Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
N-acylethanolamine-hydrolyzing acid amidase
Ligand
BDBM50151222
Substrate
n/a
Meas. Tech.
ChEBML_1558887
IC50
323±n/a nM
Citation
 Nuzzi, A; Fiasella, A; Ortega, JA; Pagliuca, C; Ponzano, S; Pizzirani, D; Bertozzi, SM; Ottonello, G; Tarozzo, G; Reggiani, A; Bandiera, T; Bertozzi, F; Piomelli, D Potenta-amino-�-lactam carbamic acid ester as NAAA inhibitors. Synthesis and structure-activity relationship (SAR) studies. Eur J Med Chem 111:138-59 (2016) [PubMed] Article
Nuzzi, A; Fiasella, A; Ortega, JA; Pagliuca, C; Ponzano, S; Pizzirani, D; Bertozzi, SM; Ottonello, G; Tarozzo, G; Reggiani, A; Bandiera, T; Bertozzi, F; Piomelli, D Potenta-amino-�-lactam carbamic acid ester as NAAA inhibitors. Synthesis and structure-activity relationship (SAR) studies. Eur J Med Chem 111:138-59 (2016) [PubMed] Article More Info.:
Target
Name:
N-acylethanolamine-hydrolyzing acid amidase
Synonyms:
ASAH-like protein | ASAHL | Acid ceramidase-like protein | N-acylethanolamine-hydrolyzing acid amidase | N-acylsphingosine amidohydrolase-like | N-acylsphingosine-amidohydrolase | NAAA | NAAA_HUMAN | PLT
Type:
Enzyme
Mol. Mass.:
40073.12
Organism:
Homo sapiens (Human)
Description:
Q02083
Residue:
359
Sequence:
MRTADREARPGLPSLLLLLLAGAGLSAASPPAAPRFNVSLDSVPELRWLPVLRHYDLDLVRAAMAQVIGDRVPKWVHVLIGKVVLELERFLPQPFTGEIRGMCDFMNLSLADCLLVNLAYESSVFCTSIVAQDSRGHIYHGRNLDYPFGNVLRKLTVDVQFLKNGQIAFTGTTFIGYVGLWTGQSPHKFTVSGDERDKGWWWENAIAALFRRHIPVSWLIRATLSESENFEAAVGKLAKTPLIADVYYIVGGTSPREGVVITRNRDGPADIWPLDPLNGAWFRVETNYDHWKPAPKEDDRRTSAIKALNATGQANLSLEALFQILSVVPVYNNFTIYTTVMSAGSPDKYMTRIRNPSRK
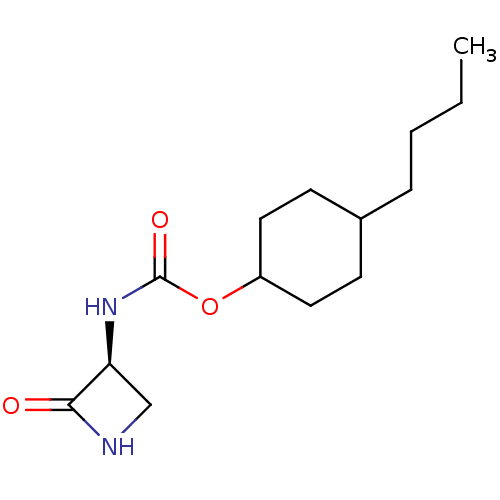
Inhibitor
Name:
BDBM50151222
Synonyms:
CHEMBL3770519
Type:
Small organic molecule
Emp. Form.:
C14H24N2O3
Mol. Mass.:
268.352
SMILES:
CCCCC1CCC(CC1)OC(=O)N[C@H]1CNC1=O |r,wU:14.14,(-6.41,.92,;-5.34,1.53,;-4,.77,;-2.67,1.54,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;,-1.54,;-1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;4,-5.4,;5.08,-6.44,;3.99,-7.52,;2.9,-6.43,;1.67,-6.41,)|