Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prostaglandin G/H synthase 2
Ligand
BDBM50057002
Substrate
n/a
Meas. Tech.
ChEMBL_159740 (CHEMBL762904)
IC50
510±n/a nM
Citation
 Lazer, ES; Miao, CK; Cywin, CL; Sorcek, R; Wong, HC; Meng, Z; Potocki, I; Hoermann, M; Snow, RJ; Tschantz, MA; Kelly, TA; McNeil, DW; Coutts, SJ; Churchill, L; Graham, AG; David, E; Grob, PM; Engel, W; Meier, H; Trummlitz, G Effect of structural modification of enol-carboxamide-type nonsteroidal antiinflammatory drugs on COX-2/COX-1 selectivity. J Med Chem 40:980-9 (1997) [PubMed] Article
Lazer, ES; Miao, CK; Cywin, CL; Sorcek, R; Wong, HC; Meng, Z; Potocki, I; Hoermann, M; Snow, RJ; Tschantz, MA; Kelly, TA; McNeil, DW; Coutts, SJ; Churchill, L; Graham, AG; David, E; Grob, PM; Engel, W; Meier, H; Trummlitz, G Effect of structural modification of enol-carboxamide-type nonsteroidal antiinflammatory drugs on COX-2/COX-1 selectivity. J Med Chem 40:980-9 (1997) [PubMed] Article More Info.:
Target
Name:
Prostaglandin G/H synthase 2
Synonyms:
COX2 | Cyclooxygenase | Cyclooxygenase 2 (COX-2) | Cyclooxygenase-2 | Cyclooxygenase-2 (COX-2 AA) | Cyclooxygenase-2 (COX-2 AEA) | Cyclooxygenase-2 (COX-2) | PGH synthase 2 | PGH2_HUMAN | PGHS-2 | PHS II | PTGS2 | Prostaglandin E synthase/G/H synthase 2 | Prostaglandin H2 synthase 2 | Prostaglandin-endoperoxide synthase 2
Type:
Enzyme
Mol. Mass.:
69003.89
Organism:
Human
Description:
Recombinant Cox-2 provided by Cayman (Cayman Chemical Co.,Ann Arbor, MI).
Residue:
604
Sequence:
MLARALLLCAVLALSHTANPCCSHPCQNRGVCMSVGFDQYKCDCTRTGFYGENCSTPEFLTRIKLFLKPTPNTVHYILTHFKGFWNVVNNIPFLRNAIMSYVLTSRSHLIDSPPTYNADYGYKSWEAFSNLSYYTRALPPVPDDCPTPLGVKGKKQLPDSNEIVEKLLLRRKFIPDPQGSNMMFAFFAQHFTHQFFKTDHKRGPAFTNGLGHGVDLNHIYGETLARQRKLRLFKDGKMKYQIIDGEMYPPTVKDTQAEMIYPPQVPEHLRFAVGQEVFGLVPGLMMYATIWLREHNRVCDVLKQEHPEWGDEQLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNKQFQYQNRIAAEFNTLYHWHPLLPDTFQIHDQKYNYQQFIYNNSILLEHGITQFVESFTRQIAGRVAGGRNVPPAVQKVSQASIDQSRQMKYQSFNEYRKRFMLKPYESFEELTGEKEMSAELEALYGDIDAVELYPALLVEKPRPDAIFGETMVEVGAPFSLKGLMGNVICSPAYWKPSTFGGEVGFQIINTASIQSLICNNVKGCPFTSFSVPDPELIKTVTINASSSRSGLDDINPTVLLKERSTEL
Inhibitor
Name:
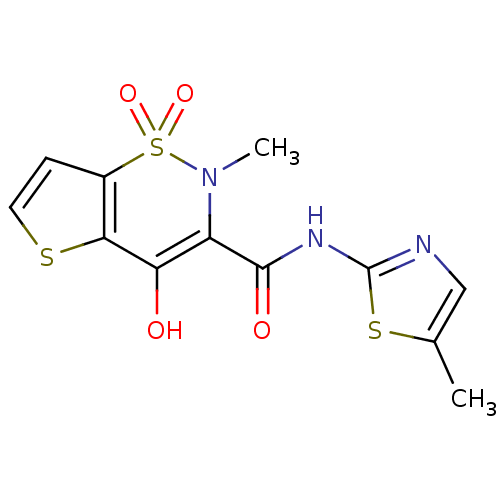
BDBM50057002
Synonyms:
4-Hydroxy-2-methyl-1,1-dioxo-1,2-dihydro-1lambda*6*-thieno[2,3-e][1,2]thiazine-3-carboxylic acid (5-methyl-thiazol-2-yl)-amide | CHEMBL367739
Type:
Small organic molecule
Emp. Form.:
C12H11N3O4S3
Mol. Mass.:
357.428
SMILES:
CN1C(C(=O)Nc2ncc(C)s2)=C(O)c2sccc2S1(=O)=O |t:12|