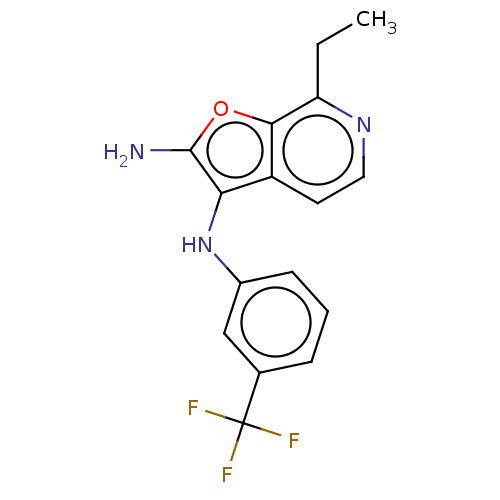
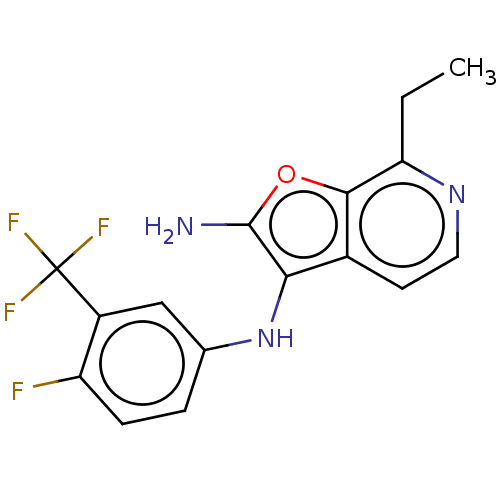
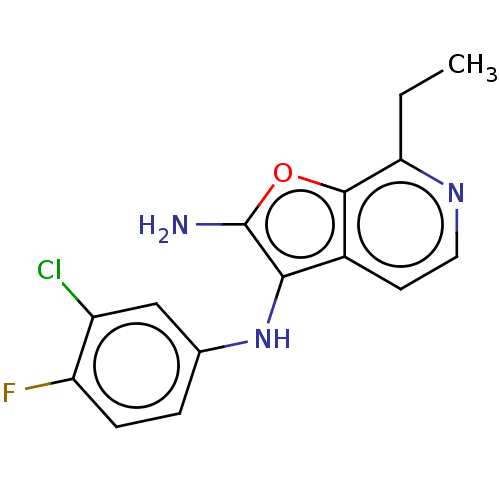
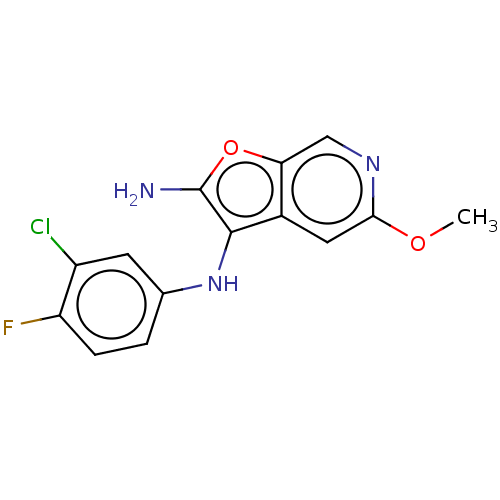
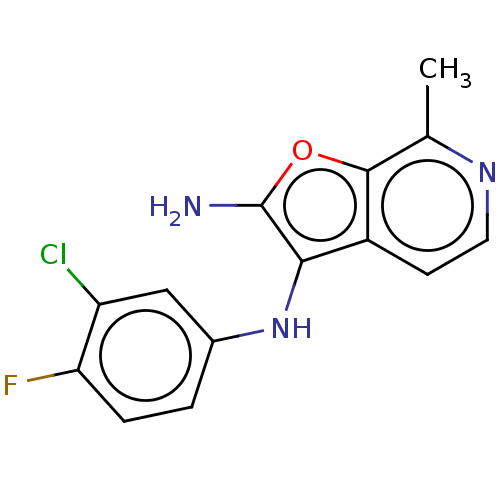
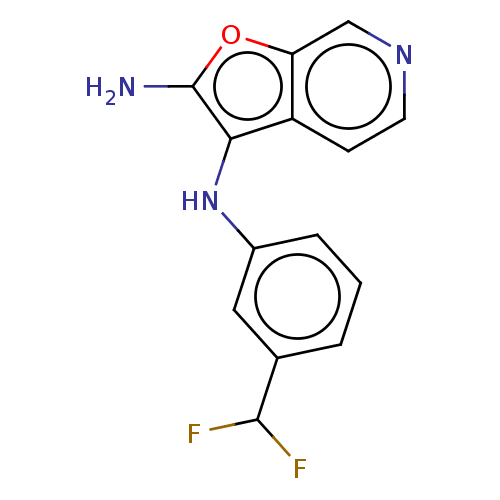
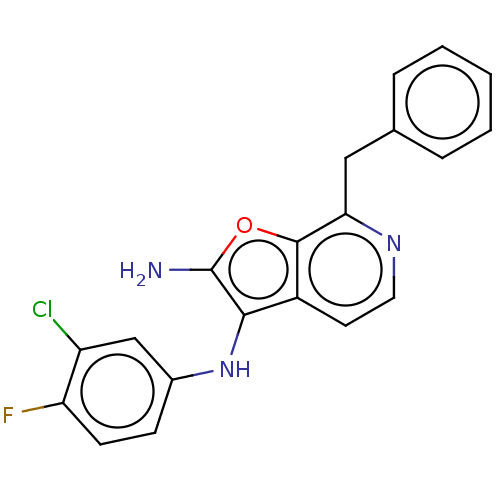
