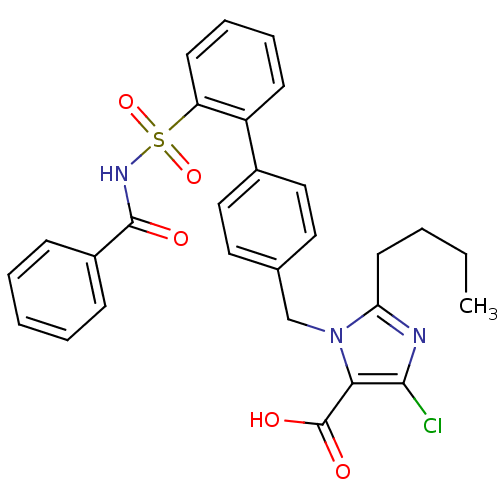
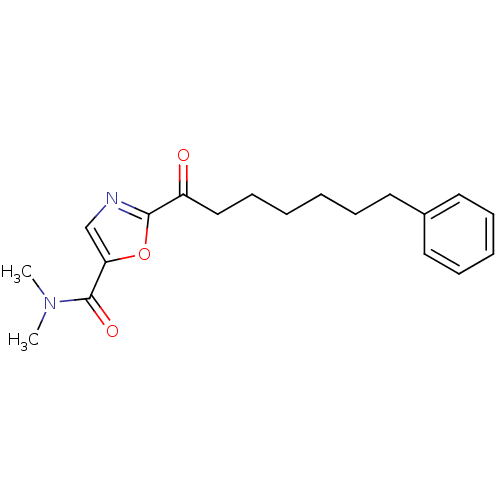
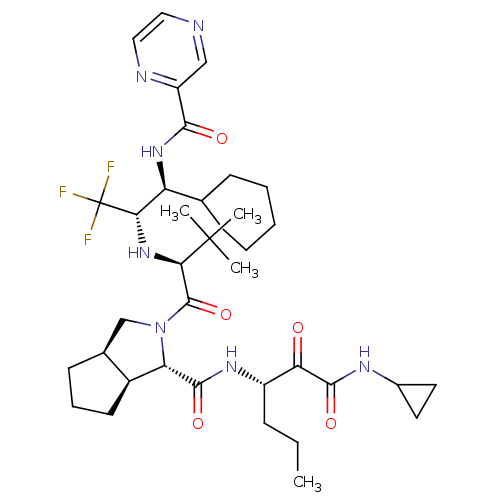
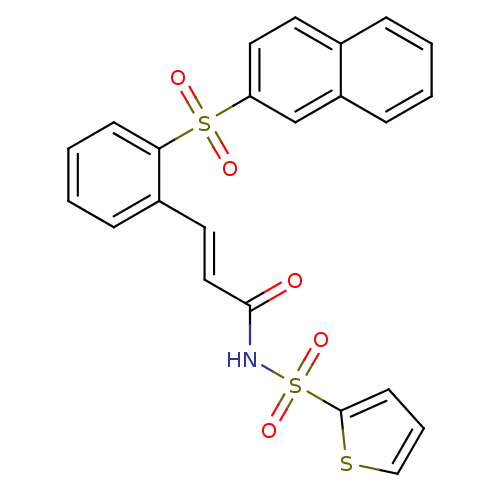
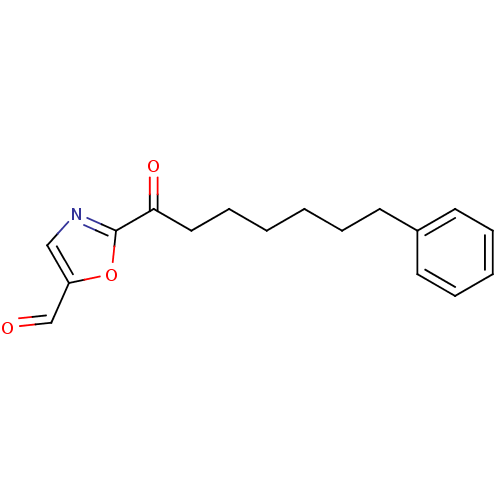
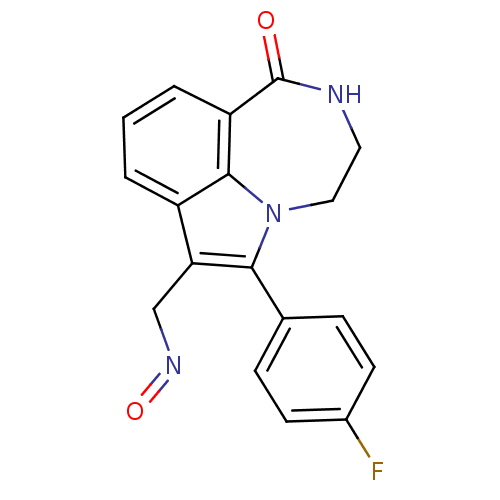
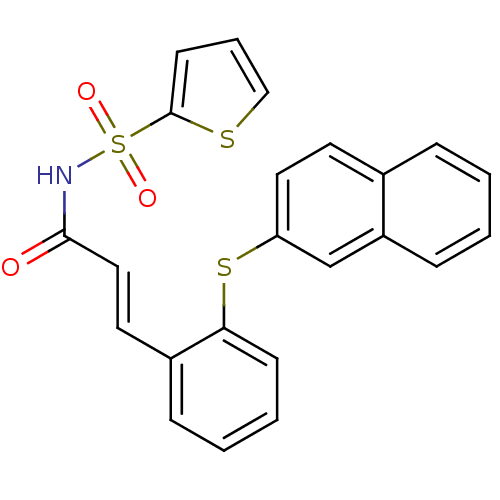
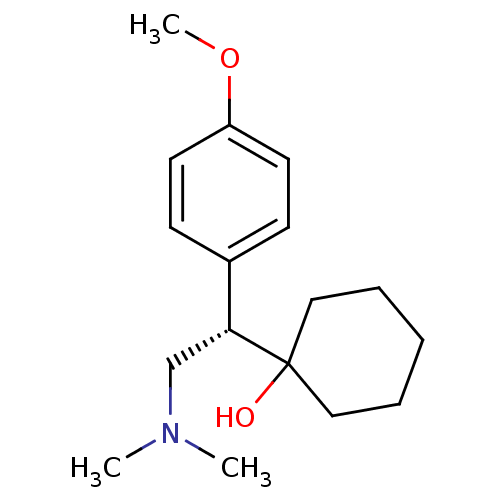
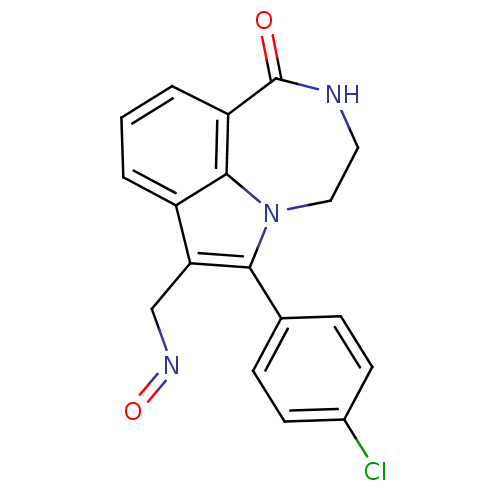
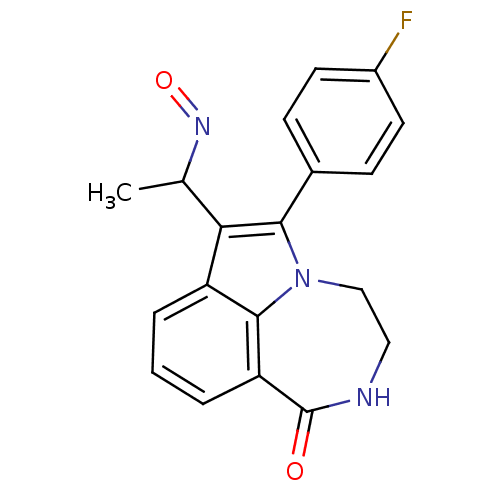
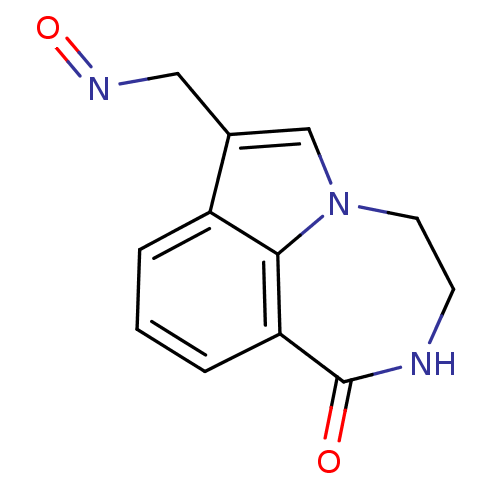
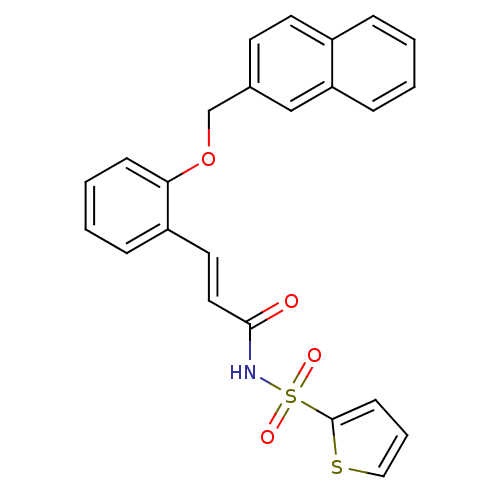
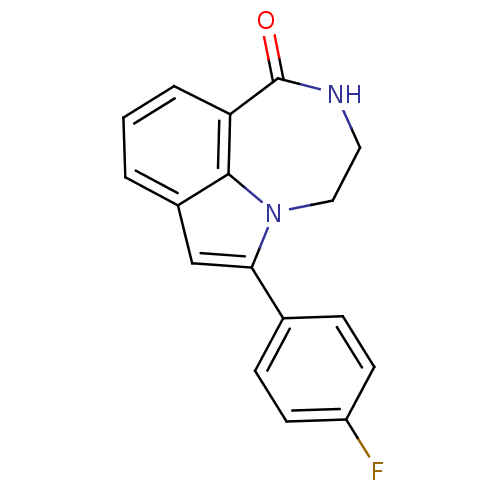
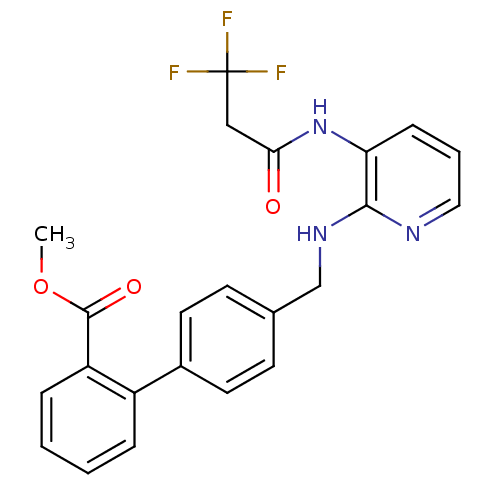
Report error Found 170 Enz. Inhib. hit(s) with all data for entry = 50040833
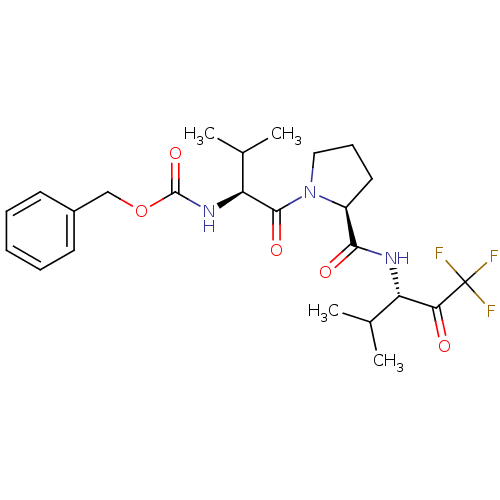
TargetNeutrophil elastase(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
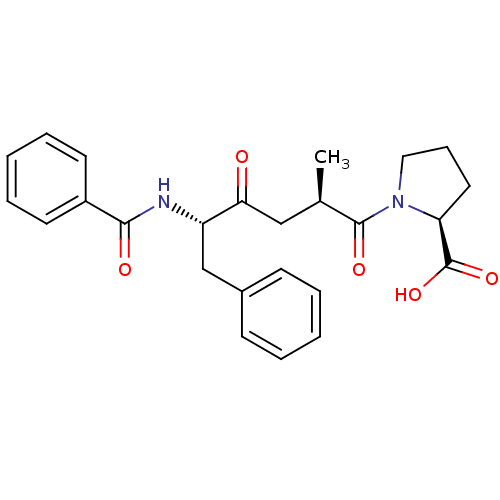
Affinity DataKi: 0.0250nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 0.200nMAssay Description:Antagonist activity at angiotensin 1 receptorMore data for this Ligand-Target Pair
