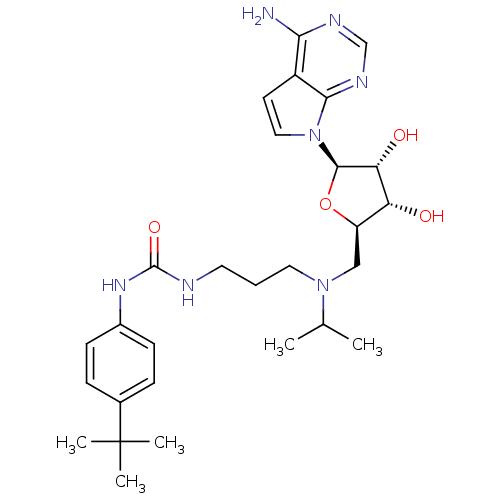
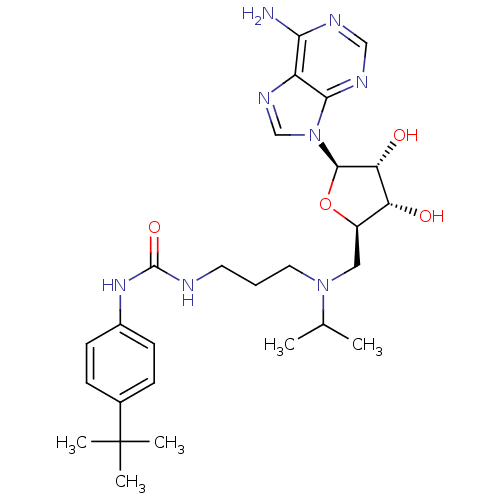
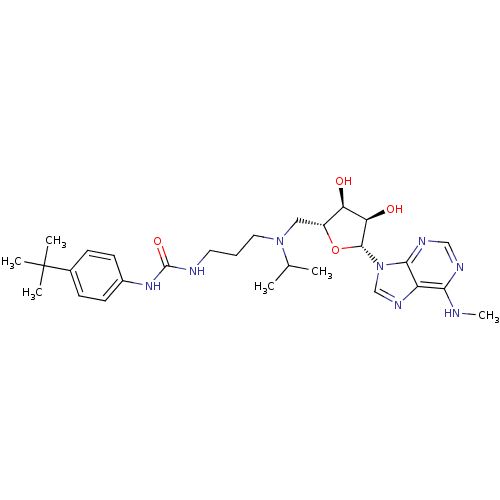
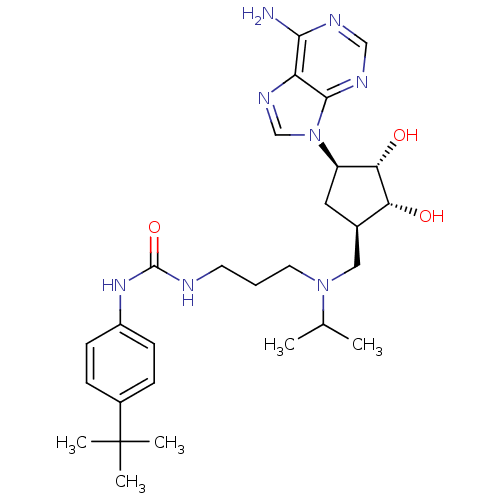
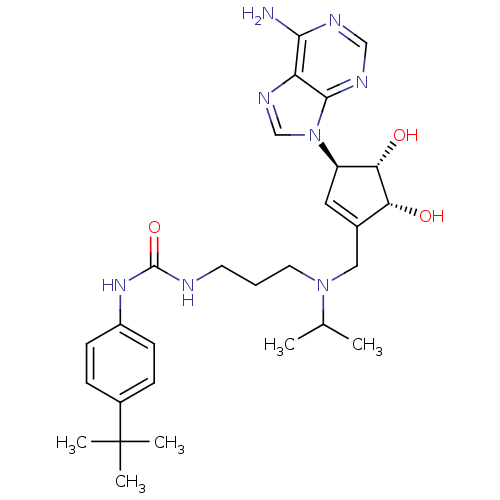
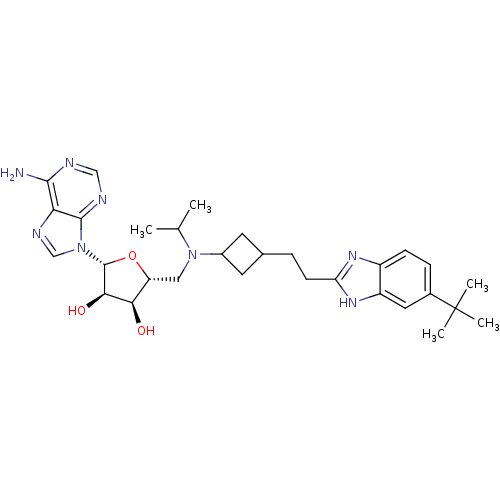
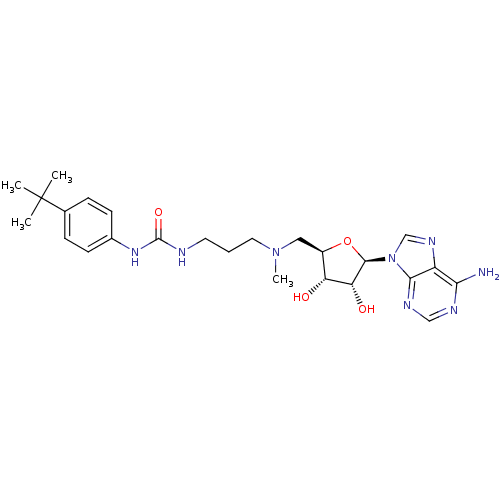
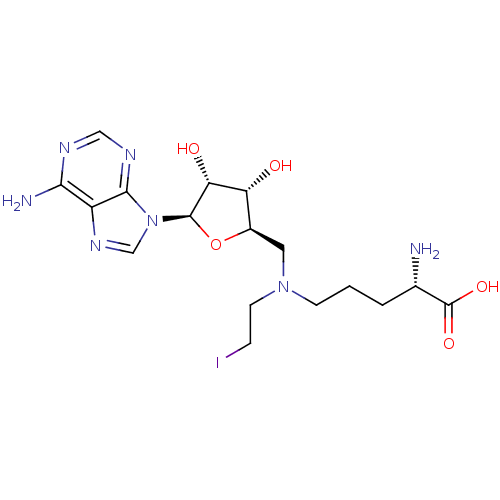
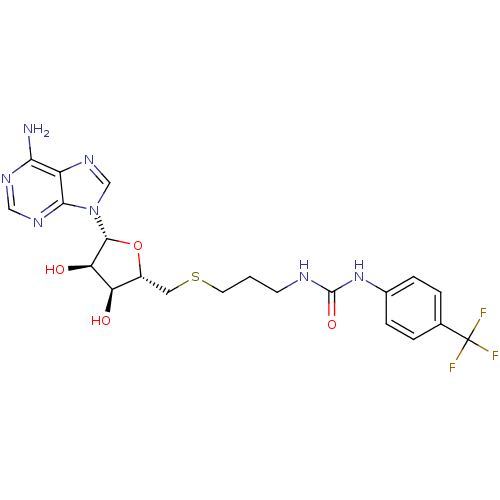
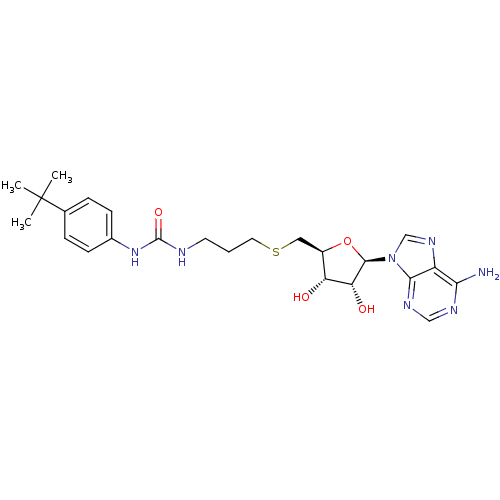
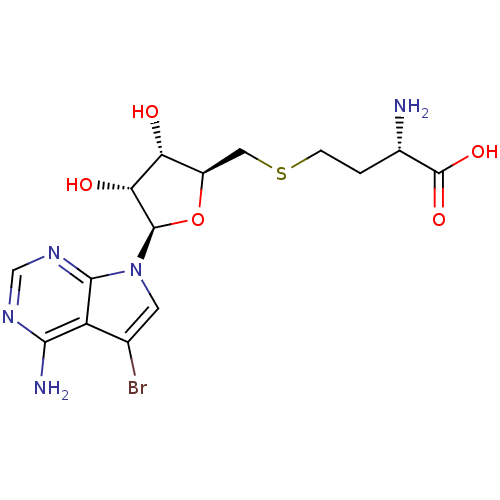
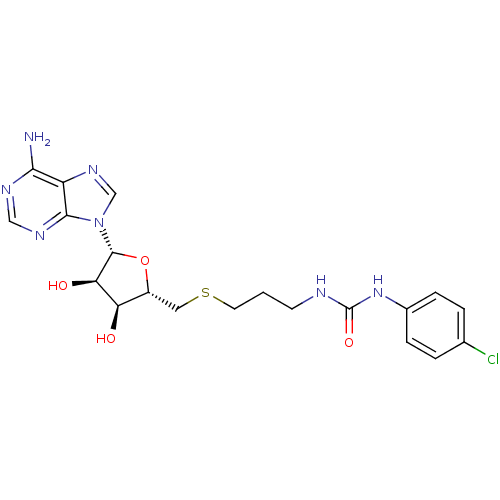
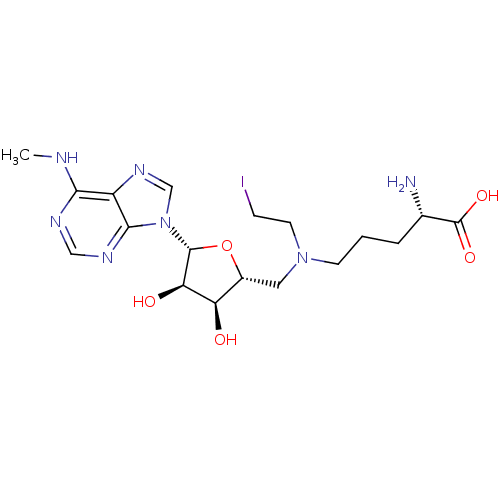
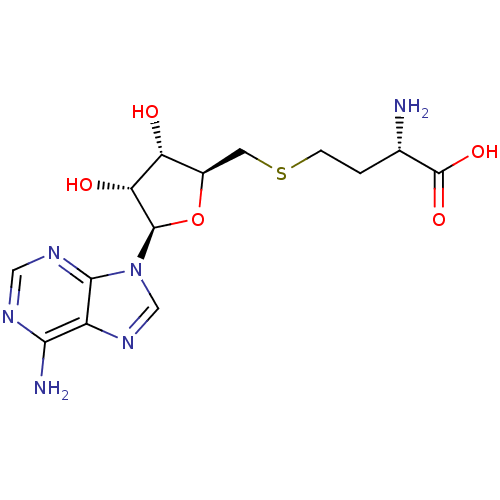
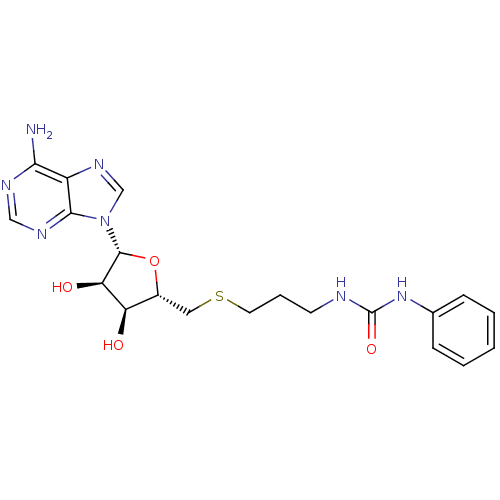
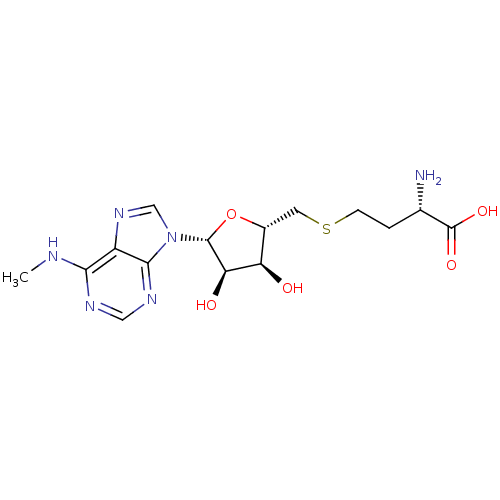
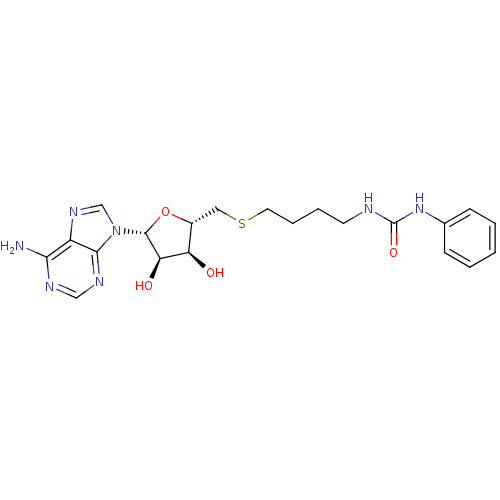
