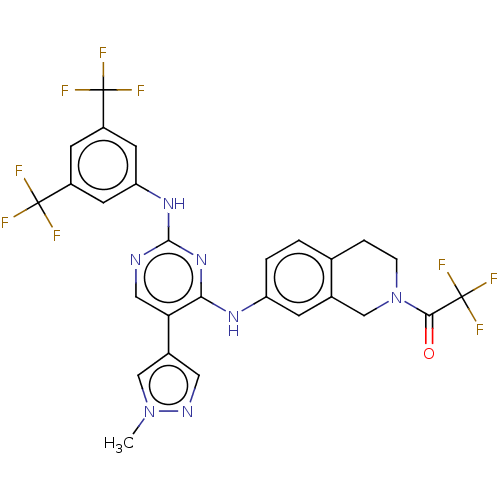
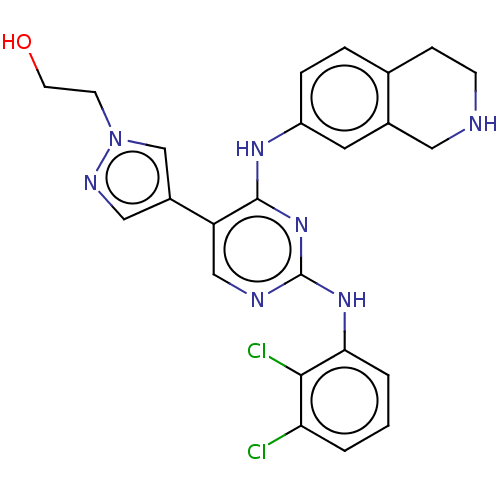
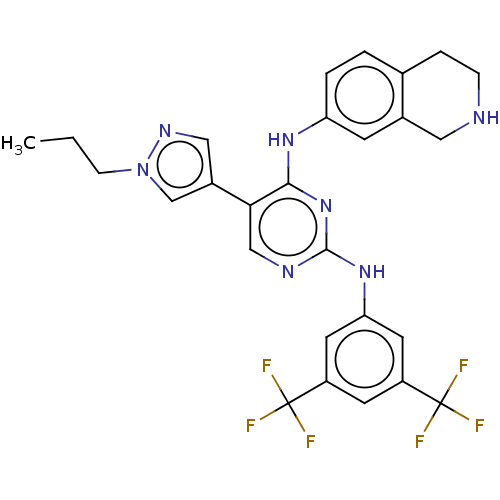
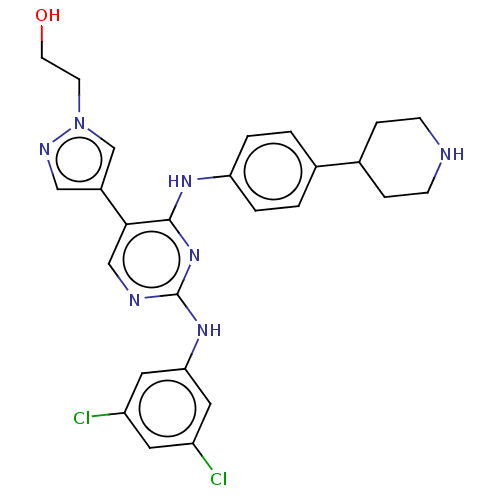
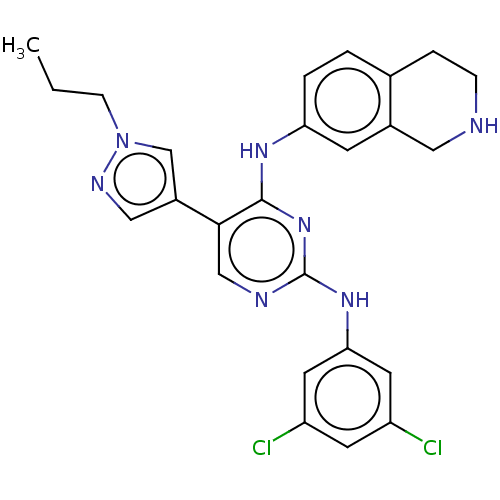
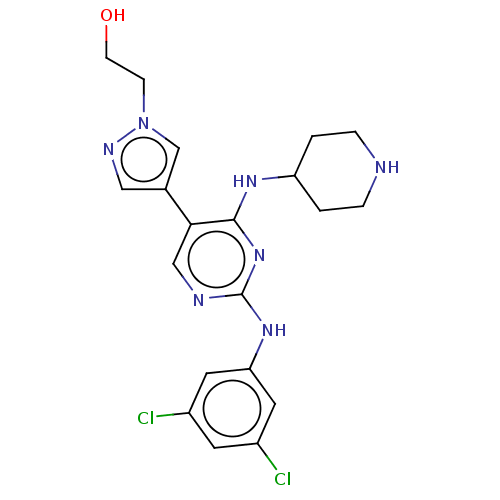
