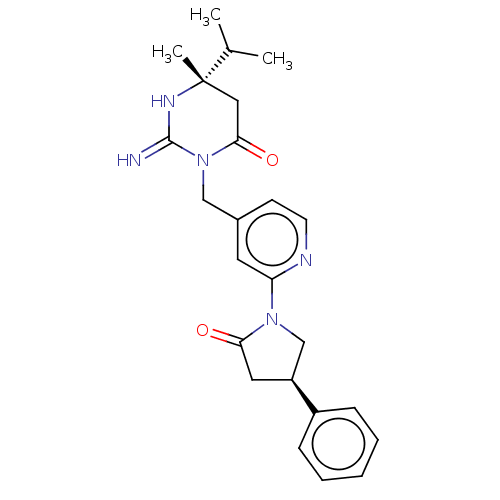
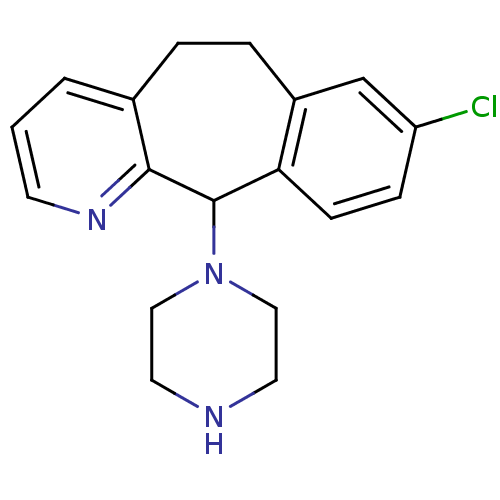
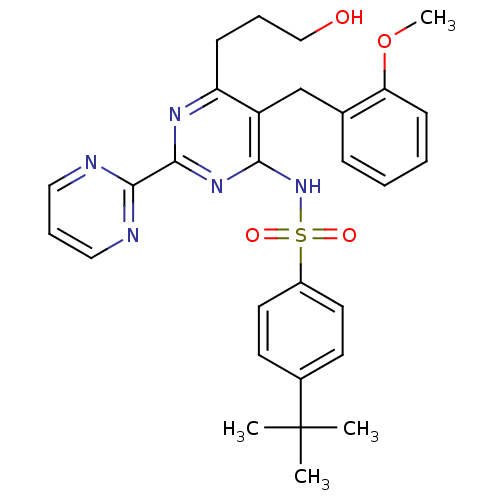
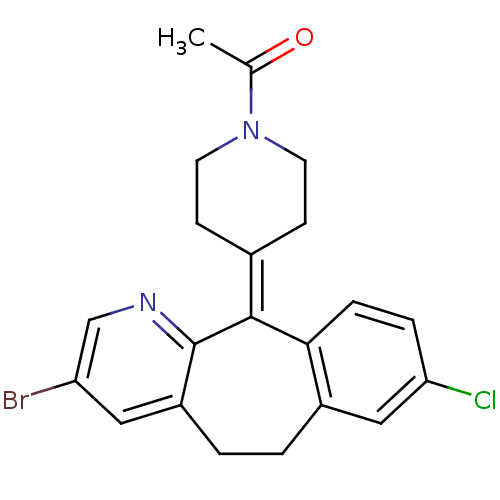
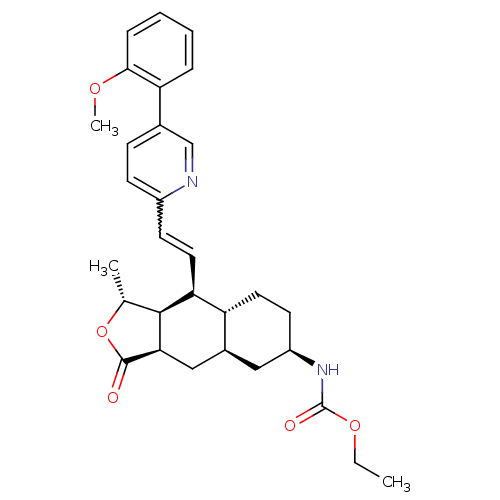
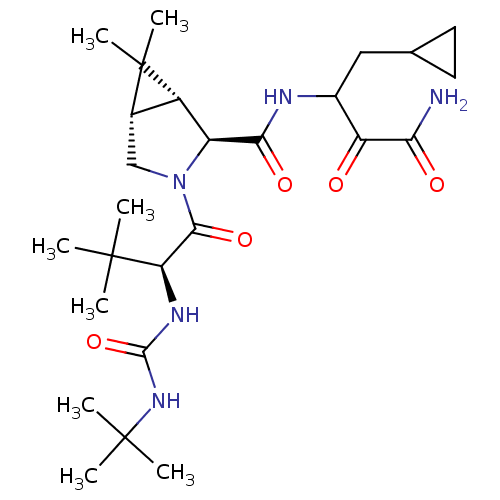
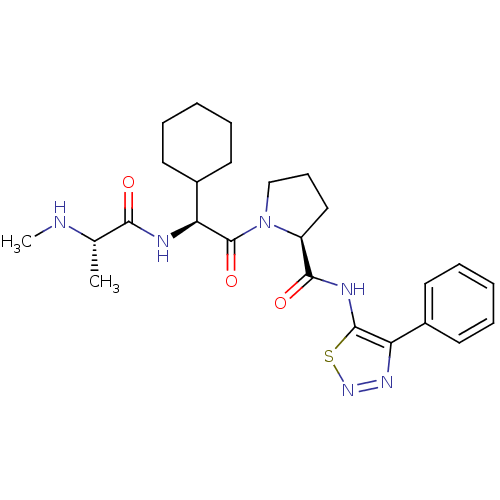
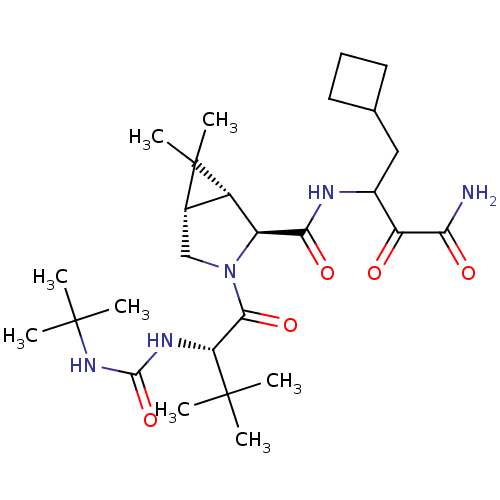
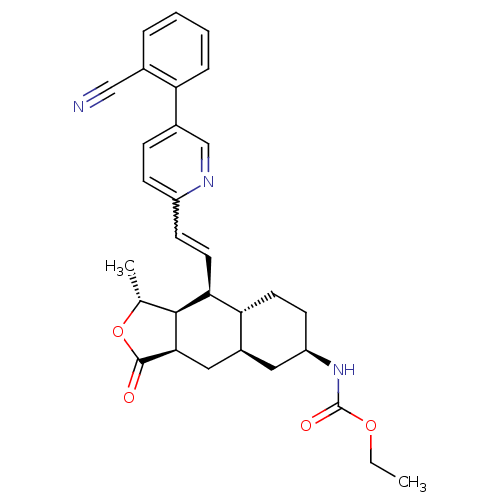
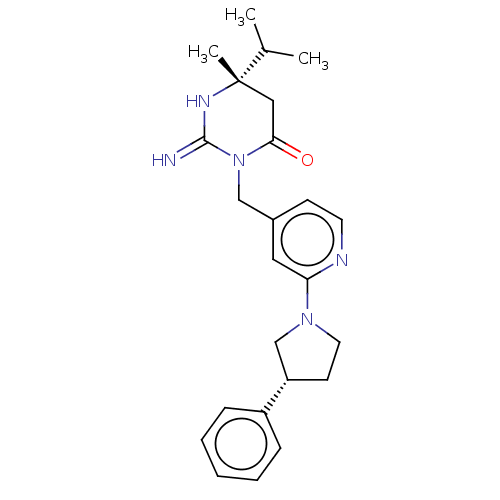
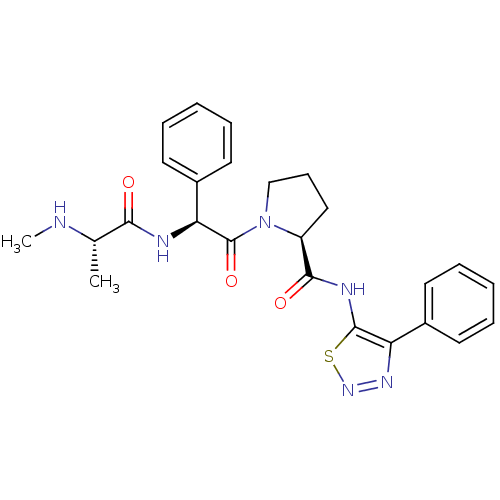
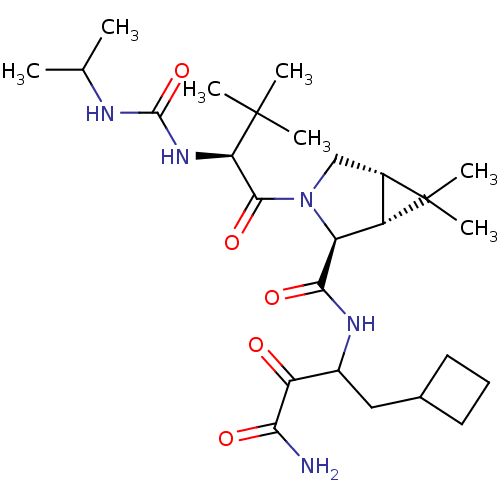
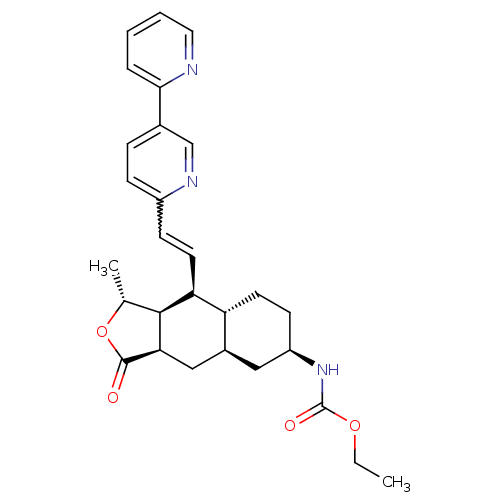
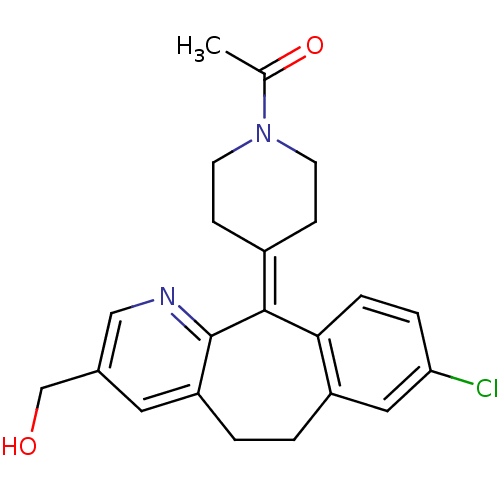
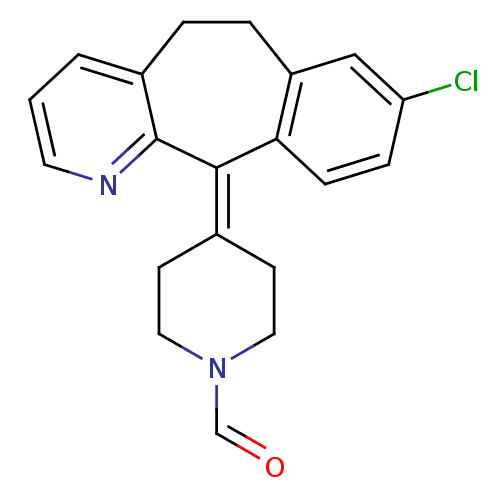
Report error Found 2533 with Last Name = 'wong' and Initial = 'j'
Affinity DataKi: 0.0340nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0690nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.130nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.130nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
Affinity DataKi: 0.170nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.170nMAssay Description:Displacement of [3H]5-(6,7-dimethoxycinnolin-4-yl)-N-isopropyl-3-methylpyridin-2-amine from PDE10A in Sprague-Dawley rat striatumMore data for this Ligand-Target Pair
Affinity DataKi: 0.170nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