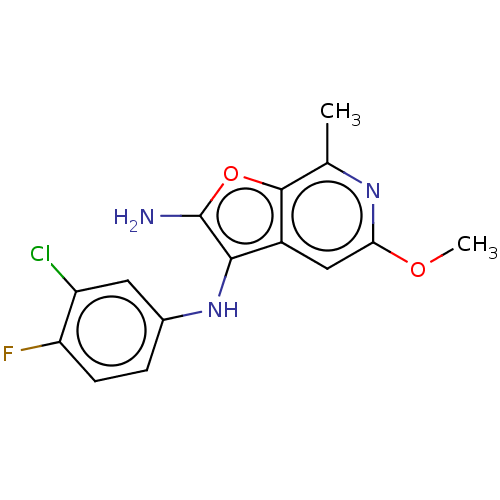
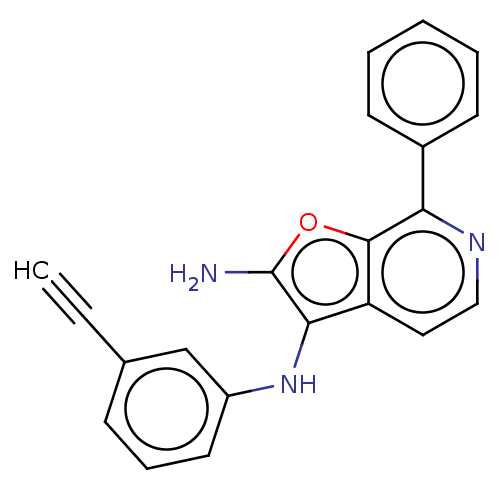
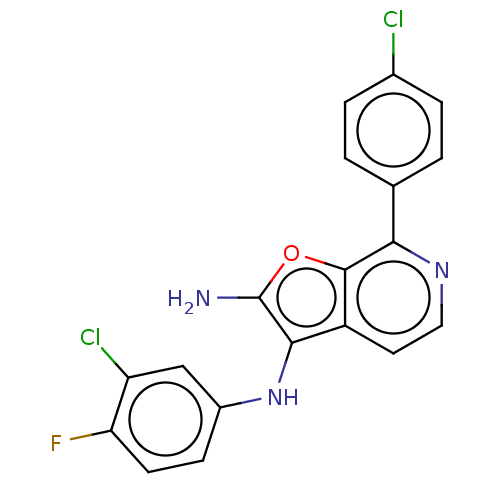
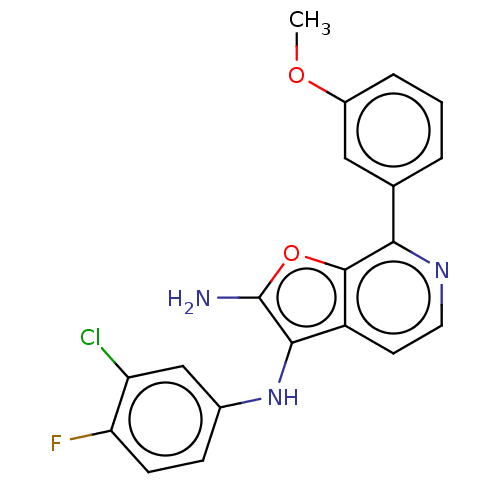
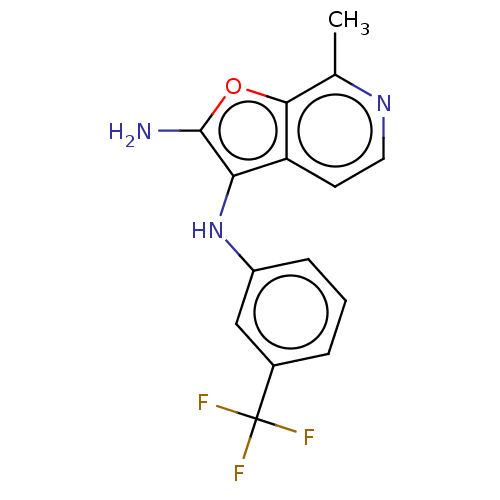
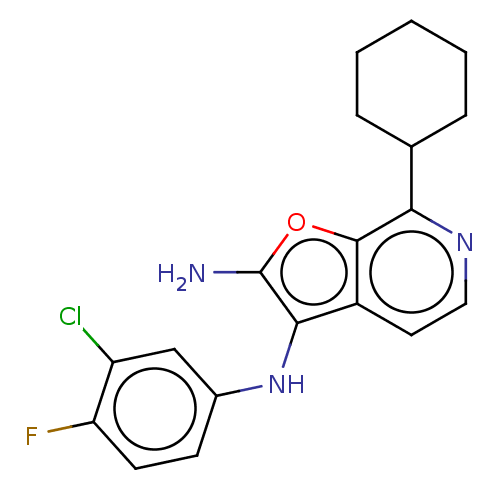
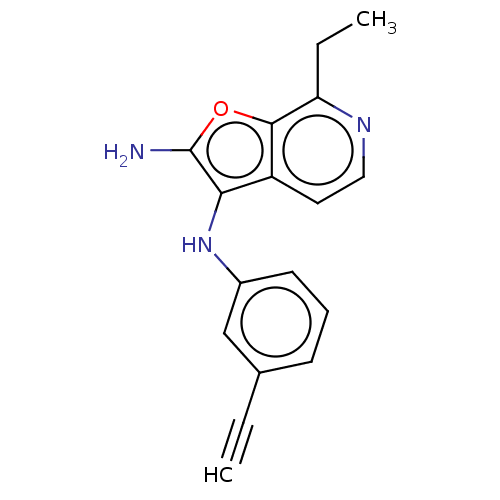
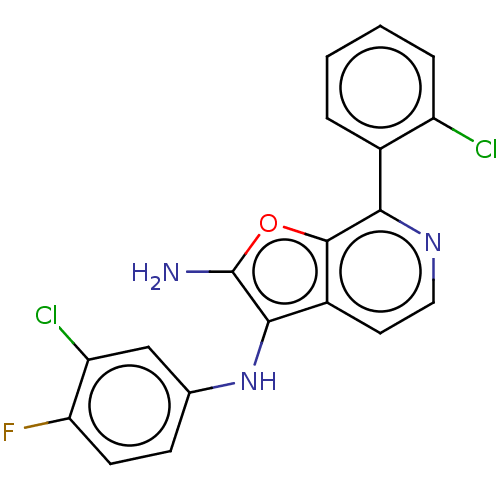
