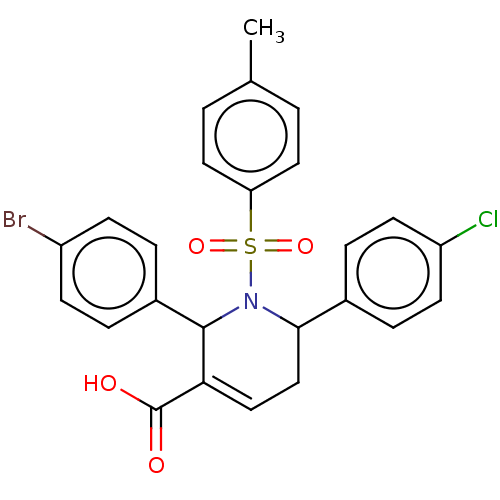
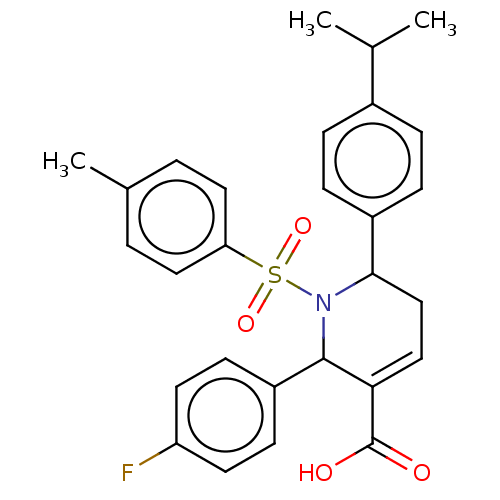
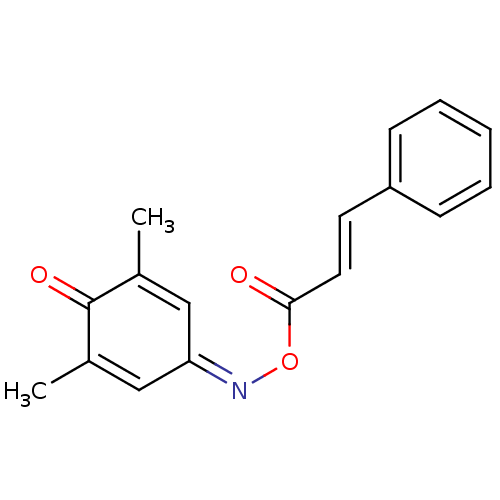
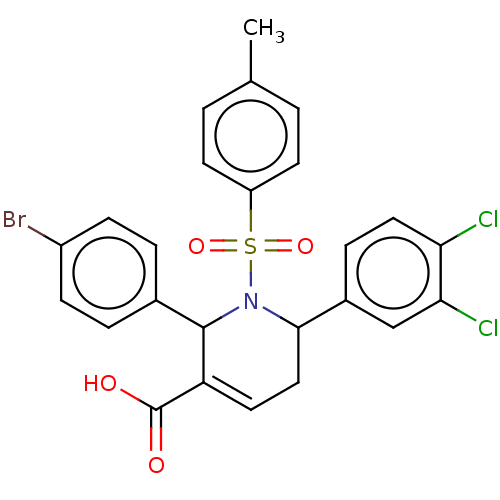
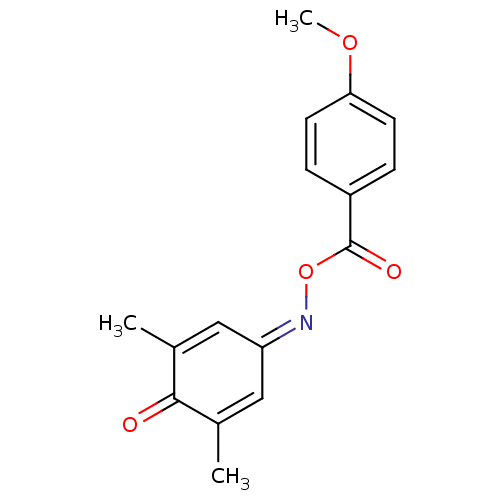
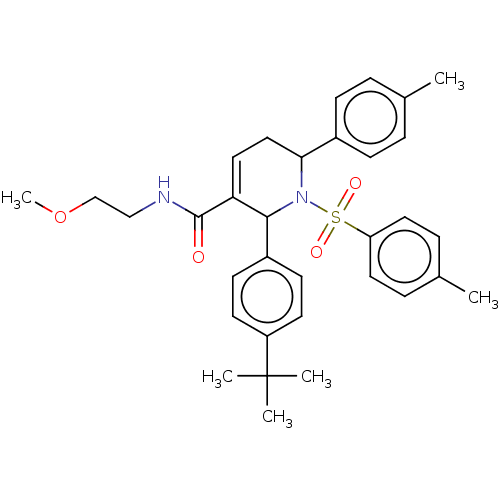
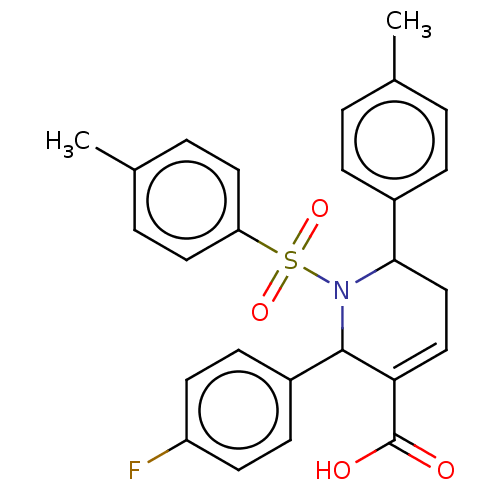
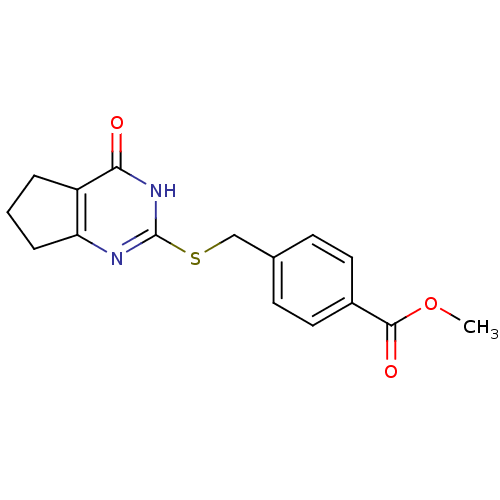
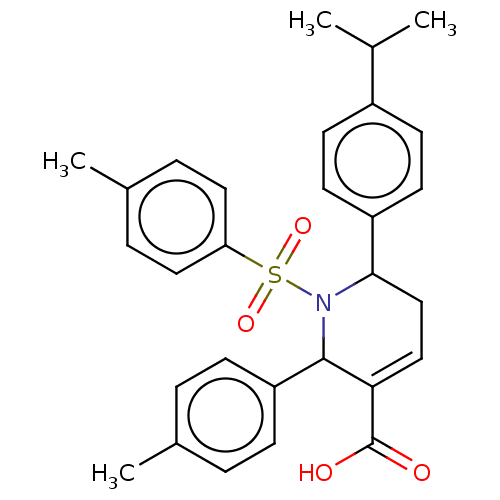
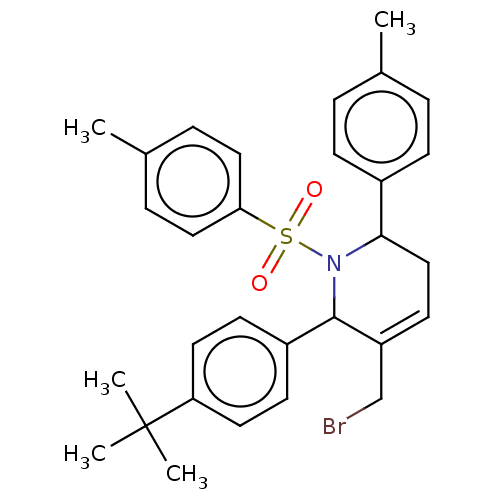
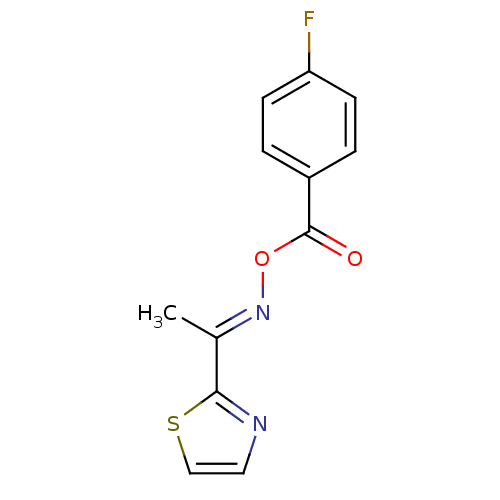
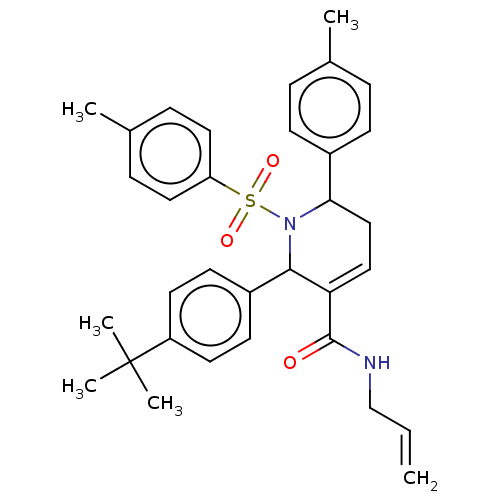
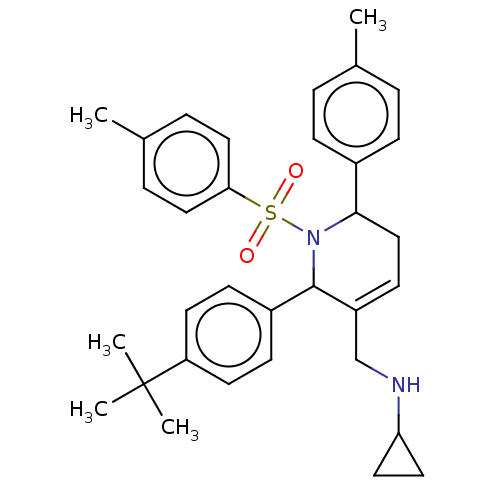
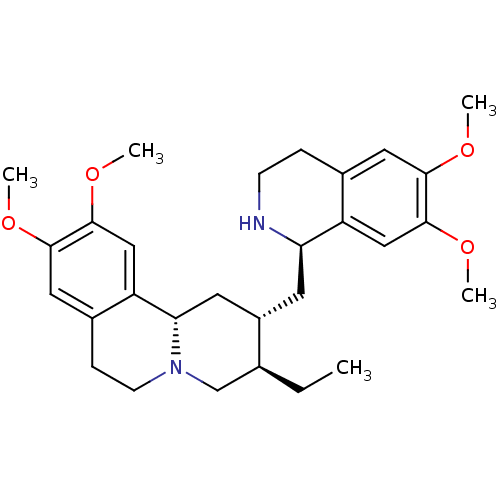
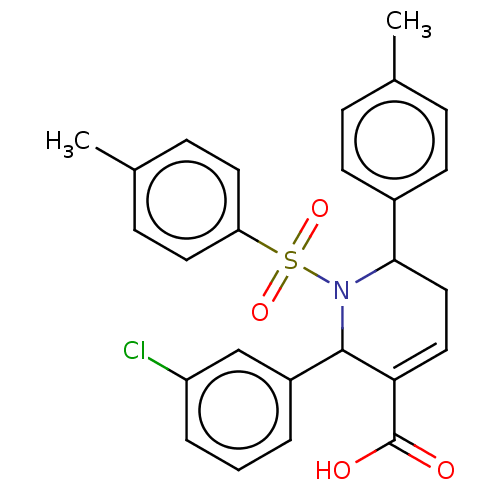
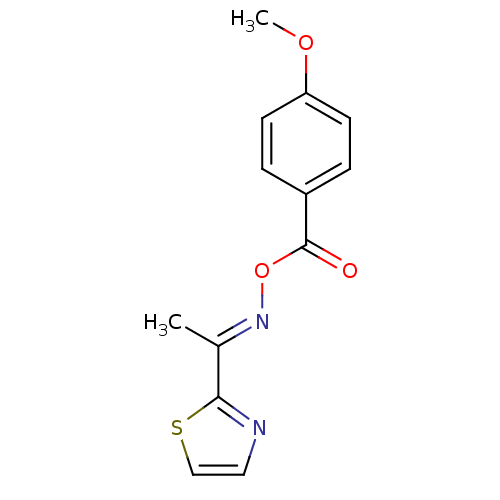
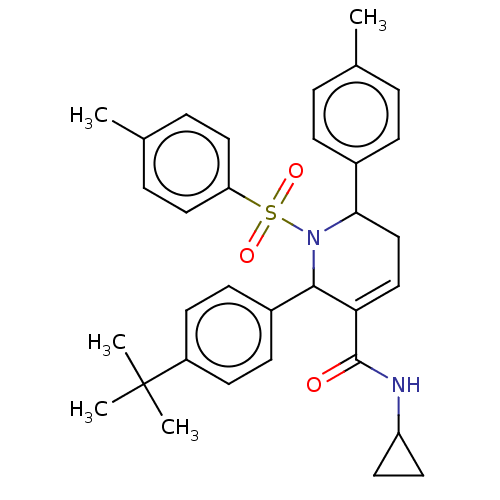
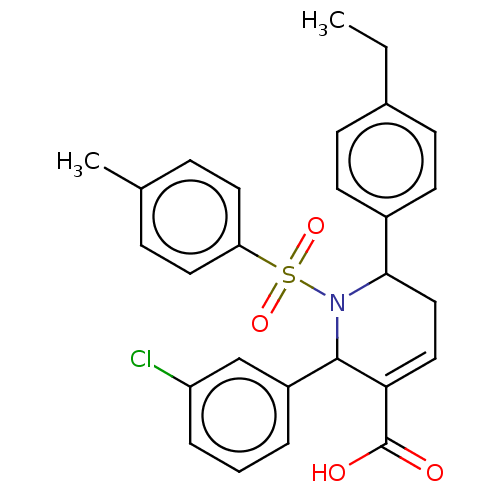
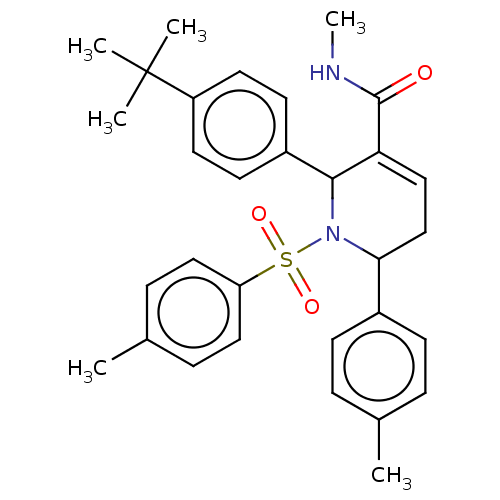
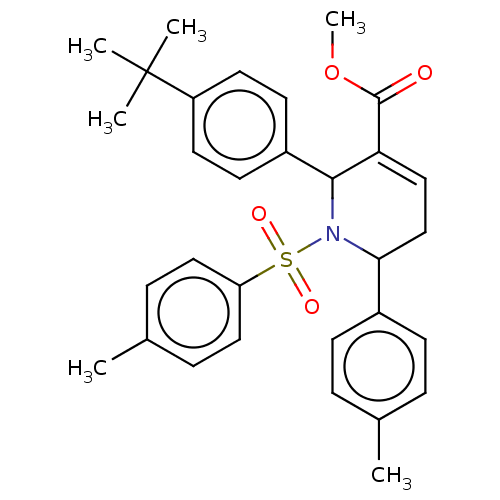
Report error Found 146 with Last Name = 'spicer' and Initial = 'tp'
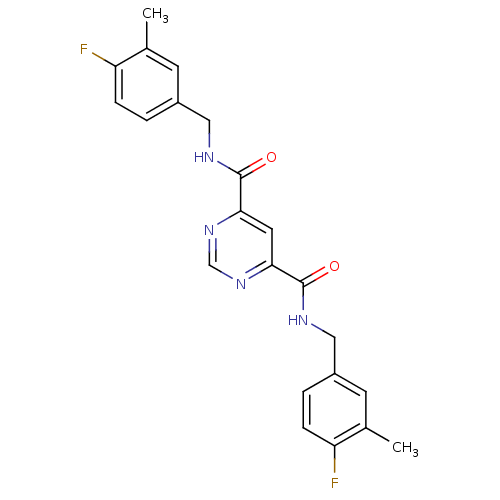
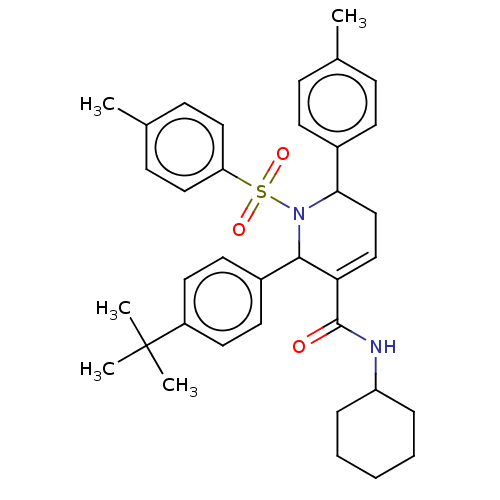
