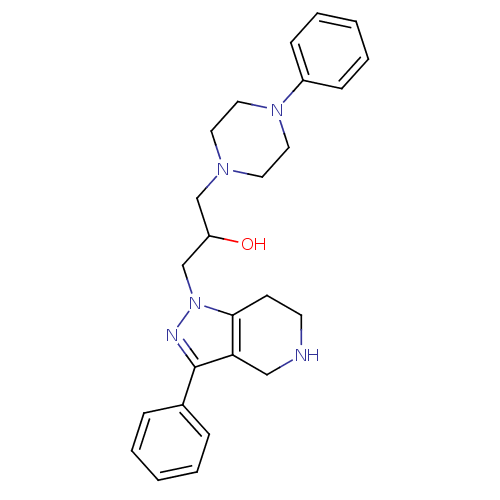
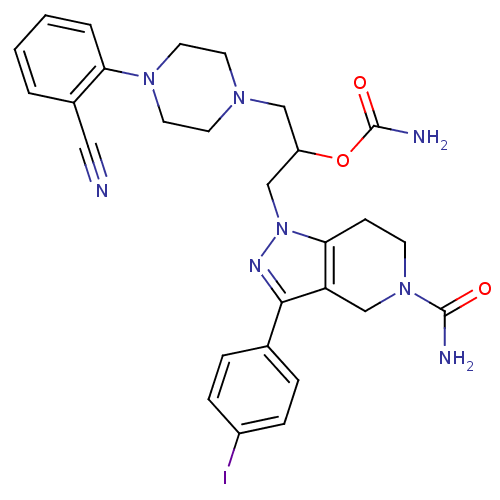
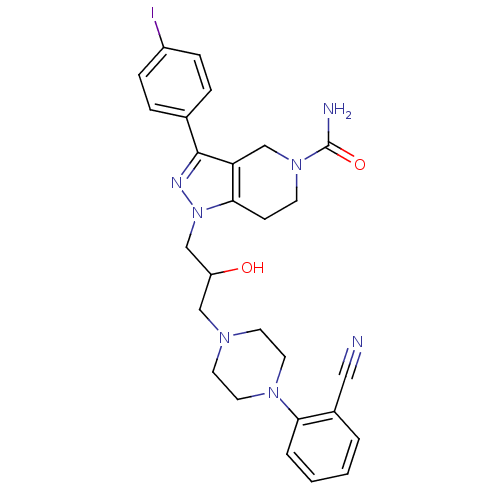
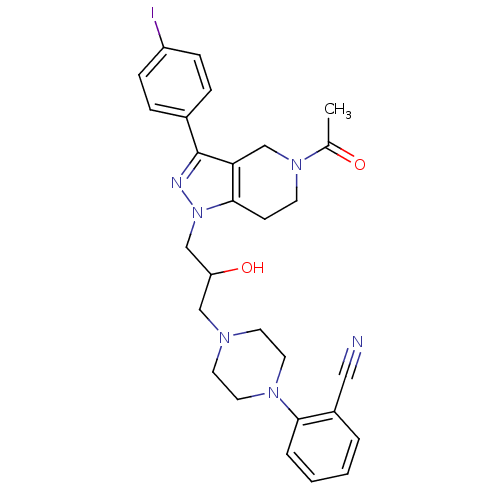
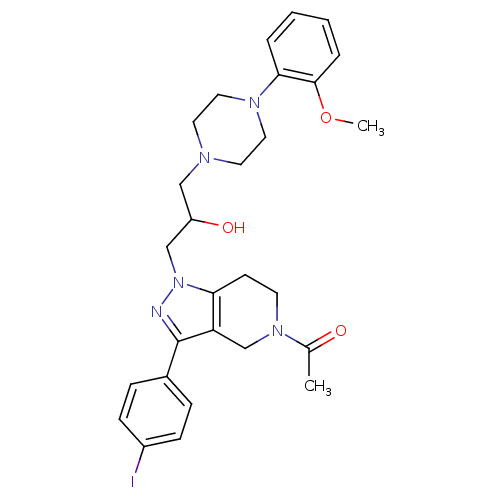
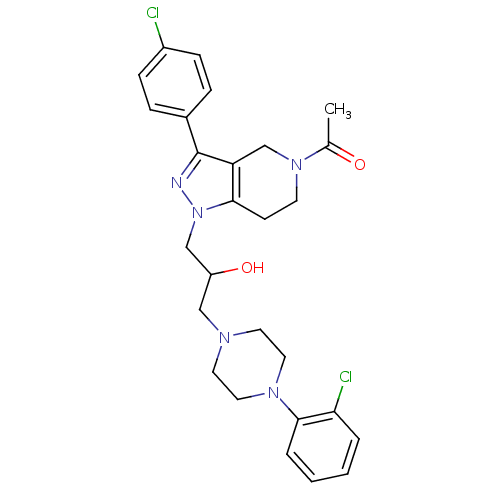
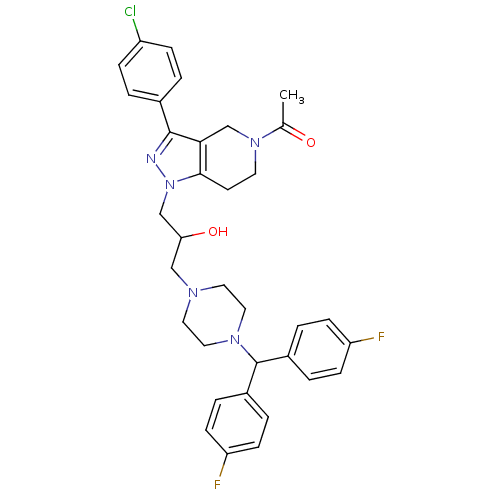
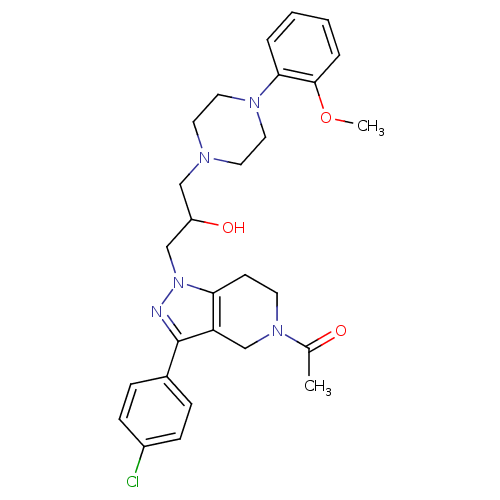
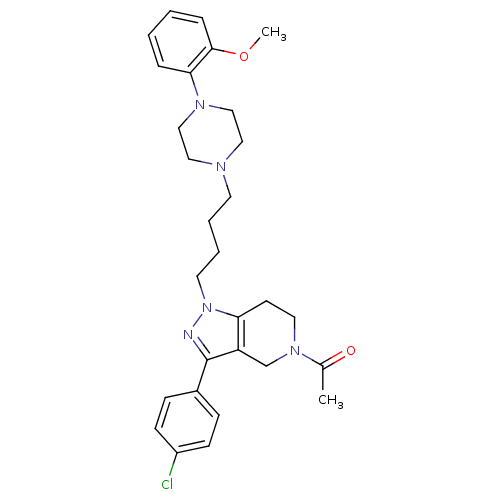
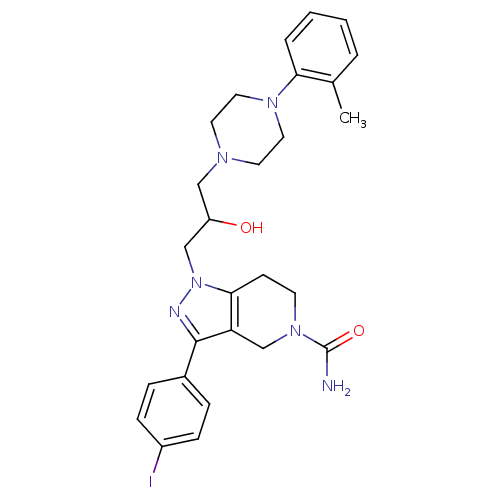
Report error Found 10 Enz. Inhib. hit(s) with all data for entry = 50015205
Affinity DataIC50: 5nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 600nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 9.50E+3nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+4nMAssay Description:Inhibitory concentration against human cysteine protease cathepsin SMore data for this Ligand-Target Pair