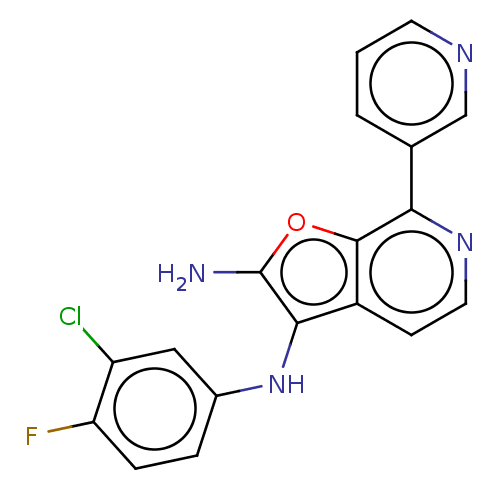
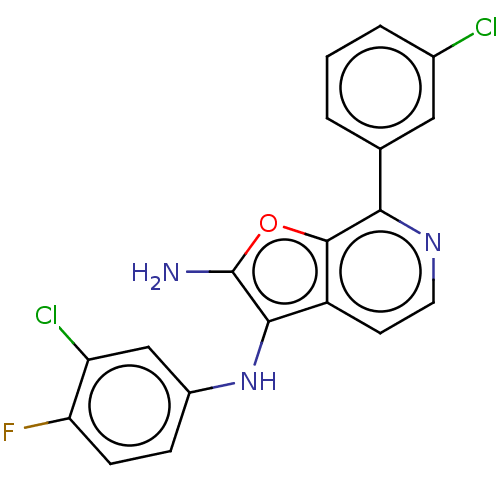
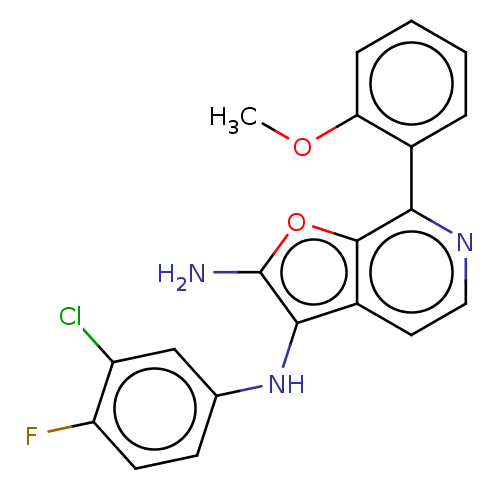
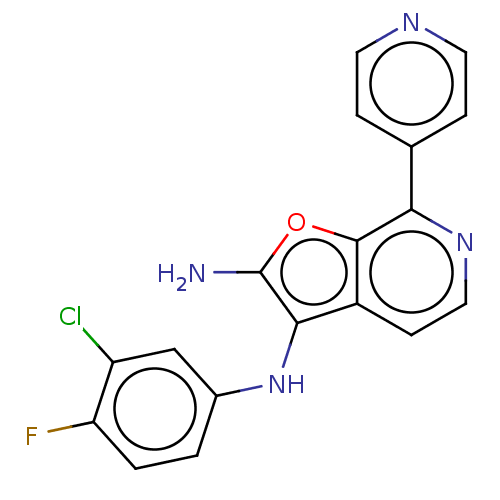
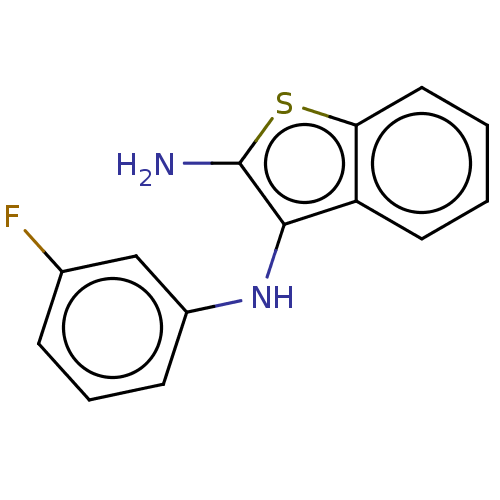
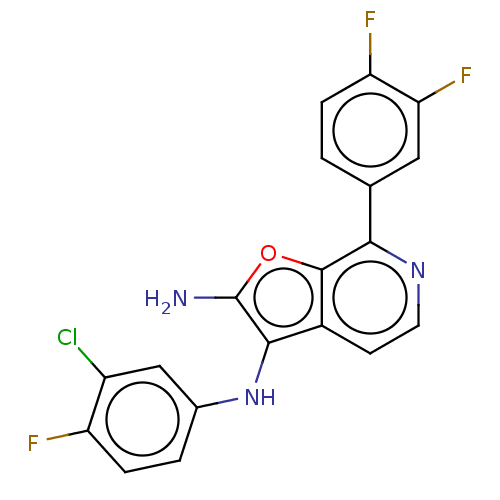
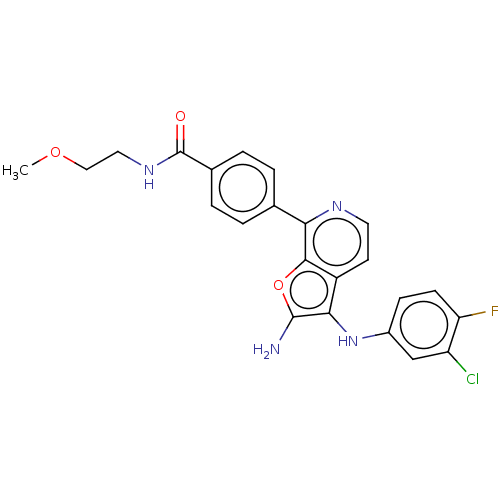
