Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Sodium/glucose cotransporter 1
Ligand
BDBM20880
Substrate
n/a
Meas. Tech.
ChEMBL_864703 (CHEMBL2174861)
IC50
800±n/a nM
Citation
 Ohtake, Y; Sato, T; Kobayashi, T; Nishimoto, M; Taka, N; Takano, K; Yamamoto, K; Ohmori, M; Yamaguchi, M; Takami, K; Yeu, SY; Ahn, KH; Matsuoka, H; Morikawa, K; Suzuki, M; Hagita, H; Ozawa, K; Yamaguchi, K; Kato, M; Ikeda, S Discovery of tofogliflozin, a novel C-arylglucoside with an O-spiroketal ring system, as a highly selective sodium glucose cotransporter 2 (SGLT2) inhibitor for the treatment of type 2 diabetes. J Med Chem 55:7828-40 (2012) [PubMed] Article
Ohtake, Y; Sato, T; Kobayashi, T; Nishimoto, M; Taka, N; Takano, K; Yamamoto, K; Ohmori, M; Yamaguchi, M; Takami, K; Yeu, SY; Ahn, KH; Matsuoka, H; Morikawa, K; Suzuki, M; Hagita, H; Ozawa, K; Yamaguchi, K; Kato, M; Ikeda, S Discovery of tofogliflozin, a novel C-arylglucoside with an O-spiroketal ring system, as a highly selective sodium glucose cotransporter 2 (SGLT2) inhibitor for the treatment of type 2 diabetes. J Med Chem 55:7828-40 (2012) [PubMed] Article More Info.:
Target
Name:
Sodium/glucose cotransporter 1
Synonyms:
High affinity sodium-glucose cotransporter | NAGT | Na(+)/glucose cotransporter 1 | SC5A1_HUMAN | SGLT1 | SLC5A1 | Sodium-dependent glucose cotransporter 1 (SGLT1) | Sodium/glucose cotransporter 1 | Sodium/glucose cotransporter 1 (SGLT1) | Solute carrier family 5 member 1
Type:
Protein
Mol. Mass.:
73503.78
Organism:
Homo sapiens (Human)
Description:
P13866
Residue:
664
Sequence:
MDSSTWSPKTTAVTRPVETHELIRNAADISIIVIYFVVVMAVGLWAMFSTNRGTVGGFFLAGRSMVWWPIGASLFASNIGSGHFVGLAGTGAASGIAIGGFEWNALVLVVVLGWLFVPIYIKAGVVTMPEYLRKRFGGQRIQVYLSLLSLLLYIFTKISADIFSGAIFINLALGLNLYLAIFLLLAITALYTITGGLAAVIYTDTLQTVIMLVGSLILTGFAFHEVGGYDAFMEKYMKAIPTIVSDGNTTFQEKCYTPRADSFHIFRDPLTGDLPWPGFIFGMSILTLWYWCTDQVIVQRCLSAKNMSHVKGGCILCGYLKLMPMFIMVMPGMISRILYTEKIACVVPSECEKYCGTKVGCTNIAYPTLVVELMPNGLRGLMLSVMLASLMSSLTSIFNSASTLFTMDIYAKVRKRASEKELMIAGRLFILVLIGISIAWVPIVQSAQSGQLFDYIQSITSYLGPPIAAVFLLAIFWKRVNEPGAFWGLILGLLIGISRMITEFAYGTGSCMEPSNCPTIICGVHYLYFAIILFAISFITIVVISLLTKPIPDVHLYRLCWSLRNSKEERIDLDAEEENIQEGPKETIEIETQVPEKKKGIFRRAYDLFCGLEQHGAPKMTEEEEKAMKMKMTDTSEKPLWRTVLNVNGIILVTVAVFCHAYFA
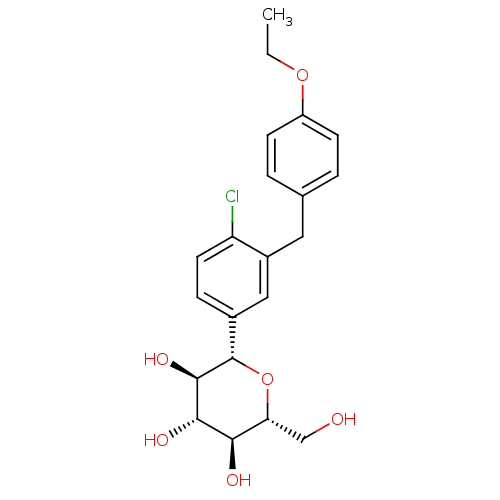
Inhibitor
Name:
BDBM20880
Synonyms:
(2S,3R,4R,5S,6R)-2-{4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl}-6-(hydroxymethyl)oxane-3,4,5-triol | BMS-512148 | C-aryl glucoside, 6 | CHEMBL429910 | Dapagliflozin
Type:
Small organic molecule
Emp. Form.:
C21H25ClO6
Mol. Mass.:
408.873
SMILES:
CCOc1ccc(Cc2cc(ccc2Cl)[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)cc1