Affinity DataIC50: 14nMAssay Description:Non-competitive inhibition of full-length recombinant human MMP-13 assessed as fTHP-15 substrate hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 70nMAssay Description:Inhibition of full-length recombinant human MMP-13 assessed as bovine type-2 collagen hydrolysis after 18 hrs by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Non-competitive inhibition of full-length recombinant human MMP-13 assessed as fTHP-15 substrate hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibition of full-length recombinant human MMP-13 assessed as bovine type-2 collagen hydrolysis after 18 hrs by ELISAMore data for this Ligand-Target Pair
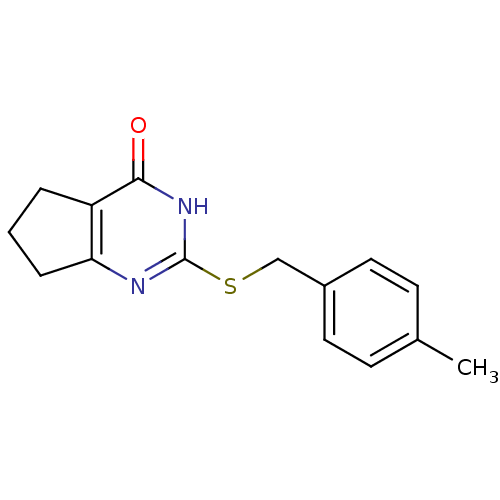
Affinity DataIC50: 640nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Inhibition of full-length recombinant human MMP-13 assessed as bovine type-2 collagen hydrolysis after 18 hrs by ELISAMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
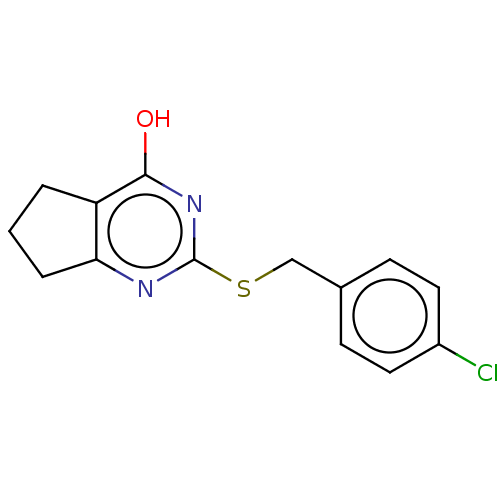
Affinity DataIC50: 800nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 1.10E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 1.10E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 