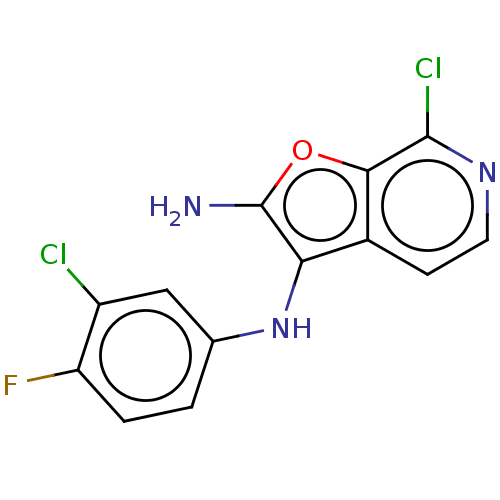
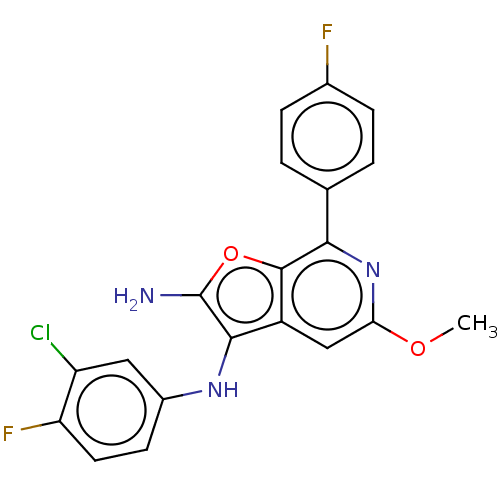
Report error Found 277 Enz. Inhib. hit(s) with all data for entry = 2987
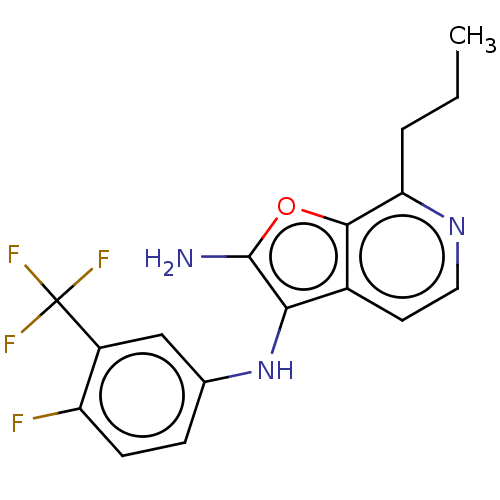
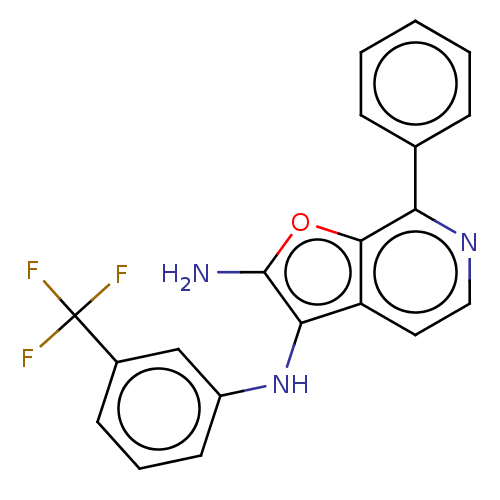
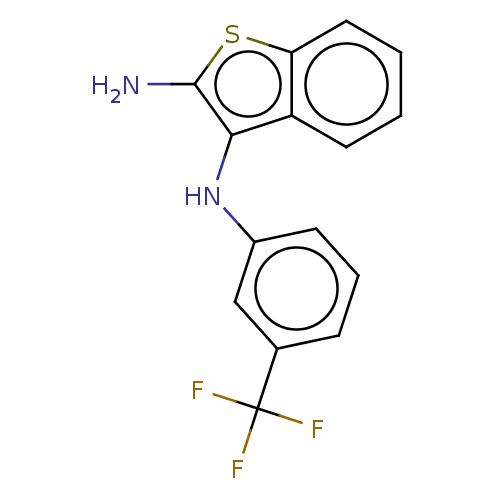
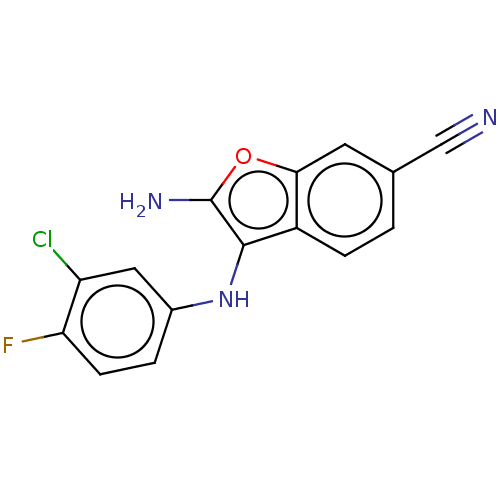
