Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
RAC-alpha serine/threonine-protein kinase
Ligand
BDBM50340304
Substrate
n/a
Meas. Tech.
ChEMBL_741473 (CHEMBL1763788)
IC50
2±n/a nM
Citation
 Xu, R; Banka, A; Blake, JF; Mitchell, IS; Wallace, EM; Bencsik, JR; Kallan, NC; Spencer, KL; Gloor, SL; Martinson, M; Risom, T; Gross, SD; Morales, TH; Wu, WI; Vigers, GP; Brandhuber, BJ; Skelton, NJ Discovery of spirocyclic sulfonamides as potent Akt inhibitors with exquisite selectivity against PKA. Bioorg Med Chem Lett 21:2335-40 (2011) [PubMed] Article
Xu, R; Banka, A; Blake, JF; Mitchell, IS; Wallace, EM; Bencsik, JR; Kallan, NC; Spencer, KL; Gloor, SL; Martinson, M; Risom, T; Gross, SD; Morales, TH; Wu, WI; Vigers, GP; Brandhuber, BJ; Skelton, NJ Discovery of spirocyclic sulfonamides as potent Akt inhibitors with exquisite selectivity against PKA. Bioorg Med Chem Lett 21:2335-40 (2011) [PubMed] Article More Info.:
Target
Name:
RAC-alpha serine/threonine-protein kinase
Synonyms:
AKT phosphorylation (p-AKT) | AKT1 | AKT1/PPP1CA | AKT1_HUMAN | C-AKT | PKB | PKB alpha | Protein kinase Akt-1 | Protein kinase B | Protein kinase B (AKT1) | Protein kinase B (Akt 1) | Protein kinase B (Akt) | Protein kinase B alpha | Protein kinase B alpha (AKT1) | Proto-oncogene Akt (Akt1) | Proto-oncogene c-Akt (AKT) | Proto-oncogene c-Akt (AKT1) | RAC | RAC-PK-alpha | RAC-alpha serine/threonine-protein kinase (AKT) | RAC-alpha serine/threonine-protein kinase (AKT1) | RAC-alpha serine/threonine-protein kinase (pAKT)
Type:
Enzyme
Mol. Mass.:
55681.25
Organism:
Homo sapiens (Human)
Description:
P31749
Residue:
480
Sequence:
MSDVAIVKEGWLHKRGEYIKTWRPRYFLLKNDGTFIGYKERPQDVDQREAPLNNFSVAQCQLMKTERPRPNTFIIRCLQWTTVIERTFHVETPEEREEWTTAIQTVADGLKKQEEEEMDFRSGSPSDNSGAEEMEVSLAKPKHRVTMNEFEYLKLLGKGTFGKVILVKEKATGRYYAMKILKKEVIVAKDEVAHTLTENRVLQNSRHPFLTALKYSFQTHDRLCFVMEYANGGELFFHLSRERVFSEDRARFYGAEIVSALDYLHSEKNVVYRDLKLENLMLDKDGHIKITDFGLCKEGIKDGATMKTFCGTPEYLAPEVLEDNDYGRAVDWWGLGVVMYEMMCGRLPFYNQDHEKLFELILMEEIRFPRTLGPEAKSLLSGLLKKDPKQRLGGGSEDAKEIMQHRFFAGIVWQHVYEKKLSPPFKPQVTSETDTRYFDEEFTAQMITITPPDQDDSMECVDSERRPHFPQFSYSASGTA
Inhibitor
Name:
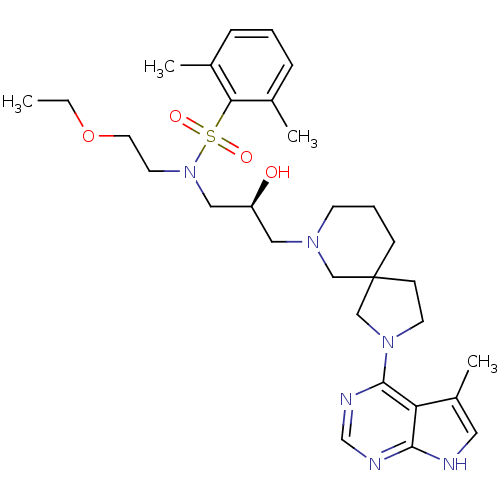
BDBM50340304
Synonyms:
CHEMBL1760759 | N-(2-ethoxyethyl)-N-((2S)-2-hydroxy-3-(2-(5-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-yl)-2,7-diazaspiro[4.5]decan-7-yl)propyl)-2,6-dimethylbenzenesulfonamide
Type:
Small organic molecule
Emp. Form.:
C30H44N6O4S
Mol. Mass.:
584.773
SMILES:
CCOCCN(C[C@@H](O)CN1CCCC2(CCN(C2)c2ncnc3[nH]cc(C)c23)C1)S(=O)(=O)c1c(C)cccc1C |r|