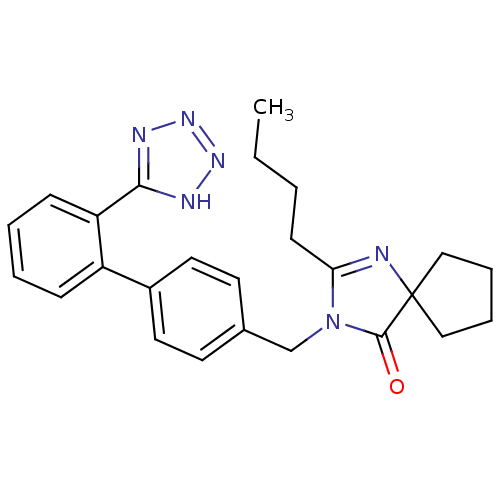
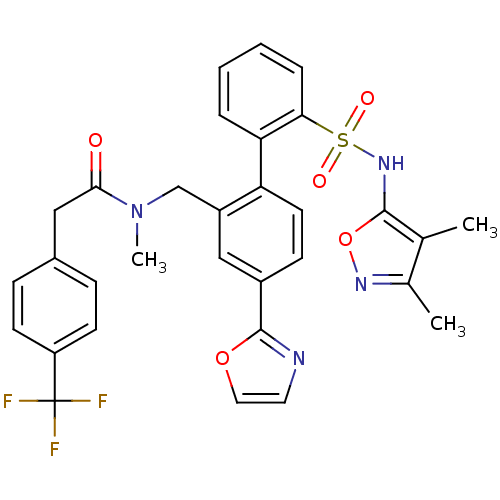
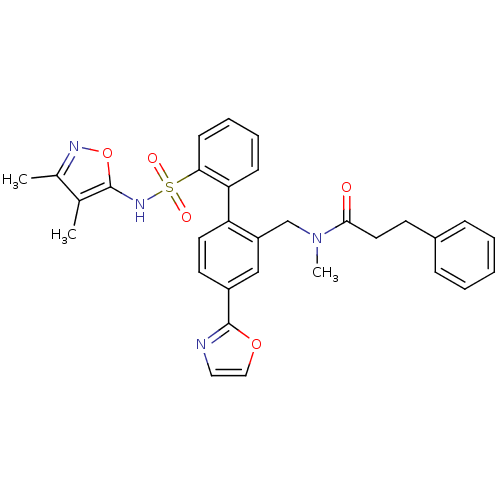
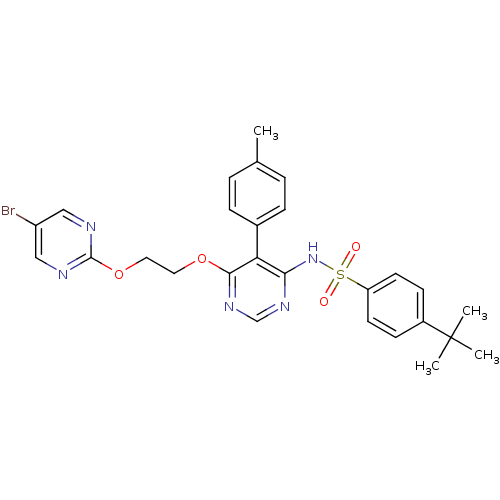
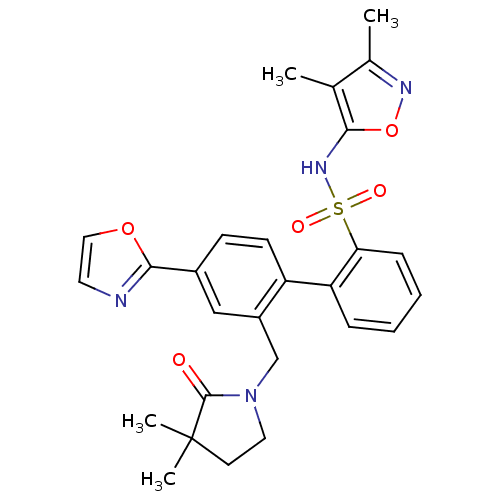
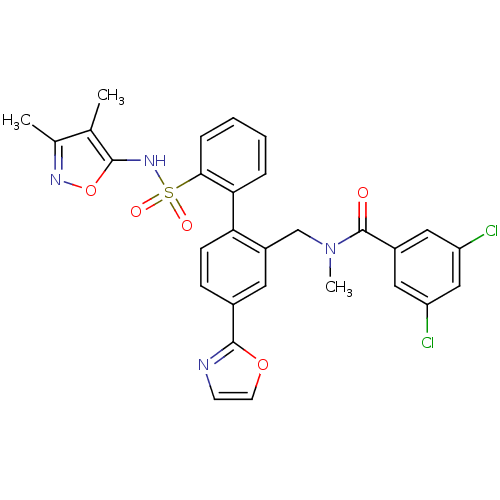
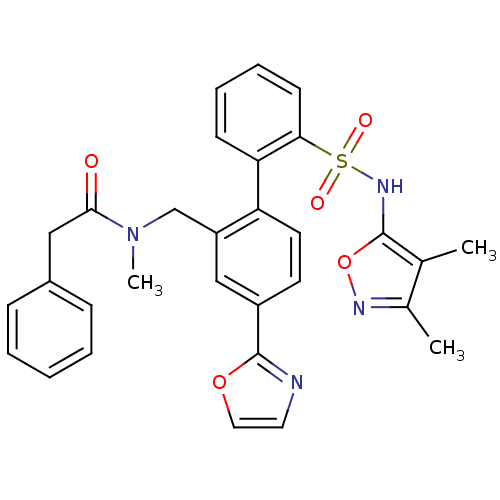
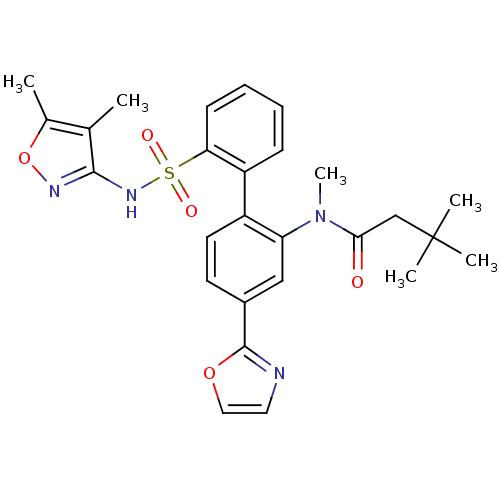
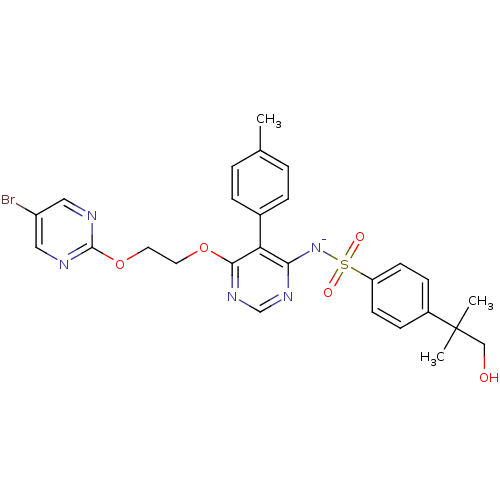
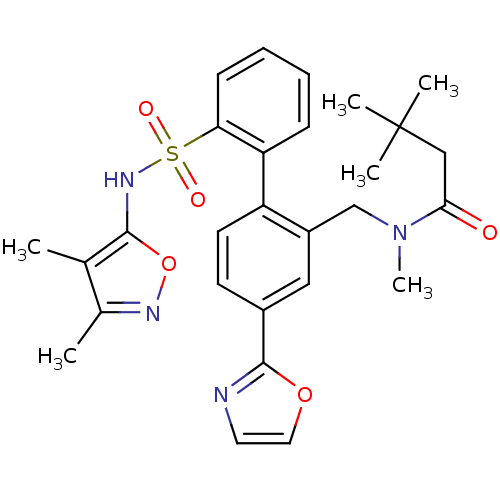
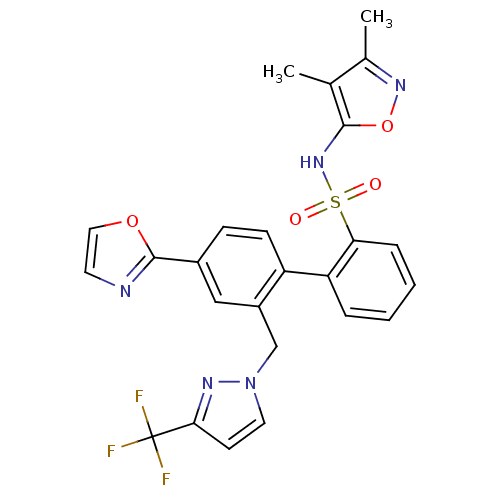
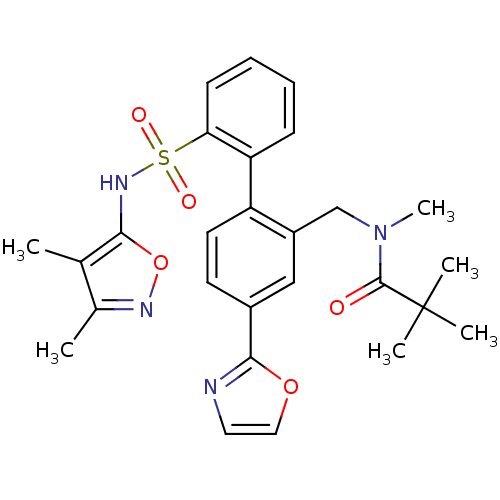
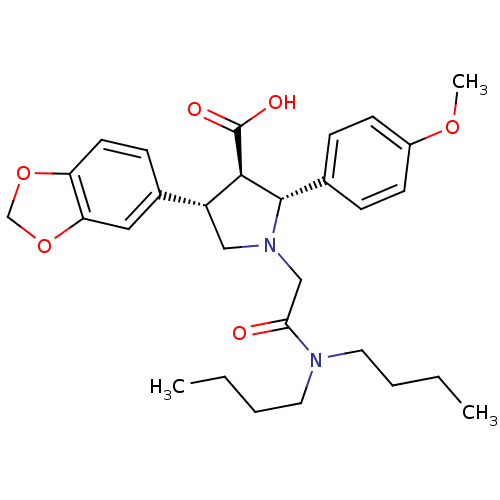
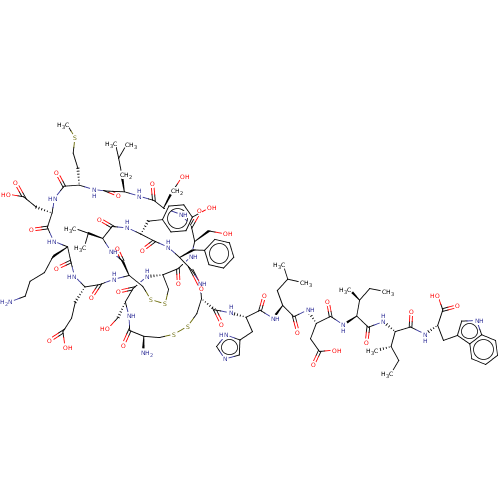
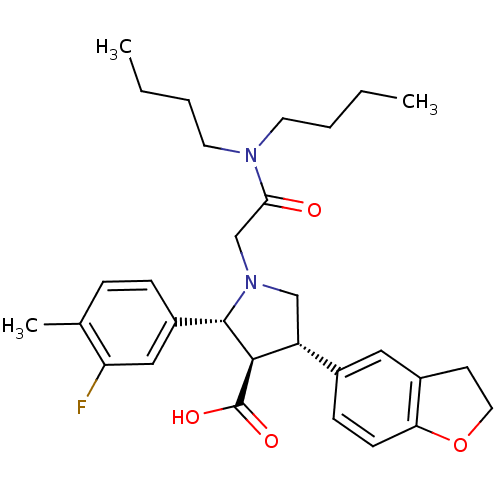
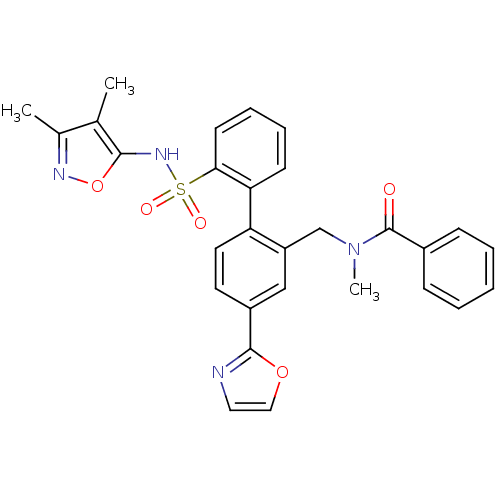
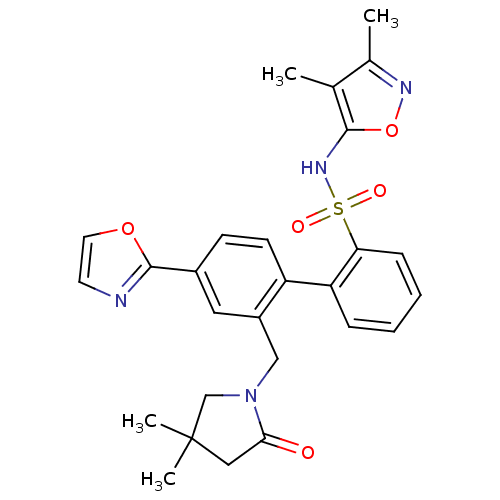
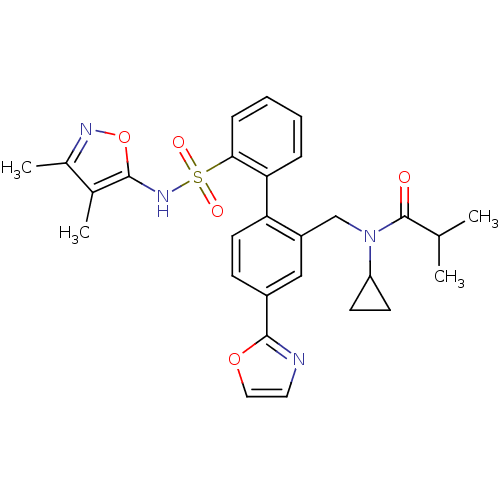
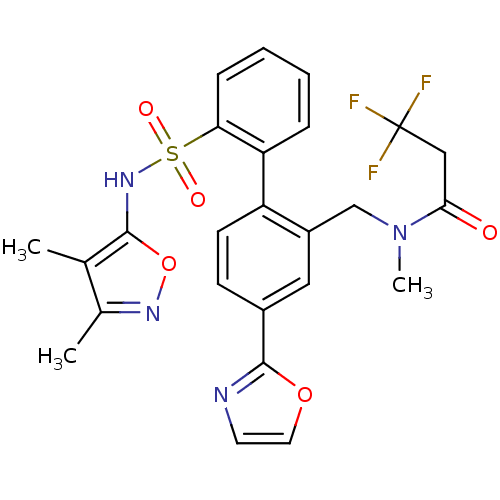
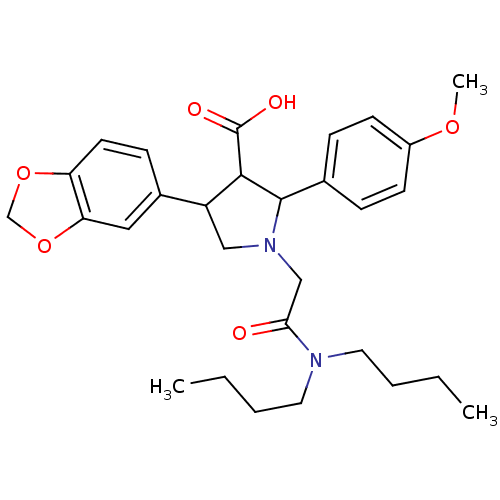