TargetSubstance-P receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Antagonist activity at neurokinin K1 receptorMore data for this Ligand-Target Pair
TargetType-1 angiotensin II receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Antagonist activity at angiotensin 1 receptorMore data for this Ligand-Target Pair
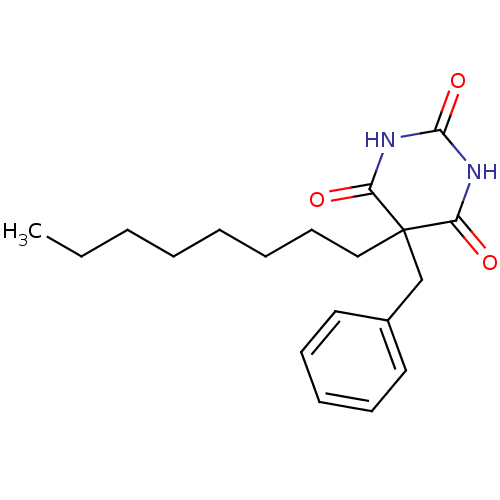
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetNeutrophil elastase(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 0.490nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 0.550nMAssay Description:Inhibition of human dopamine D2 receptorMore data for this Ligand-Target Pair
TargetSubstance-P receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 0.600nMAssay Description:Antagonist activity at neurokinin K1 receptorMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetCathepsin S(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 0.600nMAssay Description:Inhibition of cathepsin SMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetAngiotensin-converting enzyme(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
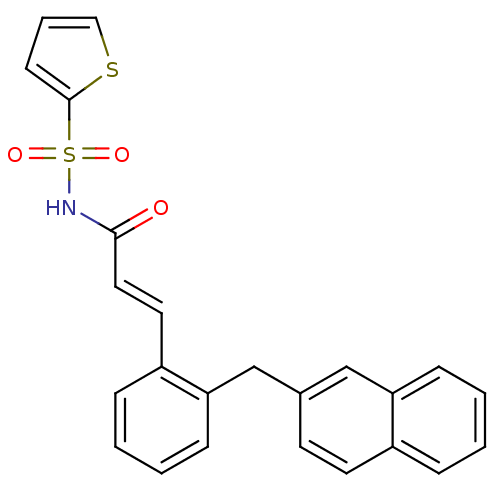