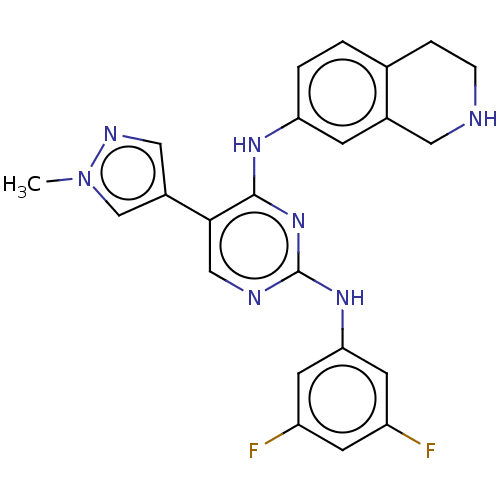
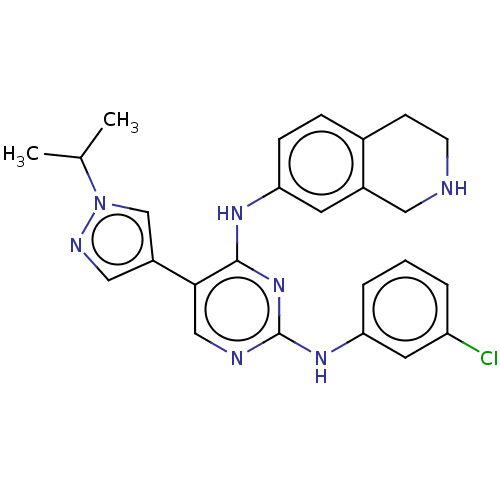
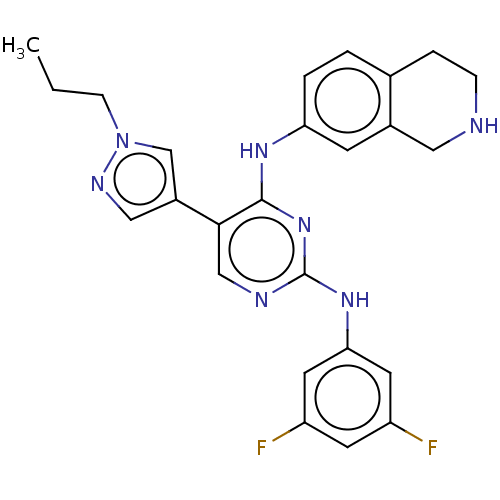
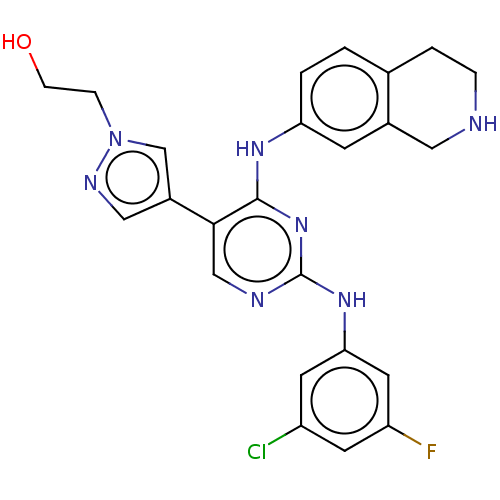
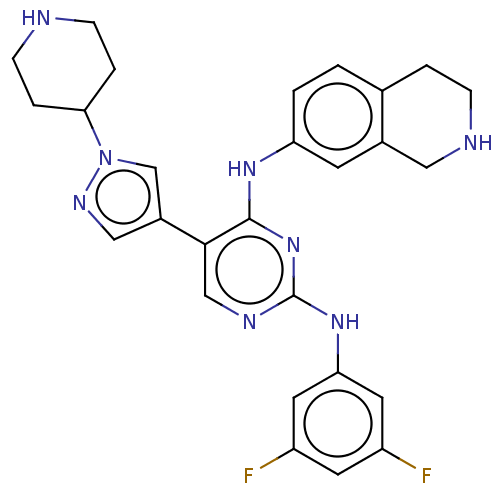
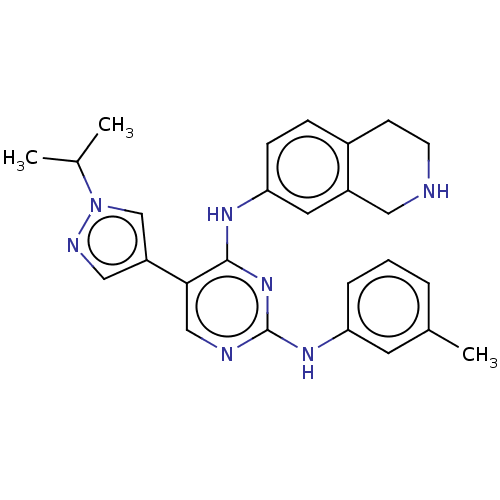
