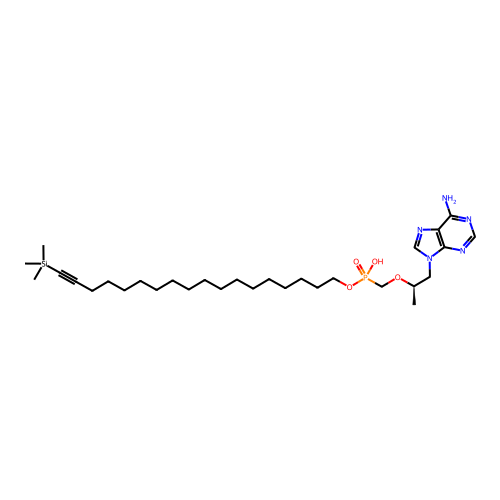
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50587157
Substrate
n/a
Meas. Tech.
ChEMBL_2171279 (CHEMBL5056413)
IC50
27000±n/a nM
Citation
 Pribut, N; D'Erasmo, M; Dasari, M; Giesler, KE; Iskandar, S; Sharma, SK; Bartsch, PW; Raghuram, A; Bushnev, A; Hwang, SS; Burton, SL; Derdeyn, CA; Basson, AE; Liotta, DC; Miller, EJ ?-Functionalized Lipid Prodrugs of HIV NtRTI Tenofovir with Enhanced Pharmacokinetic Properties. J Med Chem 64:12917-12937 (2021) [PubMed] Article
Pribut, N; D'Erasmo, M; Dasari, M; Giesler, KE; Iskandar, S; Sharma, SK; Bartsch, PW; Raghuram, A; Bushnev, A; Hwang, SS; Burton, SL; Derdeyn, CA; Basson, AE; Liotta, DC; Miller, EJ ?-Functionalized Lipid Prodrugs of HIV NtRTI Tenofovir with Enhanced Pharmacokinetic Properties. J Med Chem 64:12917-12937 (2021) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
Inhibitor
Name:
BDBM50587157
Synonyms:
CHEMBL5080093
Type:
Small organic molecule
Emp. Form.:
C30H57N6O4PSi
Mol. Mass.:
624.8706
SMILES:
[#7].[#6]-[#6@H](-[#6]-n1cnc2c(-[#7])ncnc12)-[#8]-[#6]P([#8])(=O)[#8]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]C#C[Si;v4]([#6])([#6])[#6] |r|