TargetMatrix metalloproteinase-9(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetProstaglandin E2 receptor EP3 subtype(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 1.60nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 1.90nMAssay Description:Inhibition of sigma 1 receptorMore data for this Ligand-Target Pair
TargetType-2 angiotensin II receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Antagonist activity at angiotensin 2 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetMatrix metalloproteinase-9(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetCathepsin S(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of cathepsin SMore data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 2.10nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 2.84nMAssay Description:Inhibition of human dopamine D2 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetNeutrophil elastase(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 3.40nMAssay Description:Inhibition of sigma 1 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetAngiotensin-converting enzyme(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetD(3) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 4.73nMAssay Description:Inhibition of human dopamine D3 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetCoagulation factor X(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataIC50: 5.90nMAssay Description:Inhibition of factor 10aMore data for this Ligand-Target Pair
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 6.70nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 7.5nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetD(4) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 7.94nMAssay Description:Inhibition of human dopamine D4 receptorMore data for this Ligand-Target Pair
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 9.10nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 9.40nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 9.90nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
TargetMatrix metalloproteinase-9(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
TargetD(3) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 10.4nMAssay Description:Inhibition of human dopamine D3 receptorMore data for this Ligand-Target Pair
TargetPoly [ADP-ribose] polymerase 1(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 11nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetB1 bradykinin receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 11.8nMAssay Description:Antagonist activity at Bradykinin B1 receptorMore data for this Ligand-Target Pair
TargetD(4) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 14.1nMAssay Description:Inhibition of human dopamine D4 receptorMore data for this Ligand-Target Pair
TargetD(1B) dopamine receptor(Human)
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataKi: 16.6nMAssay Description:Inhibition of human dopamine D5 receptorMore data for this Ligand-Target Pair


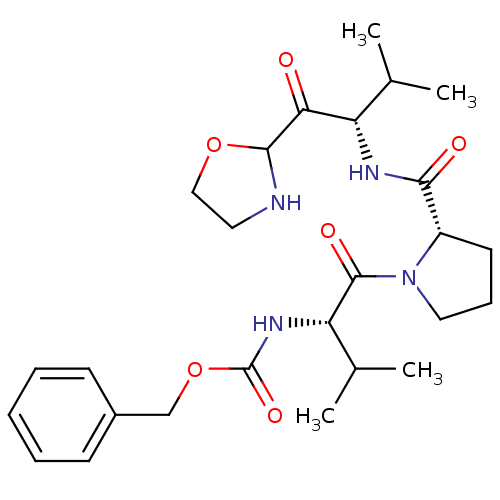
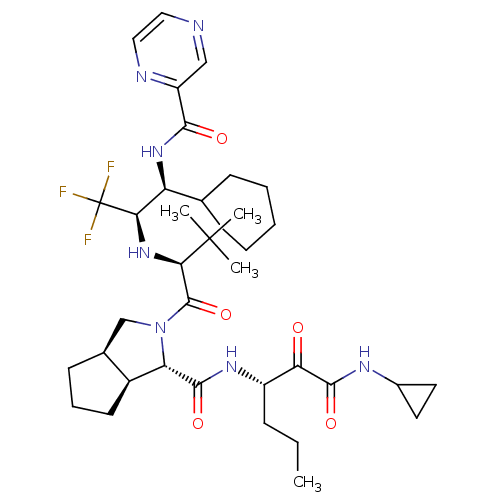
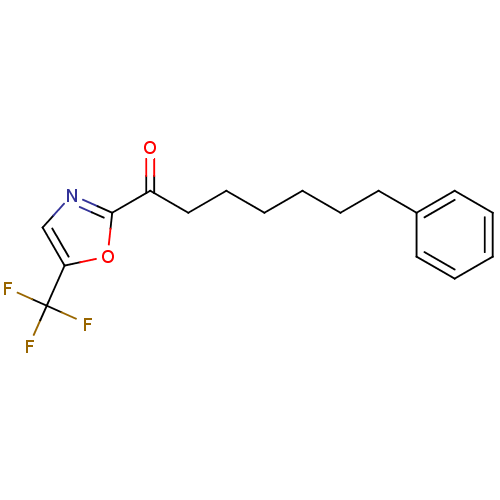
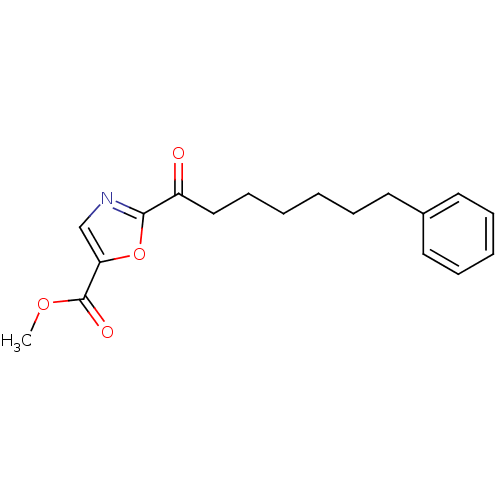
 3D Structure (crystal)
3D Structure (crystal)