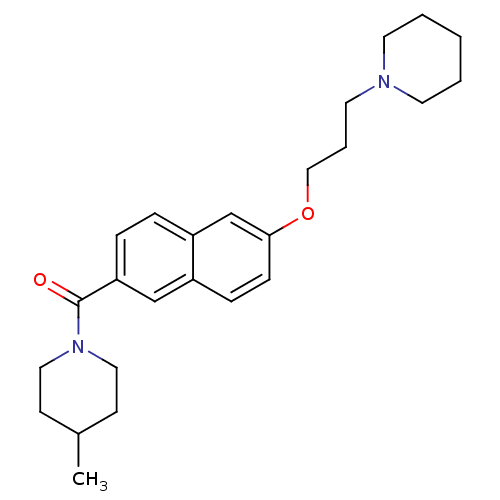
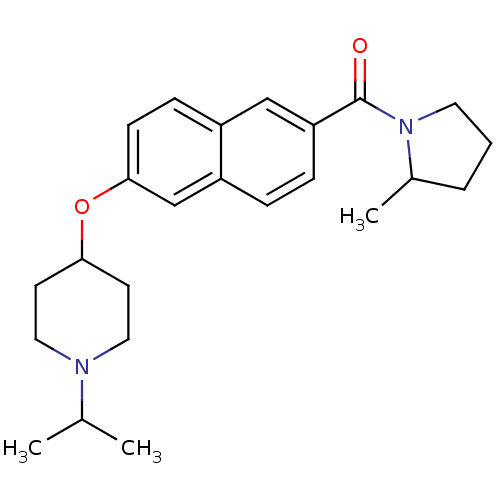
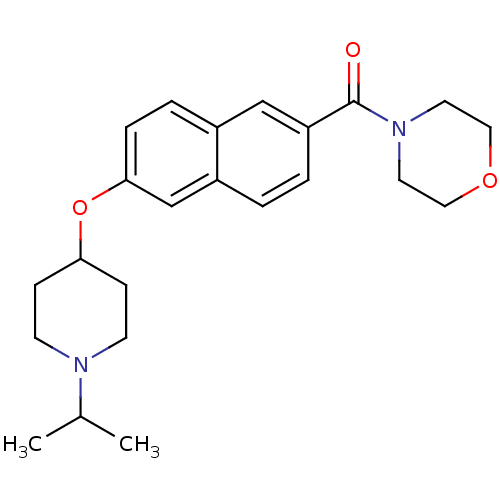
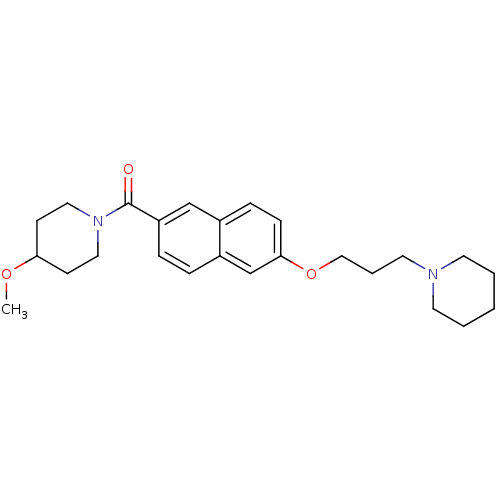
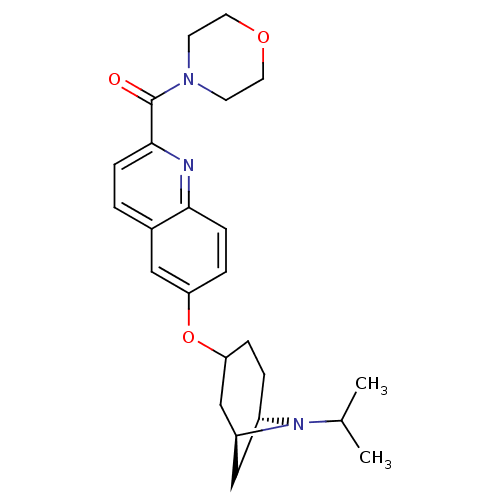
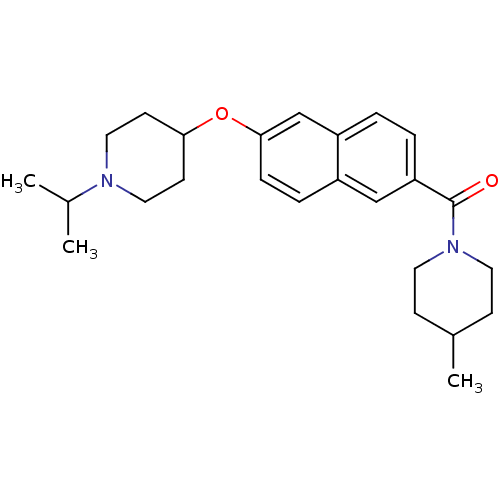
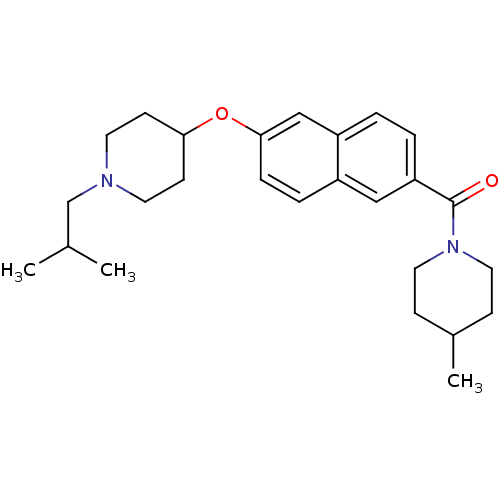
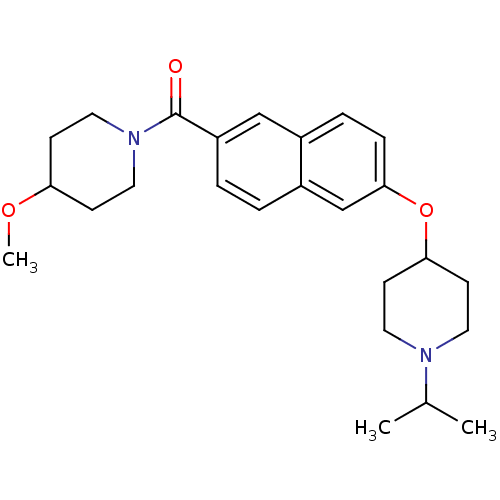
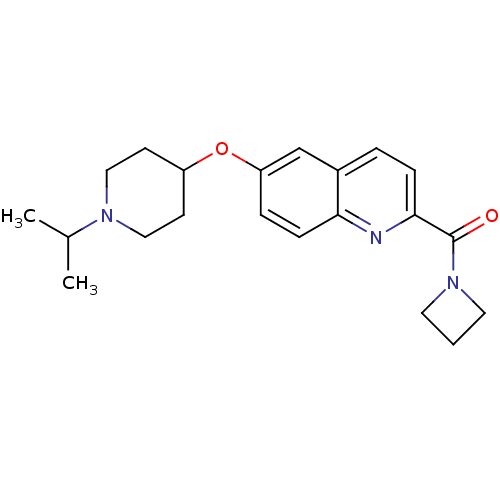
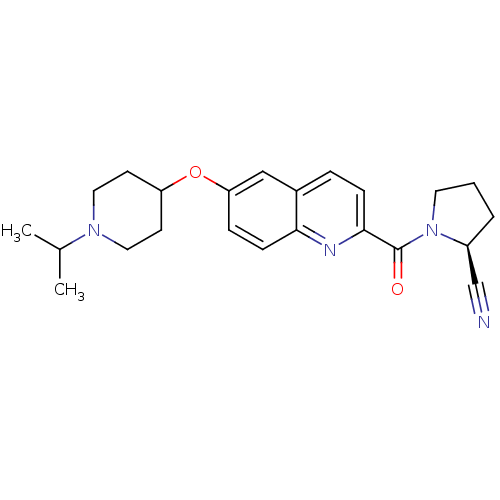
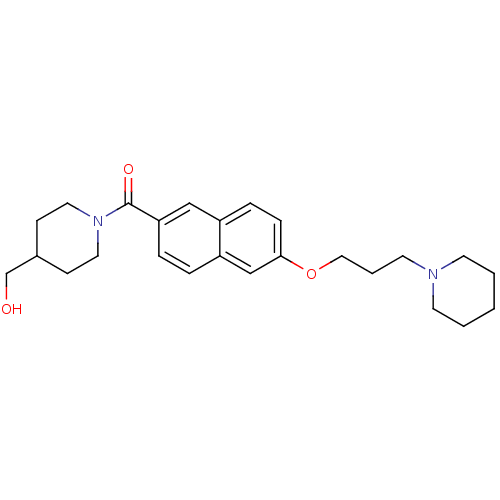
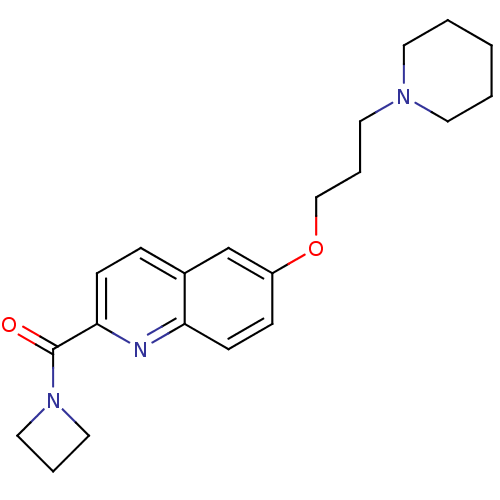
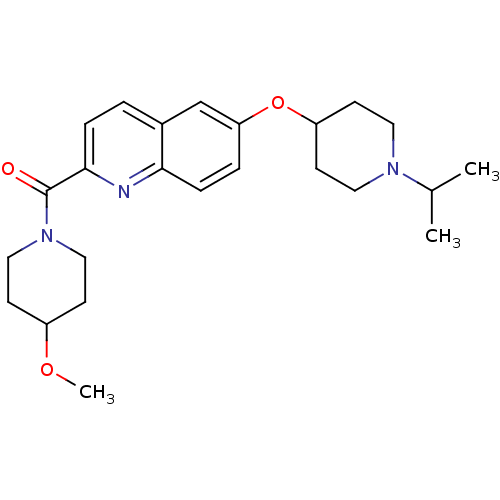
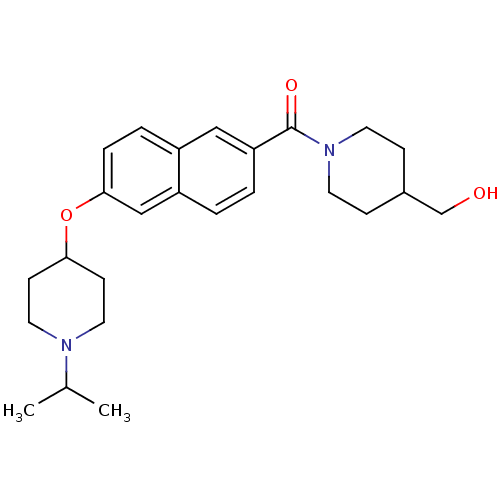