Affinity DataKi: 0.430nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.430nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
Affinity DataKi: 0.460nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
Affinity DataKi: 1.18nMAssay Description:Displacement of [3H]5-(6,7-dimethoxycinnolin-4-yl)-N-isopropyl-3-methylpyridin-2-amine from PDE10A in Sprague-Dawley rat striatumMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Inhibitory activity against human mast cell tryptase betaMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Inhibitory activity against human mast cell tryptase betaMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 3.90nMAssay Description:Binding affinity to histamine H1 receptor in rat brain membranes was evaluated using [3H]-pyrilamine as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 4.30nMAssay Description:Inhibitory activity against human mast cell tryptase betaMore data for this Ligand-Target Pair
Affinity DataKi: 5.40nMAssay Description:Inhibitory activity of the compound against human Endothelin B receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:Binding affinity against Histamine H1 receptor using receptor binding assay in rat brain membranesMore data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:Binding affinity to histamine H1 receptor in rat brain membranes was evaluated using [3H]-pyrilamine as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 6nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 6.40nMAssay Description:Binding affinity against Histamine H1 receptor using receptor binding assay in rat brain membranesMore data for this Ligand-Target Pair
Affinity DataKi: 6.5nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 6.5nMAssay Description:Binding affinity against Histamine H1 receptor using receptor binding assay in rat brain membranesMore data for this Ligand-Target Pair
TargetDihydrofolate reductase(Human)
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Affinity DataKi: 7.70nMAssay Description:Binding affinity to human recombinant DHFR expressed in Escherichia coli BL21(DE3) by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 8nMAssay Description:Displacement of high affinity TRAP form human platelet PAR1More data for this Ligand-Target Pair
Affinity DataKi: 8nMAssay Description:Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I...More data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Ability to displace [3H]-pyrilamine from H1 receptor in rat brain membrane.More data for this Ligand-Target Pair
TargetDihydrofolate reductase(Human)
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Affinity DataKi: 10.2nMAssay Description:Binding affinity to human recombinant DHFR expressed in Escherichia coli BL21(DE3) by competitive binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Displacement of high affinity TRAP form human platelet PAR1More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I...More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from MLXBIR3SG after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I...More data for this Ligand-Target Pair
Affinity DataKi: 14.7nMAssay Description:Inhibitory activity of the compound against human Endothelin B receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Displacement of high affinity TRAP form human platelet PAR1More data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from MLXBIR3SG after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I...More data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from cIAP1 BIR3 domain after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:Inhibition of renin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 19nMAssay Description:Displacement of high affinity TRAP form human platelet PAR1More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Ability to displace [3H]-pyrilamine from H1 receptor in rat brain membrane.More data for this Ligand-Target Pair
Affinity DataKi: 21nMAssay Description:Binding affinity to histamine H1 receptor in rat brain membranes was evaluated using [3H]-pyrilamine as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 25nMAssay Description:Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I...More data for this Ligand-Target Pair
Affinity DataKi: 28nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from XIAP BIR3 domain after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from MLXBIR3SG after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Displacement of 5-FAM-conjugated AVP-diPhe-FAM from MLXBIR3SG after 30 mins by fluorescence polarization-based competition assayMore data for this Ligand-Target Pair
TargetDihydrofolate reductase(Human)
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Kenya Medical Research Institute/Wellcome Trust Collaborative Research Program
Curated by ChEMBL
Affinity DataKi: 30.8nMAssay Description:Binding affinity to human recombinant DHFR expressed in Escherichia coli BL21(DE3) by competitive binding assayMore data for this Ligand-Target Pair
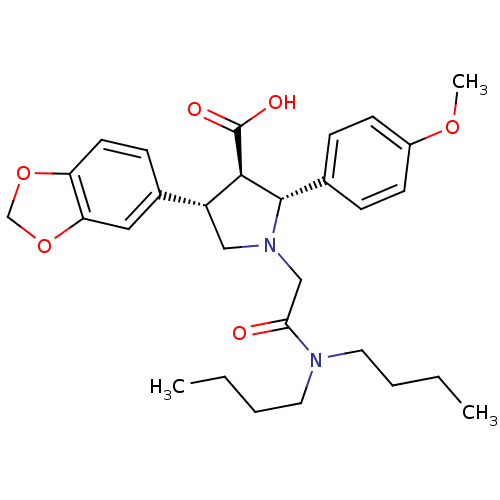
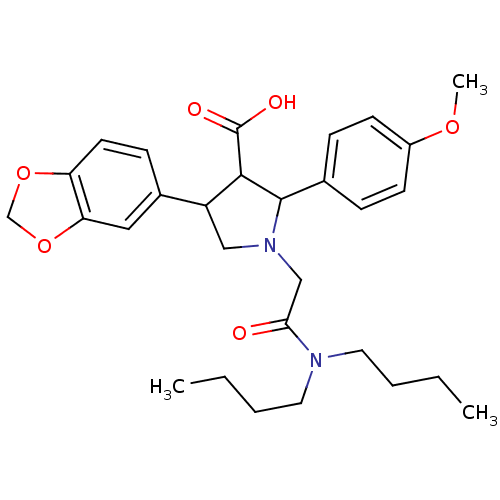

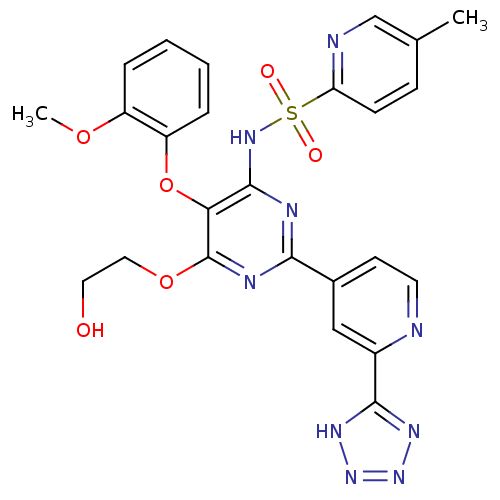
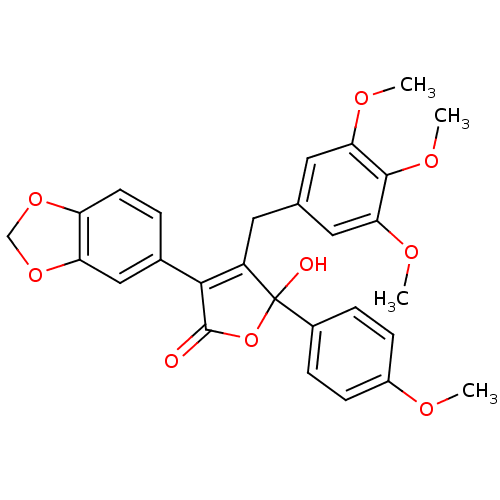
 3D Structure (crystal)
3D Structure (crystal)