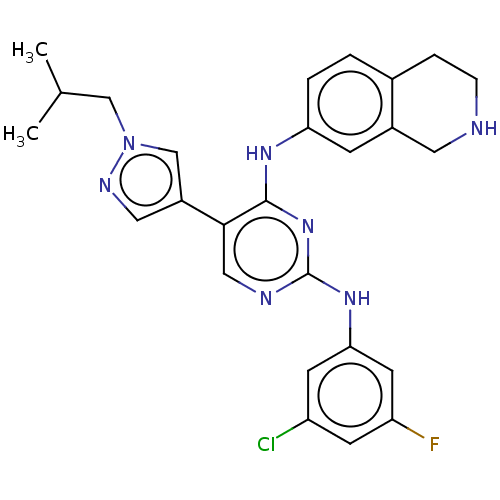
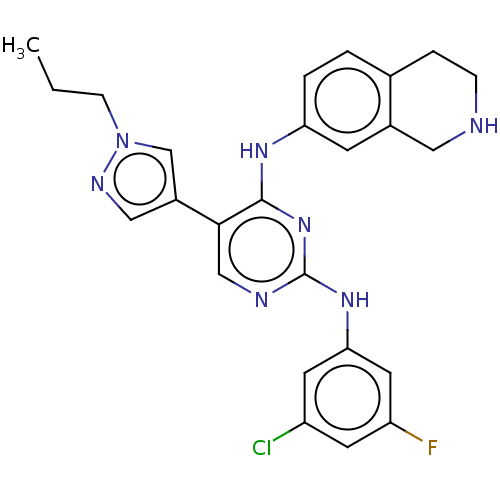
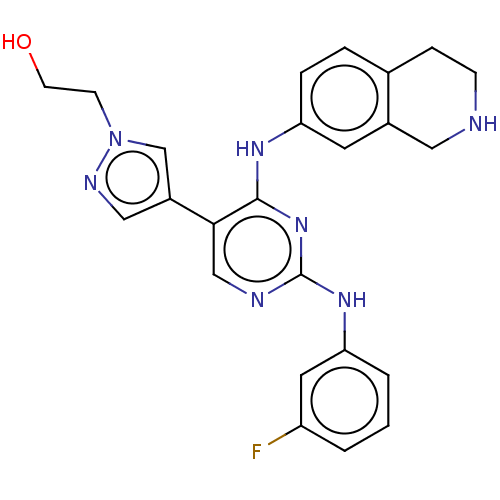
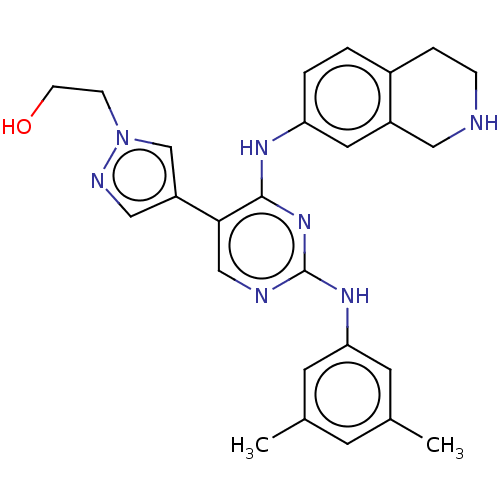
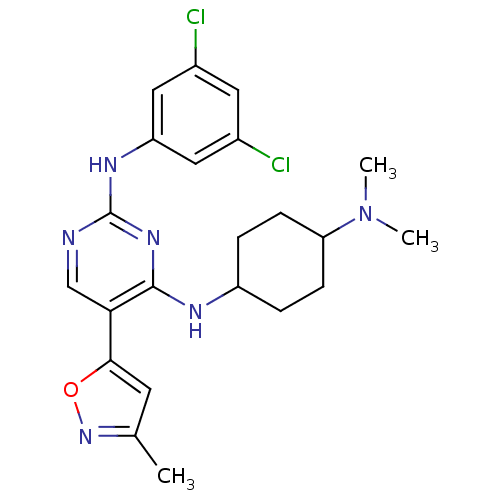
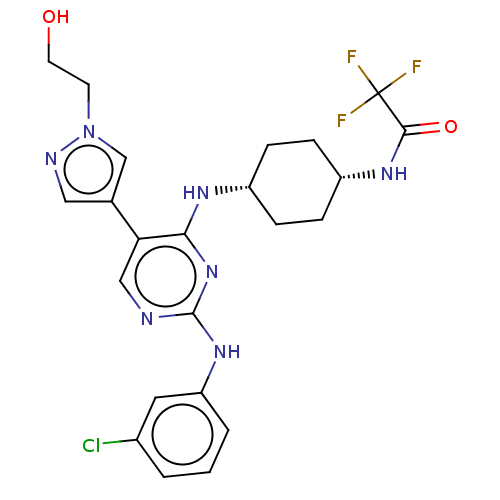
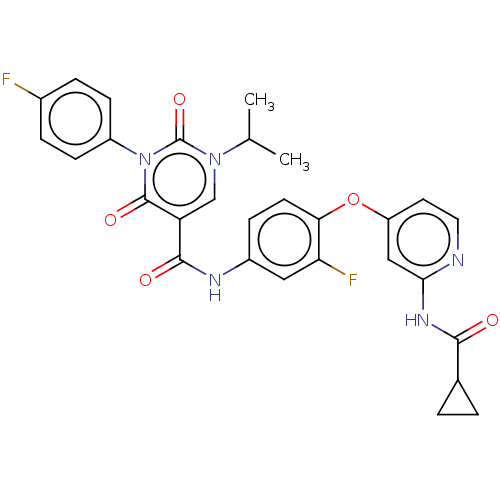