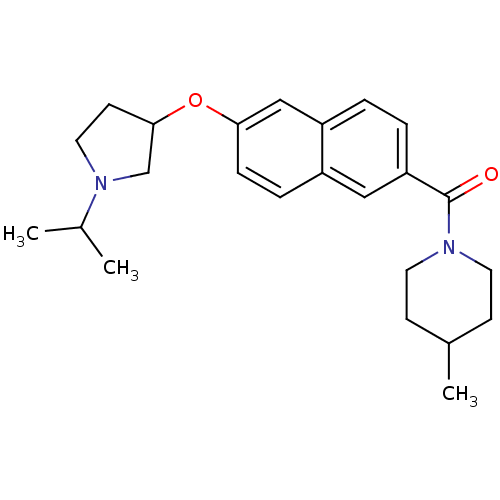
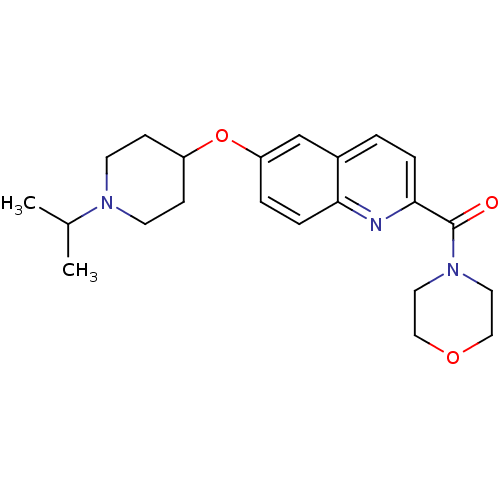
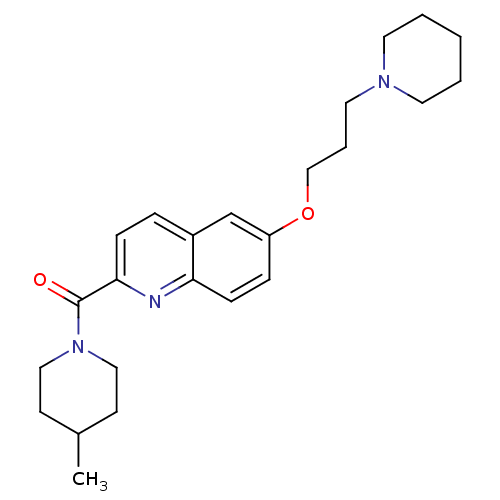
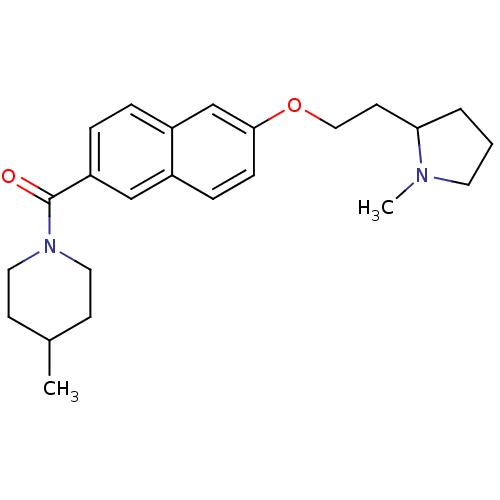
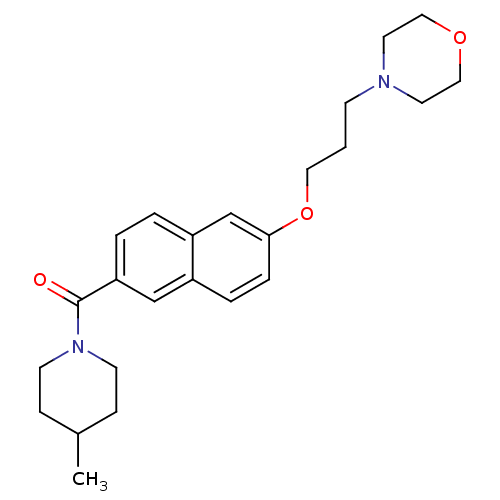
Report error Found 31 Enz. Inhib. hit(s) with all data for entry = 50030539
Affinity DataKi: 7nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:Inverse agonist activity at human histamine H3 receptor expressed in CHO cells by [35S]GTPgammaS binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 34nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 36nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 62nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 62nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 87nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 103nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 119nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 125nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 154nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataEC50: 200nMAssay Description:Inverse agonist activity at human histamine H3 receptor expressed in CHO cells by [35S]GTPgammaS binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 234nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 238nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 571nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Inhibition of human histamine H3 expressed in CHO cells by saturation binding experimentMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP2C19More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of CYP2C19More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair