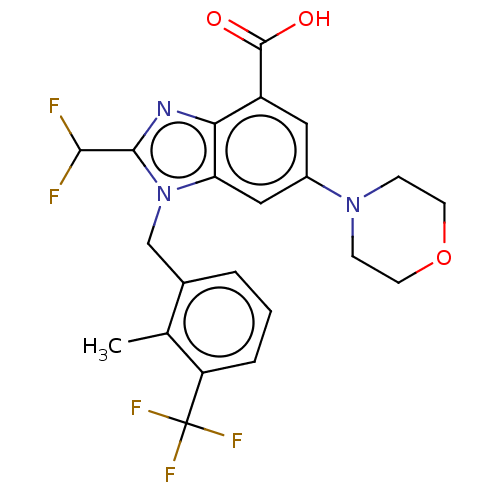
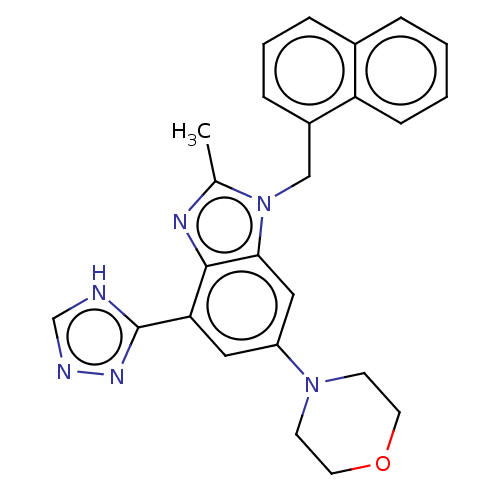
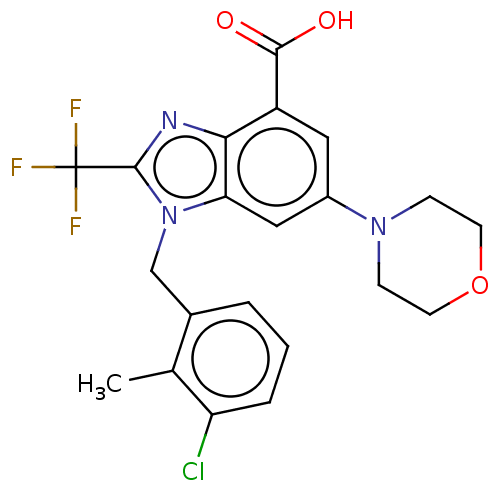
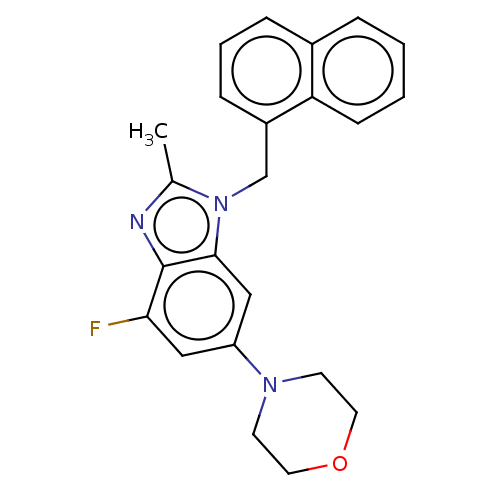
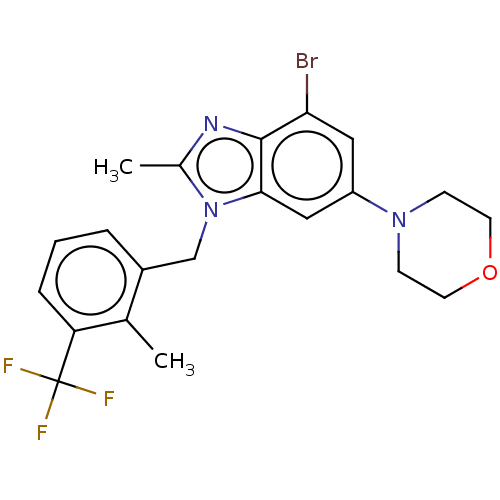
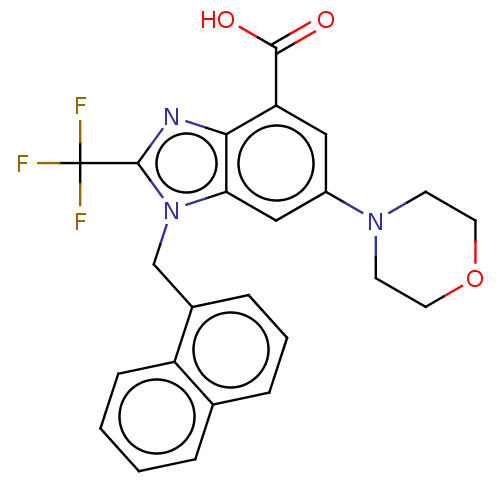
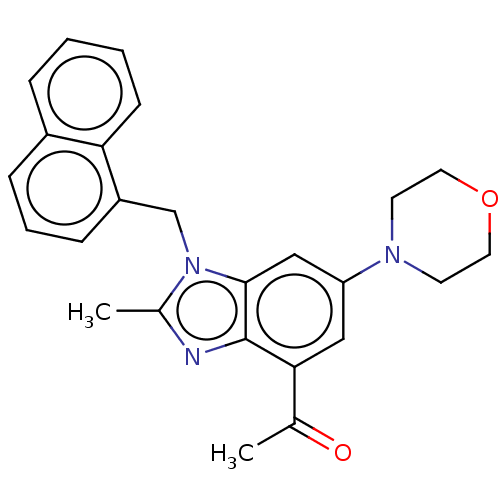
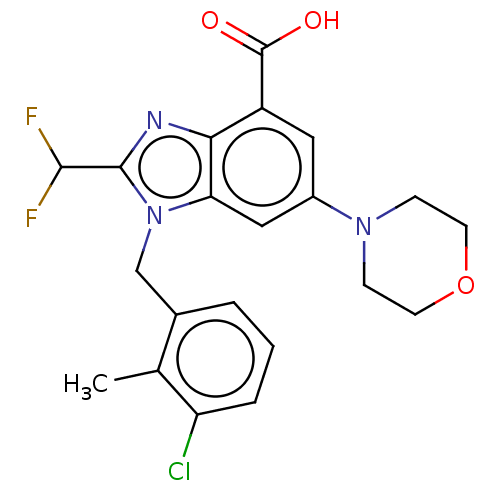
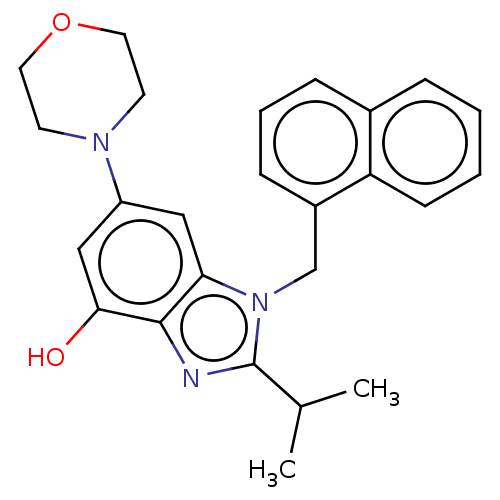
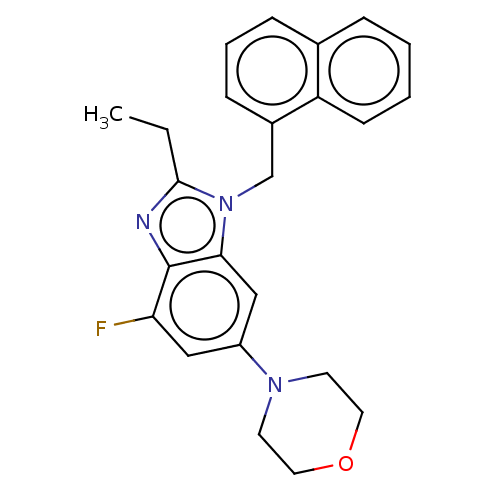
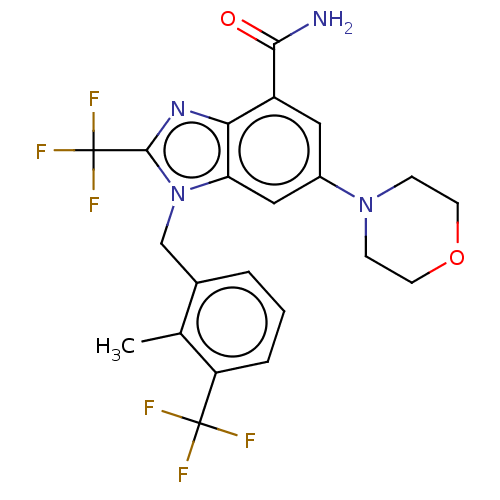
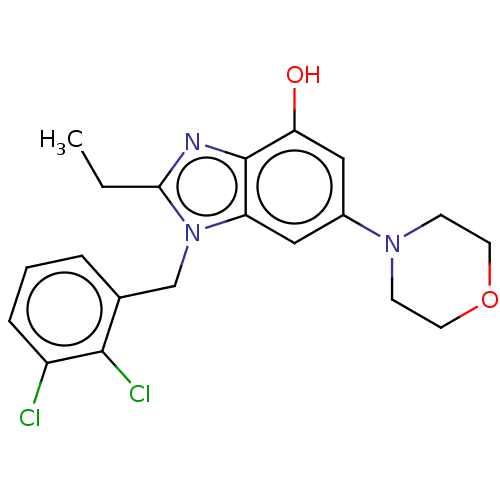
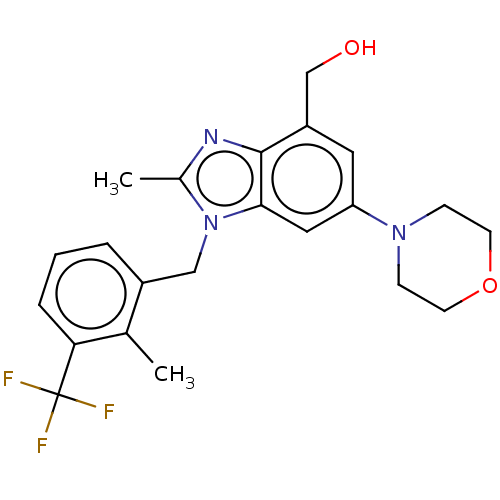
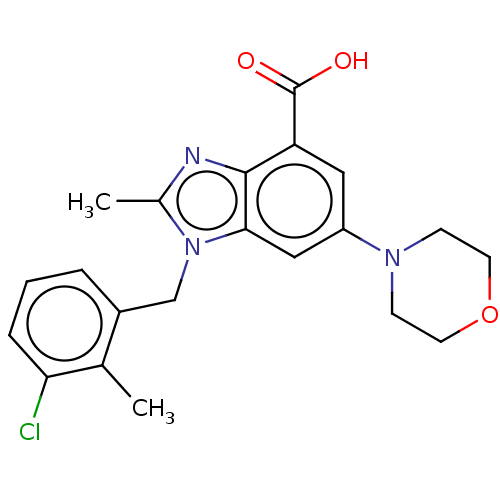
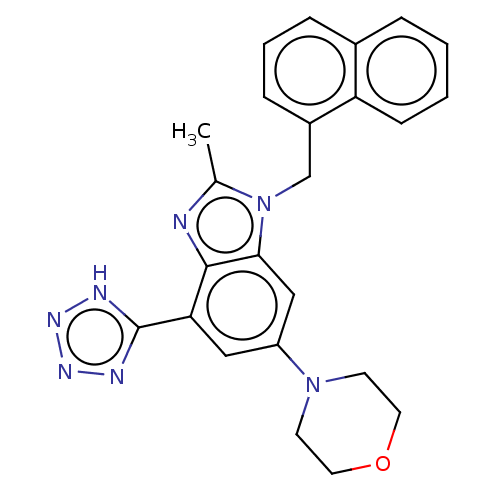
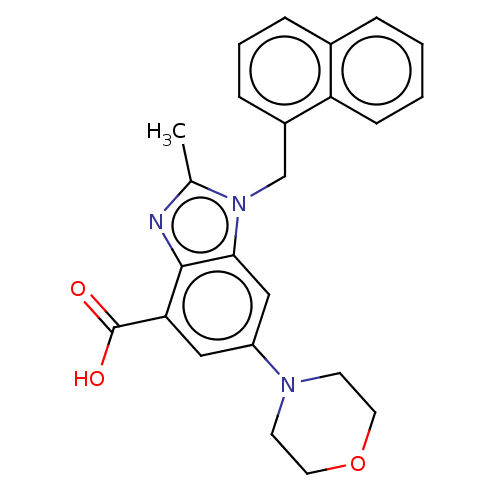
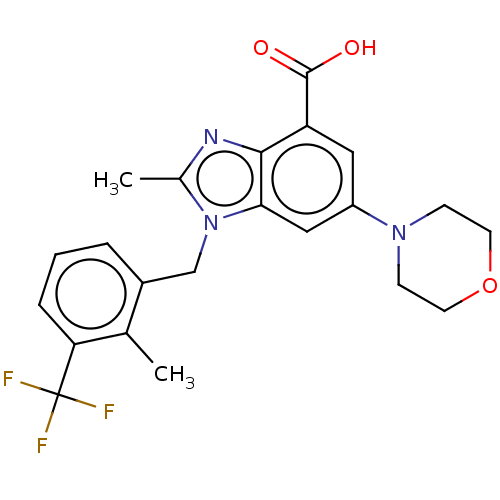
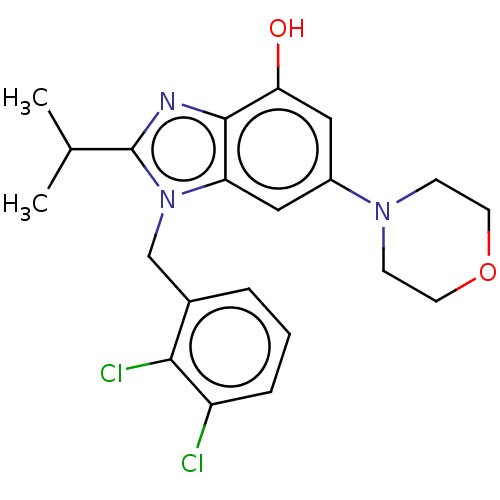
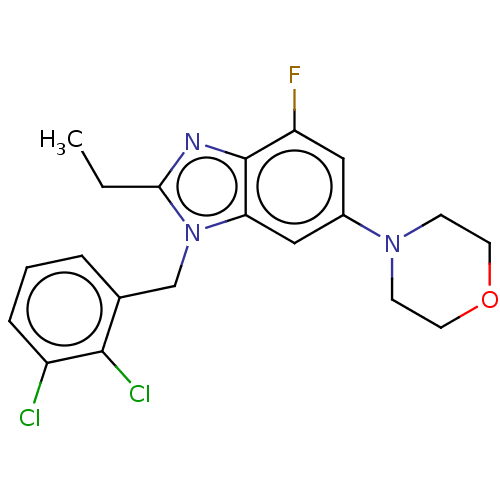
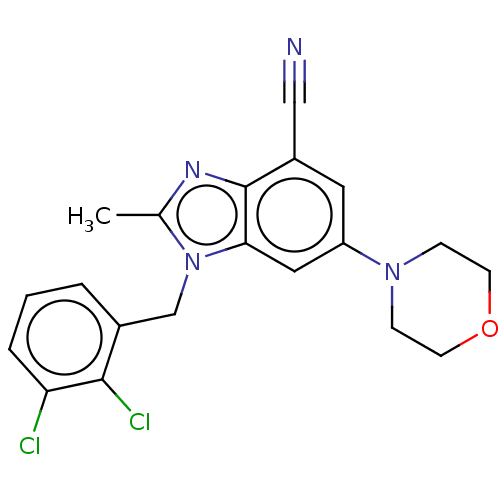
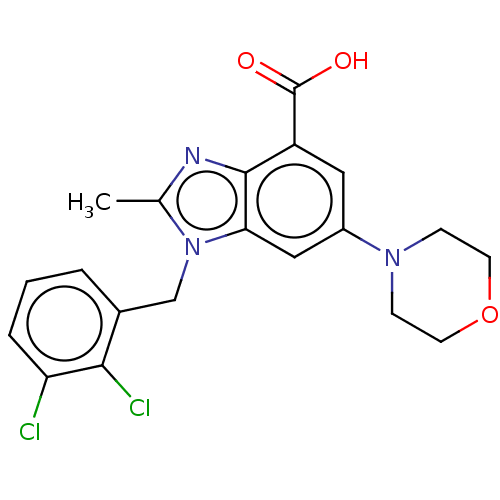
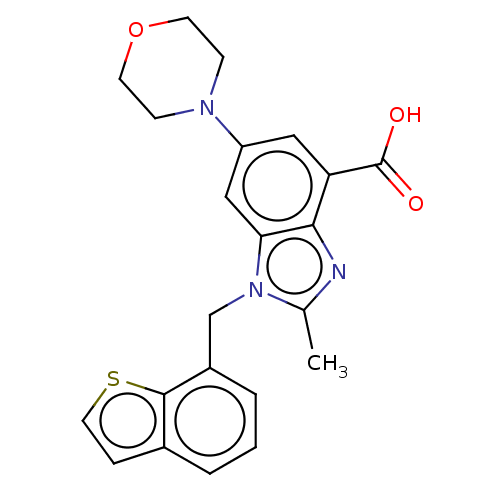
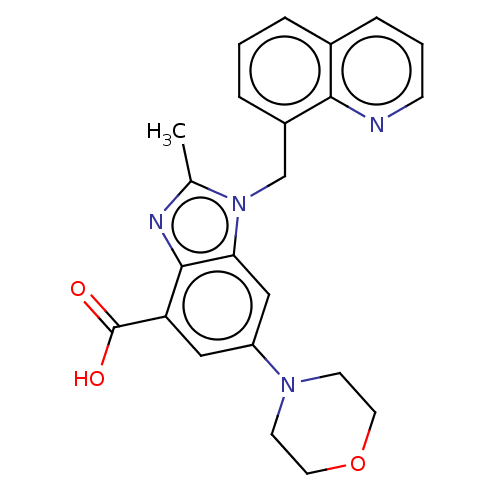
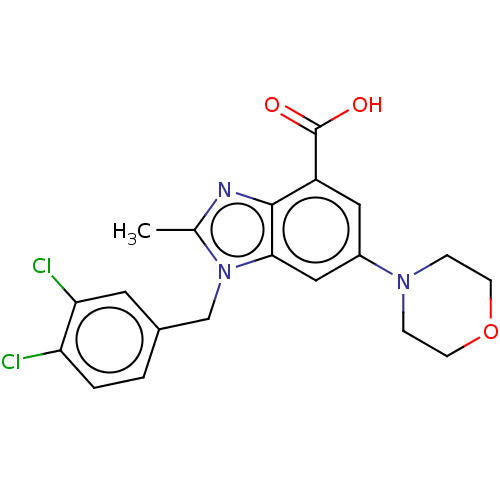
Report error Found 27 Enz. Inhib. hit(s) with all data for entry = 9131
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
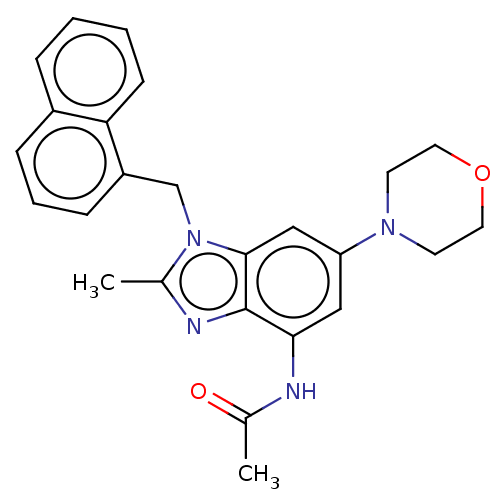
Affinity DataIC50: 0.200nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 0.251nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 0.501nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 0.631nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1.26nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1.58nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1.58nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 1.58nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 2nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 2.51nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 2.51nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 3.16nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 5.01nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 6.31nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 6.31nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 6.31nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 6.31nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 7.94nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 10nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 12.6nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 15.8nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 25.1nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 63.1nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 100nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta(Human)
Glaxosmithkline
US Patent
Glaxosmithkline
US Patent
Affinity DataIC50: 200nMAssay Description:PI3Kinase Reaction Buffer was prepared by diluting the stock 1:4 with de-ionized water. Freshly prepared DTT was added at a final concentration of 5 ...More data for this Ligand-Target Pair