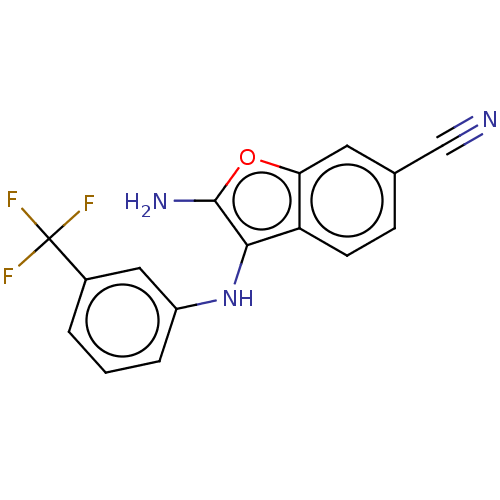
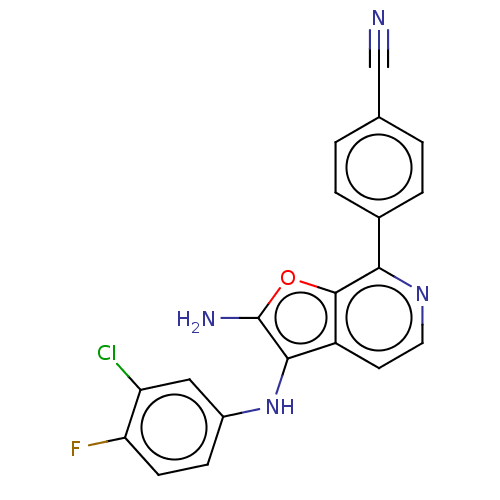
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
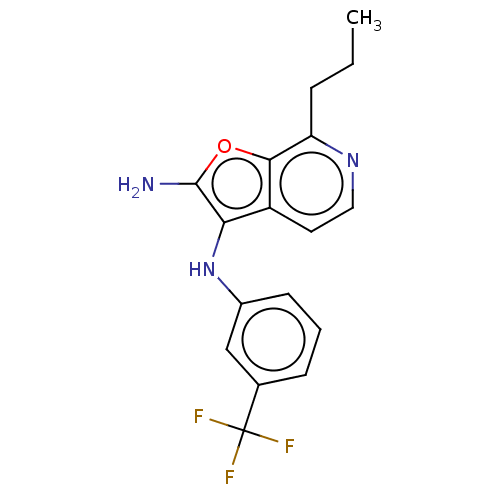
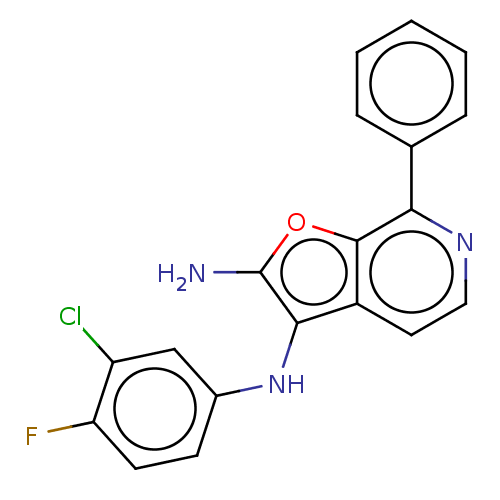
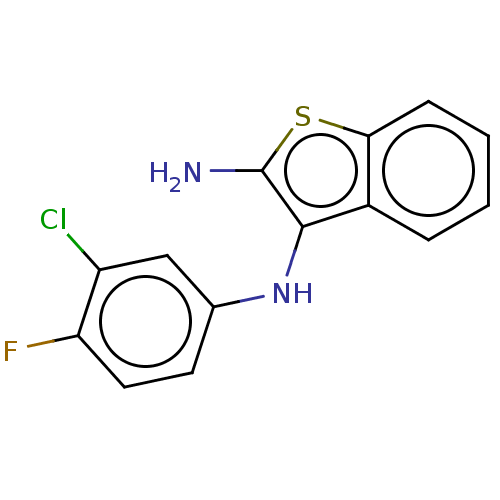
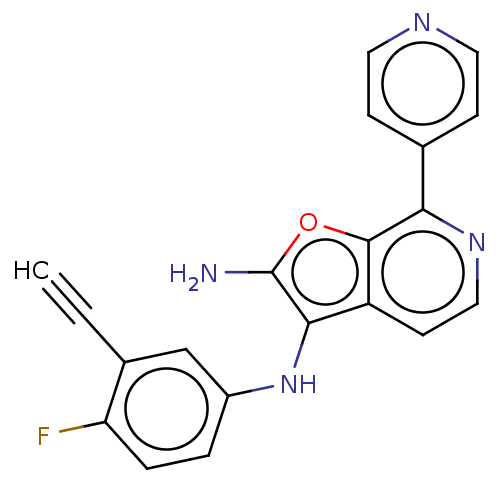
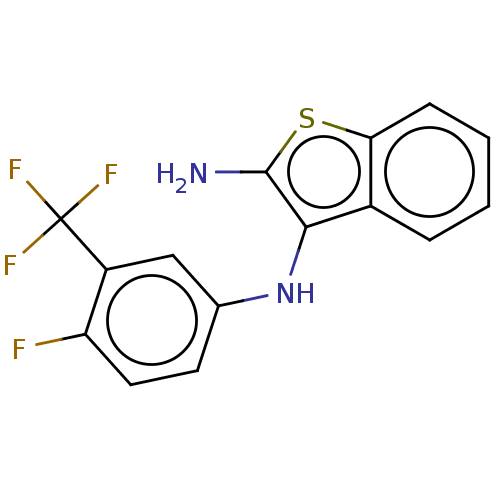
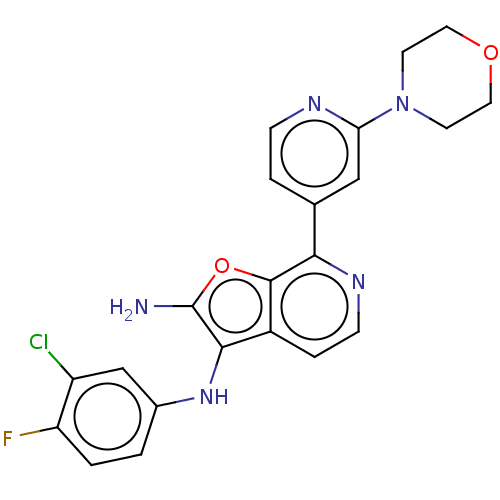
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
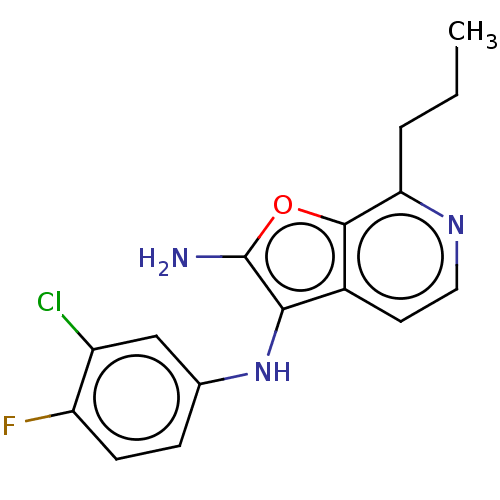
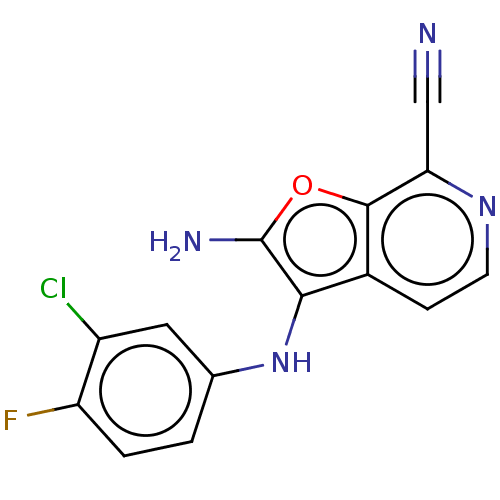
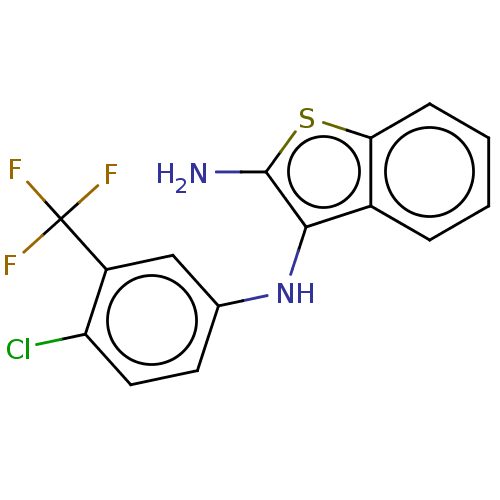
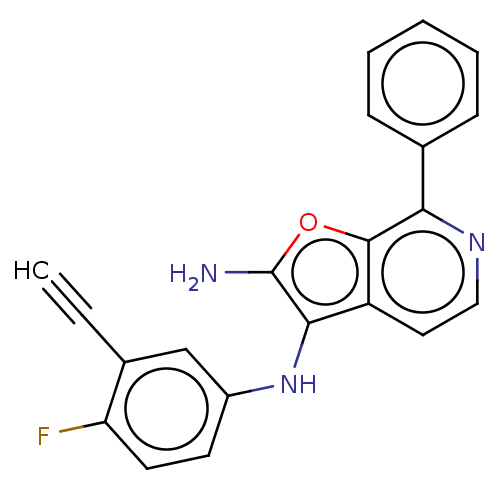
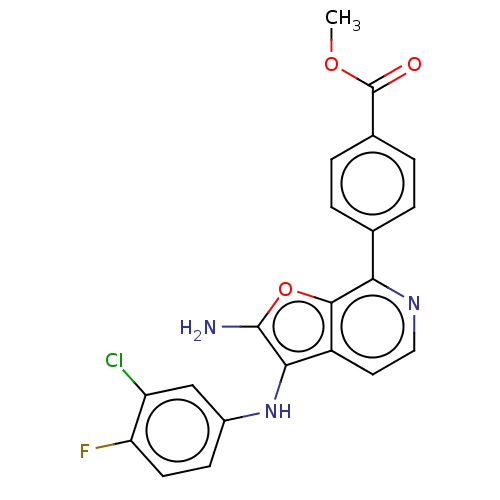
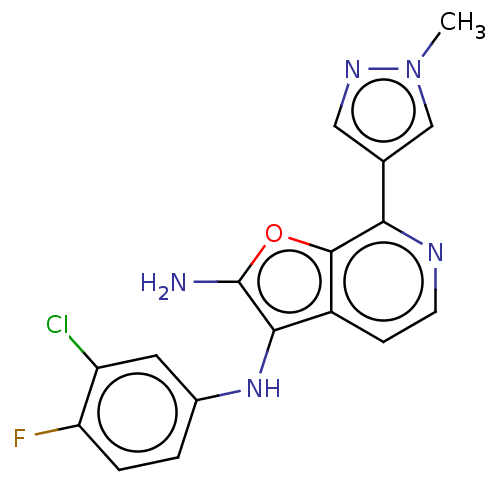
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
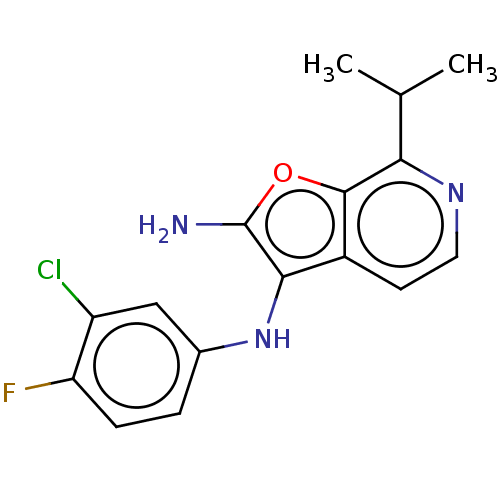
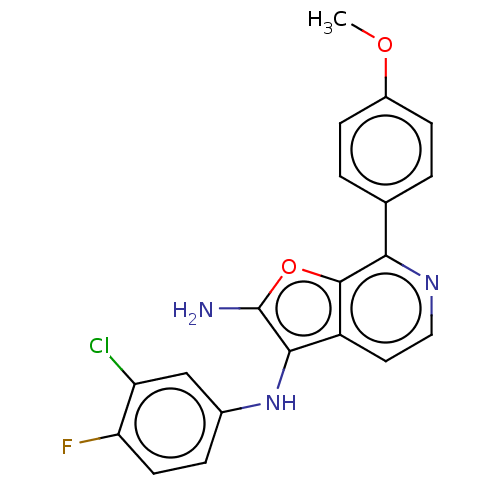
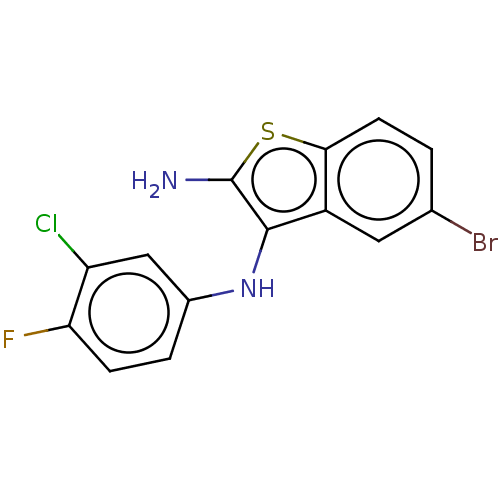
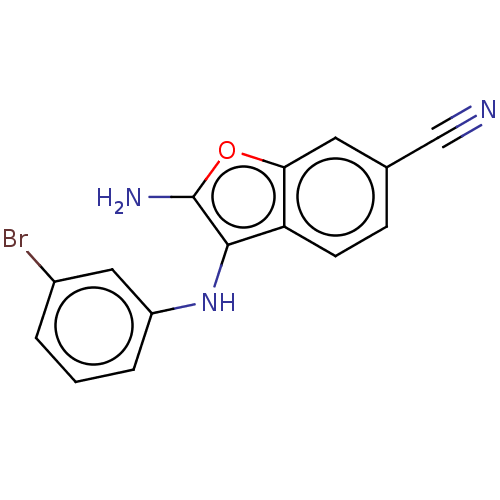