1.40E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from recombinant RBBP9 transfected in mouse brain membrane proteomes after 30 mins by SDS-PAGE gel fluoresc...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 1.70E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 2.00E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of full-length recombinant human MMP-13 assessed as bovine type-2 collagen hydrolysis after 18 hrs by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+3nMAssay Description:Non-competitive inhibition of full-length recombinant human MMP-13 assessed as fTHP-15 substrate hydrolysisMore data for this Ligand-Target Pair
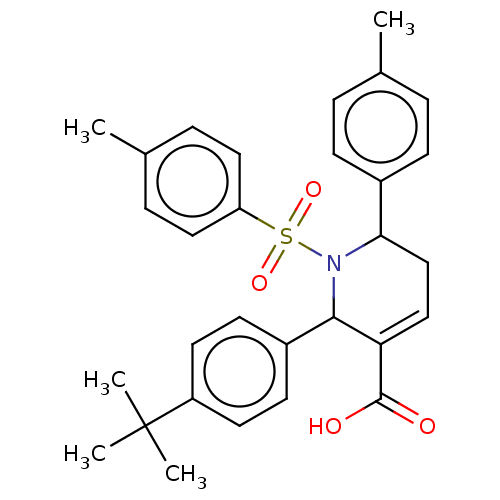
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin) using (S)-mephentoin substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin) using dextromethophan substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP2C9 (unknown origin) using diclofenac substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP2C8 (unknown origin) using amodiaquine substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP2B6 (unknown origin) using bupropion substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin) using tacrin substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using midazolam substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.50E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using testosterone substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Non-competitive inhibition of full-length recombinant human MMP-13 assessed as fTHP-15 substrate hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+3nMAssay Description:Non-competitive inhibition of full-length recombinant human MMP-13 assessed as fTHP-15 substrate hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin) using tacrin substrateMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 4.50E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 4.60E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 5.70E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.50E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin) using (S)-mephentoin substrateMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 7.50E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 7.60E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 7.80E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibition of CYP2C19 (unknown origin) using (S)-mephentoin substrateMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 8.20E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 8.60E+3nMAssay Description:Inhibition of CYP1A2 (unknown origin) using tacrin substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 8.80E+3nMAssay Description:Inhibition of CYP2C9 (unknown origin) using diclofenac substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 9.20E+3nMAssay Description:Displacement of fluorophosphate-rhodamine from RBBP9 transfected in human HEK293T cells proteomes after 30 mins by SDS-PAGE gel fluorescence assayMore data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 9.30E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: 9.40E+3nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: >1.00E+4nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: >1.00E+4nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
TargetPlatelet-activating factor acetylhydrolase IB subunit alpha2(Mus musculus (Mouse))
University of California Berkeley
University of California Berkeley
Affinity DataIC50: >1.00E+4nMpH: 7.5Assay Description:For in vitro experiments, proteomes were diluted to 1 mg/mL in PBS (pH 7.5, 50 μL total reaction volume), doped with 1 μM recombinant PAFAH...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin) using diclofenac substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.00E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin) using testosterone substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.00E+4nMAssay Description:Inhibition of CYP2C8 (unknown origin) using amodiaquine substrateMore data for this Ligand-Target Pair
Affinity DataIC50: >2.00E+4nMAssay Description:Inhibition of CYP2B6 (unknown origin) using bupropion substrateMore data for this Ligand-Target Pair
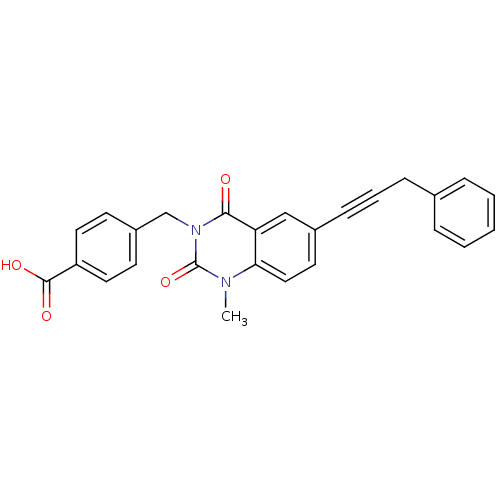
 3D Structure (crystal)
3D Structure (crystal)