Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glycogen synthase kinase-3 beta
Ligand
BDBM188515
Substrate
n/a
Meas. Tech.
Mobility Shift Microfluidics Assay
pH
7.5±n/a
Temperature
298.15±n/a K
IC50
5± 3 nM
Comments
extracted
Citation
 Wagner, FF; Bishop, JA; Gale, JP; Shi, X; Walk, M; Ketterman, J; Patnaik, D; Barker, D; Walpita, D; Campbell, AJ; Nguyen, S; Lewis, M; Ross, L; We�wer, M; An, WF; Germain, AR; Nag, PP; Metkar, S; Kaya, T; Dandapani, S; Olson, DE; Barbe, AL; Lazzaro, F; Sacher, JR; Cheah, JH; Fei, D; Perez, J; Munoz, B; Palmer, M; Stegmaier, K; Schreiber, SL; Scolnick, E; Zhang, YL; Haggarty, SJ; Holson, EB; Pan, JQ Inhibitors of Glycogen Synthase Kinase 3 with Exquisite Kinome-Wide Selectivity and Their Functional Effects. ACS Chem Biol 11:1952-63 (2016) [PubMed] Article
Wagner, FF; Bishop, JA; Gale, JP; Shi, X; Walk, M; Ketterman, J; Patnaik, D; Barker, D; Walpita, D; Campbell, AJ; Nguyen, S; Lewis, M; Ross, L; We�wer, M; An, WF; Germain, AR; Nag, PP; Metkar, S; Kaya, T; Dandapani, S; Olson, DE; Barbe, AL; Lazzaro, F; Sacher, JR; Cheah, JH; Fei, D; Perez, J; Munoz, B; Palmer, M; Stegmaier, K; Schreiber, SL; Scolnick, E; Zhang, YL; Haggarty, SJ; Holson, EB; Pan, JQ Inhibitors of Glycogen Synthase Kinase 3 with Exquisite Kinome-Wide Selectivity and Their Functional Effects. ACS Chem Biol 11:1952-63 (2016) [PubMed] Article More Info.:
Target
Name:
Glycogen synthase kinase-3 beta
Synonyms:
GSK-3 beta | GSK-3, beta | GSK3B | GSK3B_HUMAN | Glycogen synthase kinase 3 beta (GSK3B) | Glycogen synthase kinase 3-beta (GSK3B) | Glycogen synthase kinase-3 beta (GSK-3B) | Glycogen synthase kinase-3 beta (GSK3 Beta) | Glycogen synthase kinase-3 beta (GSK3B) | Glycogen synthase kinase-3B (GSK-3B) | Glycogen synthase kinase-3beta (GSK3B) | Serine/threonine-protein kinase GSK3B
Type:
Enzyme
Mol. Mass.:
46756.38
Organism:
Homo sapiens (Human)
Description:
P49841
Residue:
420
Sequence:
MSGRPRTTSFAESCKPVQQPSAFGSMKVSRDKDGSKVTTVVATPGQGPDRPQEVSYTDTKVIGNGSFGVVYQAKLCDSGELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFYSSGEKKDEVYLNLVLDYVPETVYRVARHYSRAKQTLPVIYVKLYMYQLFRSLAYIHSFGICHRDIKPQNLLLDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSSIDVWSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIKAHPWTKVFRPRTPPEAIALCSRLLEYTPTARLTPLEACAHSFFDELRDPNVKLPNGRDTPALFNFTTQELSSNPPLATILIPPHARIQAAASTPTNATAASDANTGDRGQTNNAASASASNST
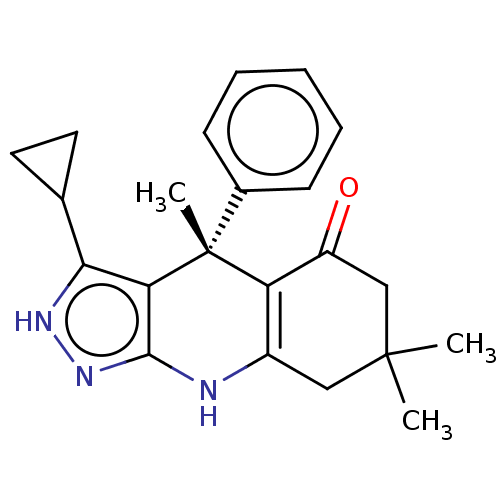
Inhibitor
Name:
BDBM188515
Synonyms:
(S)-3-cyclopropyl-4,7,7-trimethyl-4-phenyl-2,4,6,7,8,9-hexahydro-5H-pyrazolo[3,4-b]quinolin-5-one (BRD0209) | US10137122, Compound 114
Type:
Small organic molecule
Emp. Form.:
C22H25N3O
Mol. Mass.:
347.4534
SMILES:
CC1(C)CC(=O)C2=C(C1)Nc1n[nH]c(C3CC3)c1[C@]2(C)c1ccccc1 |r,c:6|