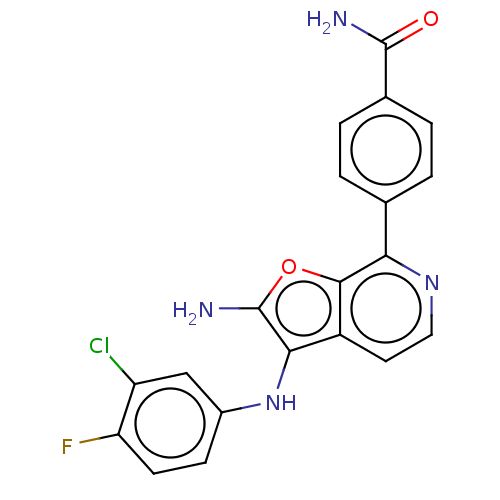
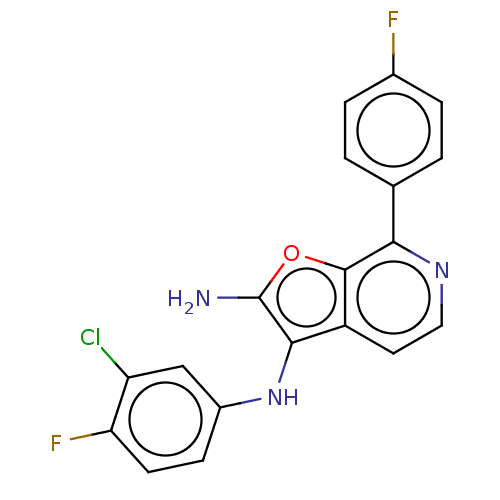
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Human indoleamine 2,3-dioxygenasel (hIDO1) catalyzes the oxidative cleavage of the pyrrole ring of the indole nucleus of tryptophan to yield N-formyl...More data for this Ligand-Target Pair