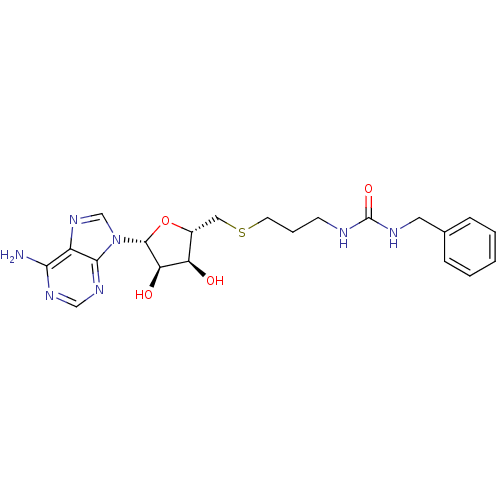
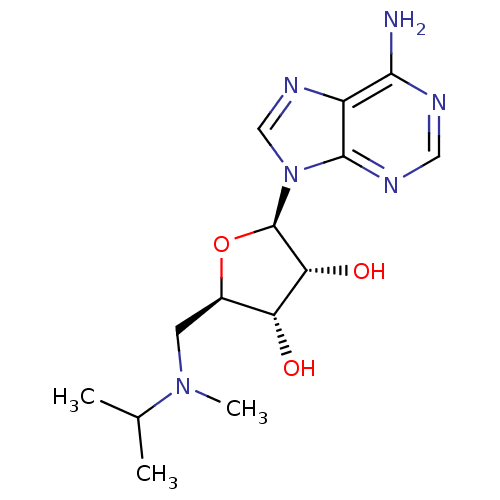
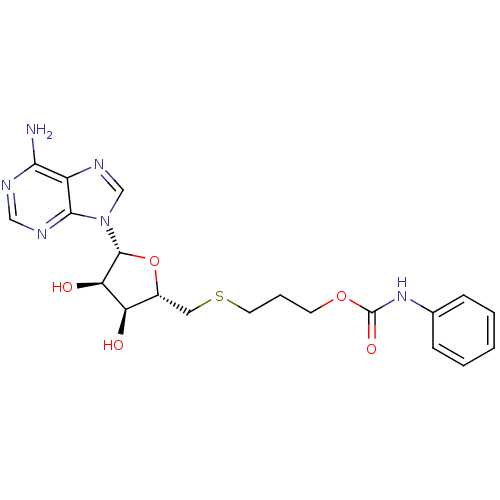
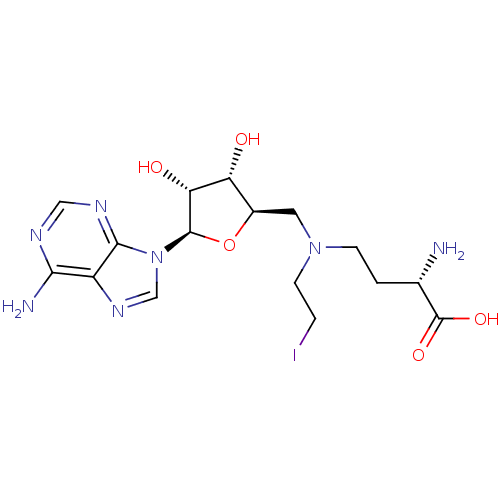
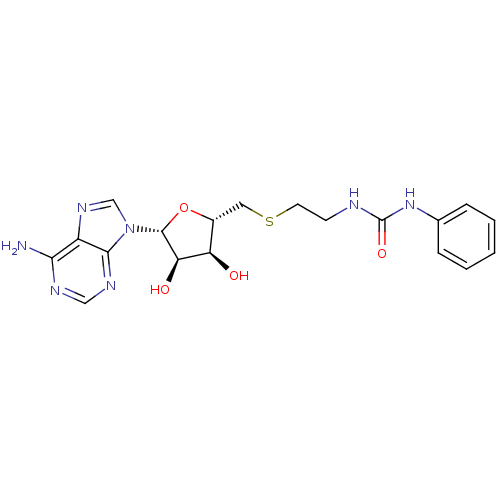
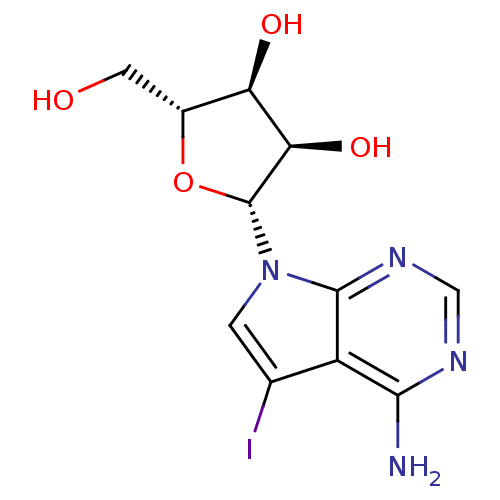
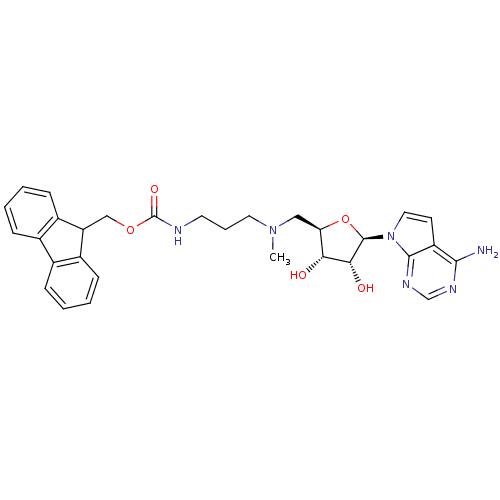
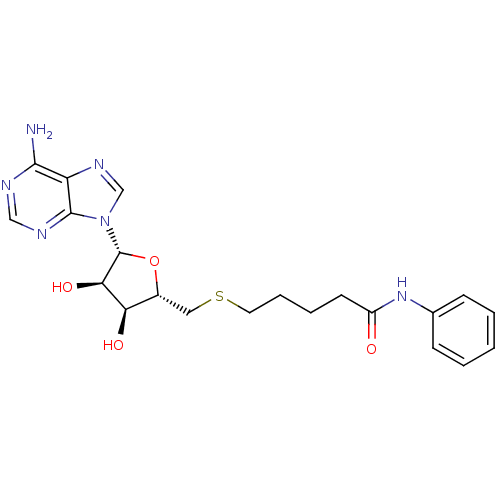
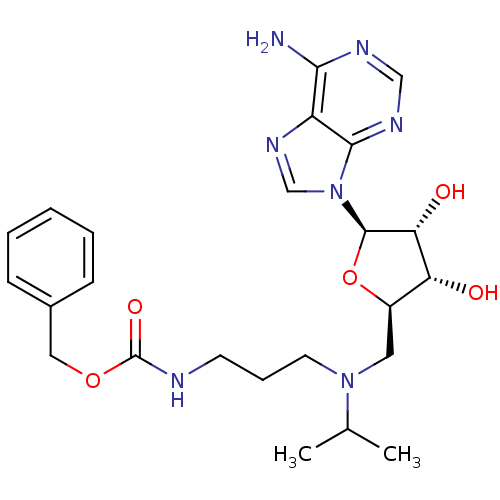
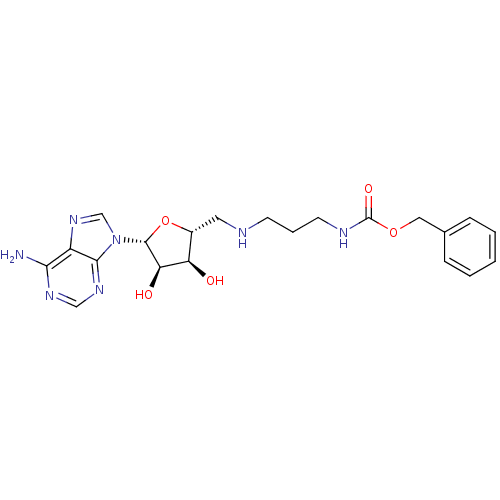
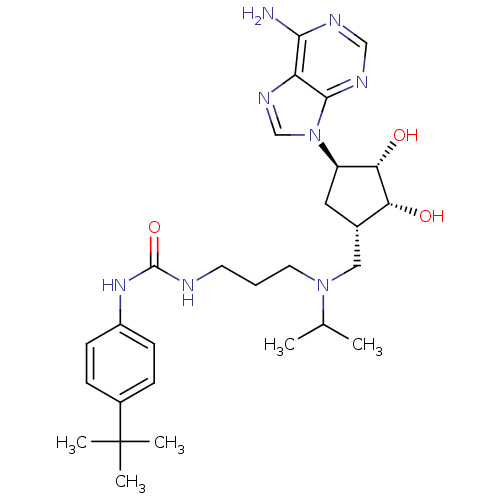
Report error Found 36 Enz. Inhib. hit(s) with all data for entry = 50043565
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
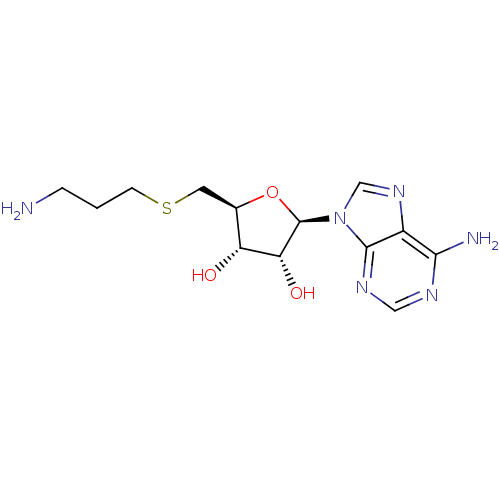