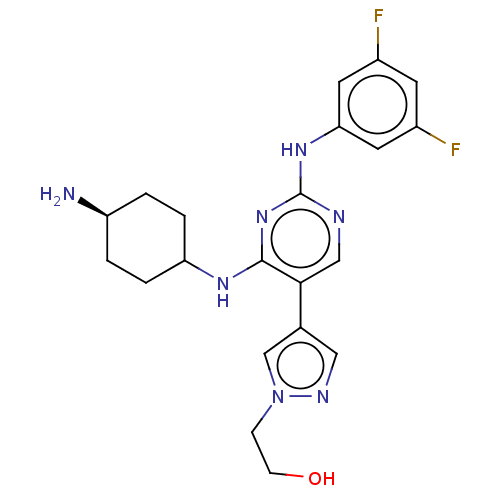
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.00600nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
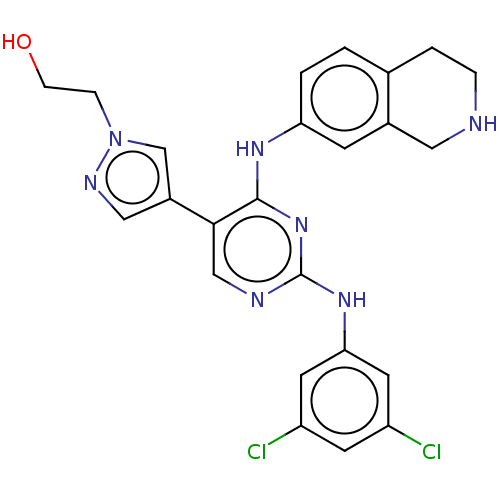
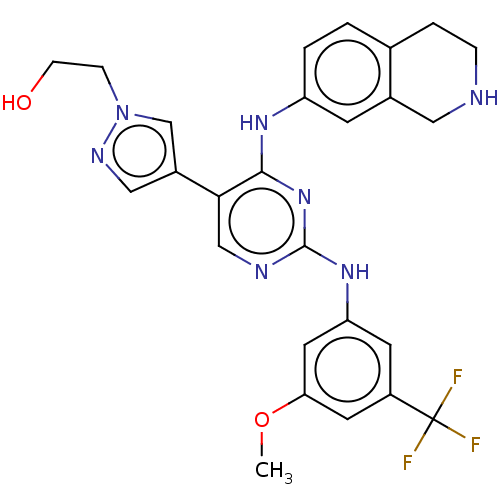