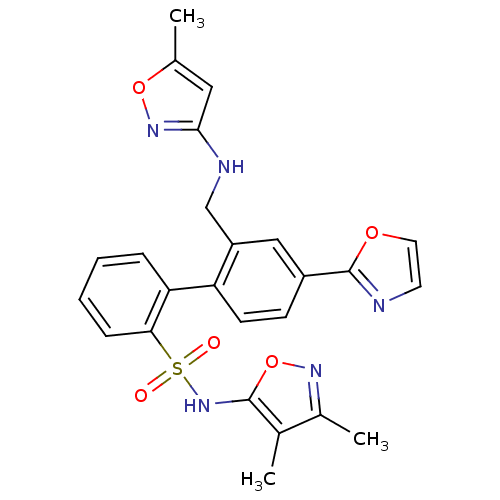
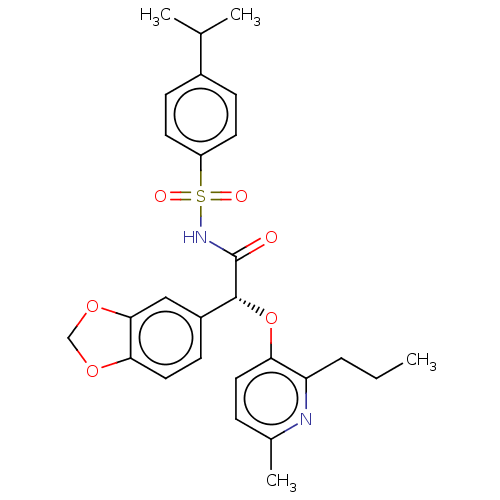
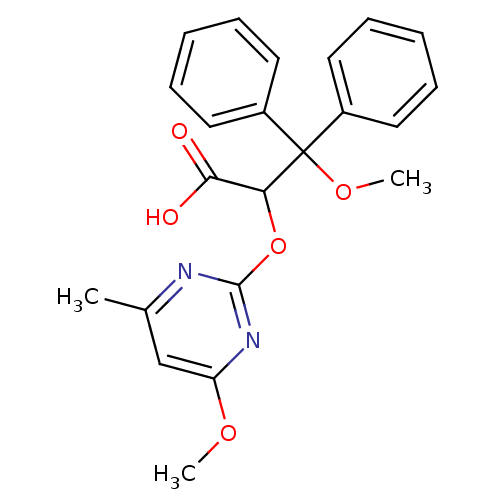
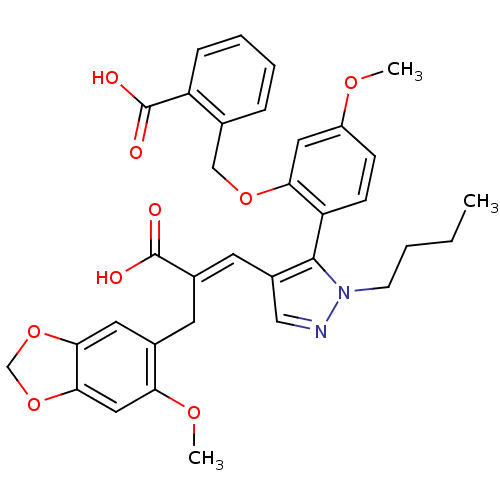
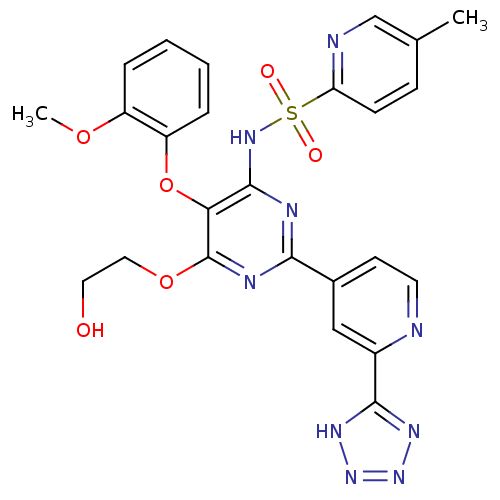
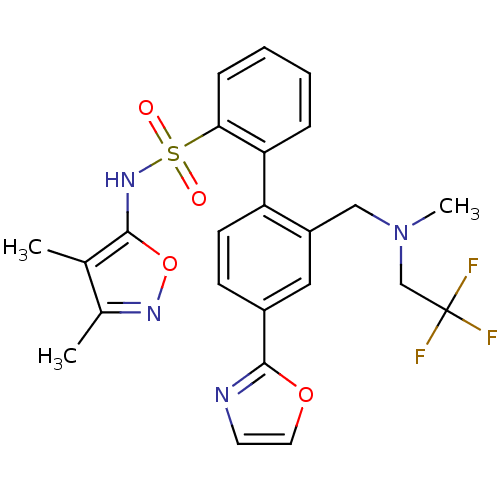
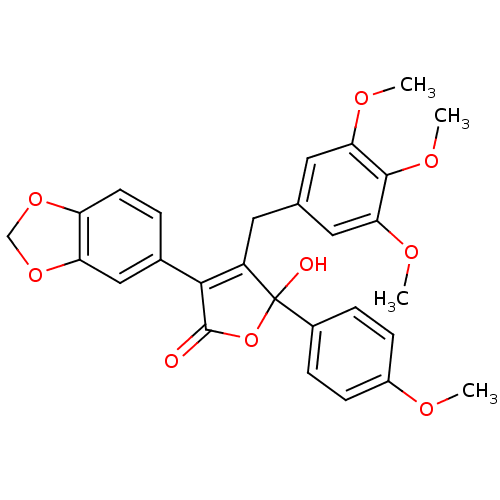
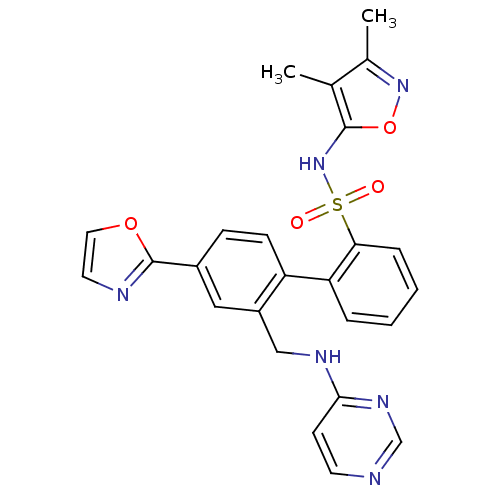
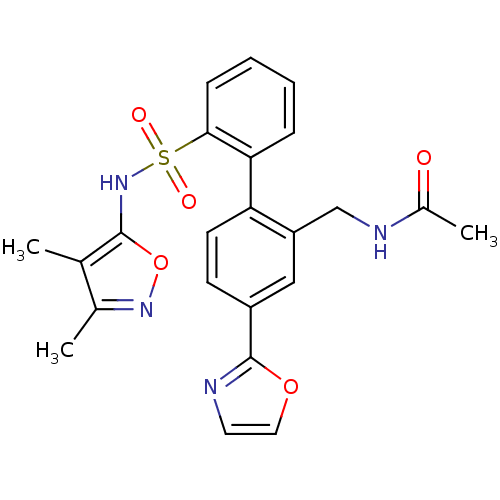
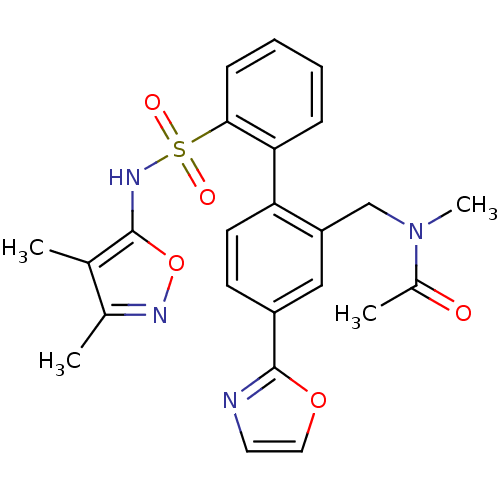
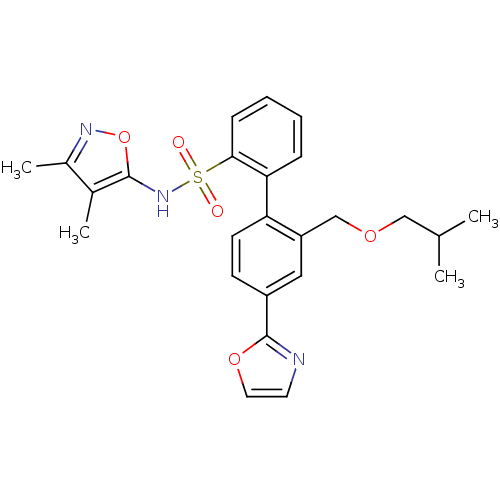
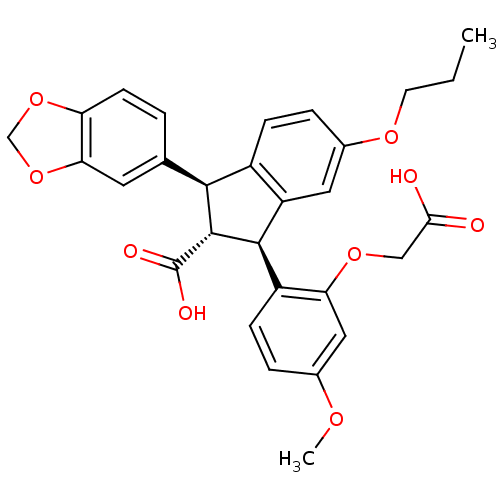
Report error Found 3252 Enz. Inhib. hit(s) with Target = 'Endothelin-1 receptor'
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: >0.000100nMAssay Description:Compound was evaluated for its binding affinity towards human Endothelin A receptorMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00100nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00400nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00400nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00420nMAssay Description:Ability to inhibit [125I]ET1 binding to human cloned endothelin A receptor expressed on CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00500nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00500nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.00900nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Inhibitory concentration against Endothelin A receptorMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0150nMAssay Description:Ability to inhibit [125I]ET1 binding to human cloned endothelin A receptor expressed on CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0300nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0340nMAssay Description:Displacement of [125I]ET-1 from human ET-A receptor expressed in CHO cell membraneMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0340nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:Compound was evaluated for the binding affinity to Endothelin A receptor in the rat atriumMore data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:Binding affinity towards Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:Binding affinity towards Endothelin A receptorMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0390nMAssay Description:Binding affinity against human Endothelin A receptorMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0400nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0400nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0400nMAssay Description:Ability to inhibit [125I]ET1 binding to the Endothelin A receptor from porcine lung membranes was evaluatedMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0450nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0470nMAssay Description:binding affinity against human Endothelin A receptorMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0500nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0500nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0690nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.0700nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.0870nMAssay Description:Compound was evaluated for the binding affinity to Endothelin A receptor in the rat atriumMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.110nMAssay Description:Displacement of [125I]-ET-1 from human ETA receptor expressed in CHO cell membranes after 2 hrs by scintillation countingMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.110nMAssay Description:Displacement of [125I]-ET-1 from human ETA receptor expressed in CHO cell membranes after 2 hrs by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 0.110nMAssay Description:Compound was evaluated for the binding affinity to Endothelin A receptor in the rat aortaMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.120nMAssay Description:Binding affinity of the compound towards human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.130nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.130nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.150nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.150nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.170nMAssay Description:Tested for binding affinity for human Endothelin A receptor by measuring its ability to displace [125I]-ET-1 from chinese hamster ovary cells(CHO)More data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.170nMAssay Description:Inhibitory activity of the compound against human Endothelin A receptor in chinese hamster ovary cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Inhibition of Human Endothelin A receptor expressed in CHO Cells.More data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Inhibitory activity against human endothelin A receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetEndothelin-1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Ability of the compound to displace endothelin ([125I]-ET-1) from human Endothelin A receptorMore data for this Ligand-Target Pair