Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Protein arginine N-methyltransferase 1
Ligand
BDBM50009672
Substrate
n/a
Meas. Tech.
ChEMBL_2210864 (CHEMBL5123813)
IC50
250±n/a nM
Citation
 Menna, M; Fiorentino, F; Marrocco, B; Lucidi, A; Tomassi, S; Cilli, D; Romanenghi, M; Cassandri, M; Pomella, S; Pezzella, M; Del Bufalo, D; Zeya Ansari, MS; Toma?evi?, N; Mladenovi?, M; Viviano, M; Sbardella, G; Rota, R; Trisciuoglio, D; Minucci, S; Mattevi, A; Rotili, D; Mai, A Novel non-covalent LSD1 inhibitors endowed with anticancer effects in leukemia and solid tumor cellular models. Eur J Med Chem 237:0 (2022) [PubMed]
Menna, M; Fiorentino, F; Marrocco, B; Lucidi, A; Tomassi, S; Cilli, D; Romanenghi, M; Cassandri, M; Pomella, S; Pezzella, M; Del Bufalo, D; Zeya Ansari, MS; Toma?evi?, N; Mladenovi?, M; Viviano, M; Sbardella, G; Rota, R; Trisciuoglio, D; Minucci, S; Mattevi, A; Rotili, D; Mai, A Novel non-covalent LSD1 inhibitors endowed with anticancer effects in leukemia and solid tumor cellular models. Eur J Med Chem 237:0 (2022) [PubMed] More Info.:
Target
Name:
Protein arginine N-methyltransferase 1
Synonyms:
2.1.1.319 | ANM1_HUMAN | HMT2 | HRMT1L2 | HRMT1L2 {ECO:0000303|PubMed:11097842 | Histone-arginine N-methyltransferase PRMT1 | IR1B4 | Interferon receptor 1-bound protein 4 | PRMT1 | Protein arginine N-methyltransferase 1 | Protein-arginine N-methyltransferase 1 | Synonyms=HMT2
Type:
PROTEIN
Mol. Mass.:
42451.61
Organism:
Homo sapiens
Description:
ChEMBL_100878
Residue:
371
Sequence:
MAAAEAANCIMENFVATLANGMSLQPPLEEVSCGQAESSEKPNAEDMTSKDYYFDSYAHFGIHEEMLKDEVRTLTYRNSMFHNRHLFKDKVVLDVGSGTGILCMFAAKAGARKVIGIECSSISDYAVKIVKANKLDHVVTIIKGKVEEVELPVEKVDIIISEWMGYCLFYESMLNTVLYARDKWLAPDGLIFPDRATLYVTAIEDRQYKDYKIHWWENVYGFDMSCIKDVAIKEPLVDVVDPKQLVTNACLIKEVDIYTVKVEDLTFTSPFCLQVKRNDYVHALVAYFNIEFTRCHKRTGFSTSPESPYTHWKQTVFYMEDYLTVKTGEEIFGTIGMRPNAKNNRDLDFTIDLDFKGQLCELSCSTDYRMR
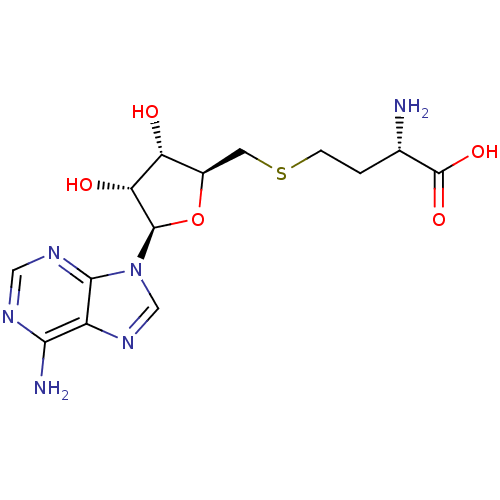
Inhibitor
Name:
BDBM50009672
Synonyms:
AdoHcy | CHEMBL418052 | S-(5'-adenosyl)-L-homocysteine | S-(5'-deoxyadenosin-5'-yl)-L-homocysteine | S-[1-(adenin-9-yl)-1,5-dideoxy-beta-D-ribofuranos-5-yl]-L-homocysteine | S-adenosyl-L-homocysteine | SAH | US8895245, S-Adenosyl-L-homocysteine (SAH) | US9175331, 1 | US9333217, S-Adenosyl-L-homocysteine (SAH)
Type:
Small organic molecule
Emp. Form.:
C14H20N6O5S
Mol. Mass.:
384.411
SMILES:
N[C@@H](CCSC[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O