Affinity DataIC50: 0.300nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 0.5nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataEC50: 9nMAssay Description:Inhibition of DOT1L (unknown origin)-mediated H3K79 methylation by cell based assayMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataEC50: 9nMAssay Description:Inhibition of DOT1L (unknown origin)-mediated H3K79 methylation by cell based assayMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataEC50: 9nMAssay Description:Inhibition of DOT1L (unknown origin)-mediated H3K79 methylation by cell based assayMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataEC50: 9nMAssay Description:Inhibition of DOT1L (unknown origin)-mediated H3K79 methylation by cell based assayMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 13nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataIC50: 38nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 58nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 70nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataIC50: 77nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 82nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataIC50: 120nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 160nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Inhibition of CARM1 (unknown origin)More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 550nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
Affinity DataKi: 570nMAssay Description:Inhibition of G9a (unknown origin)More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.10E+3nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.10E+3nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.90E+3nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.20E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.60E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataIC50: 1.60E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.80E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataIC50: 1.80E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 2.00E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 2.20E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 2.20E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 3.80E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 4.60E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: >5.00E+4nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase, H3 lysine-79 specific(Human)
Baylor College of Medicine
Curated by ChEMBL
Baylor College of Medicine
Curated by ChEMBL
Affinity DataKi: 1.00E+5nMAssay Description:Competitive inhibition of recombinant human DOT1L using adenosine/deazaadenosine as substrate and SAM cofactorMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)