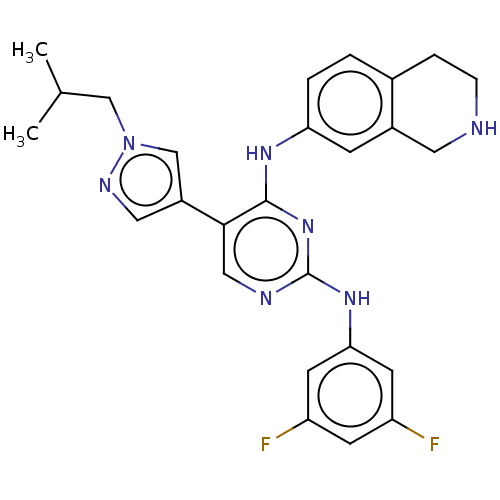
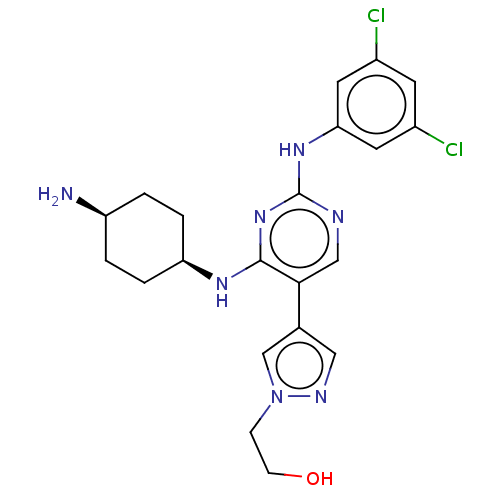
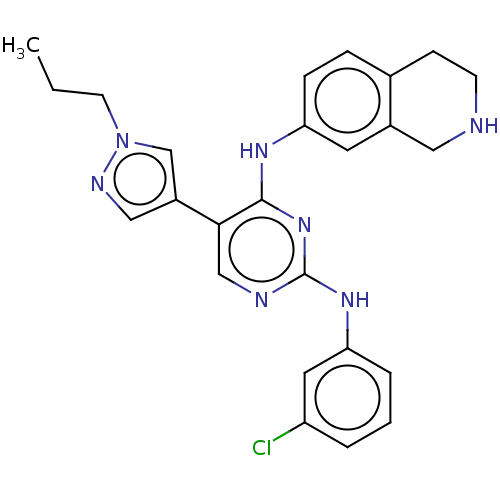
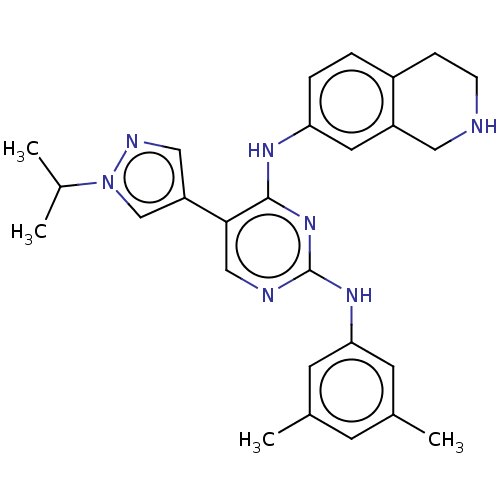
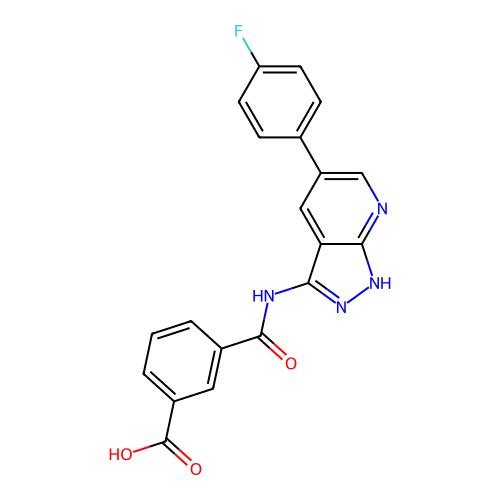
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
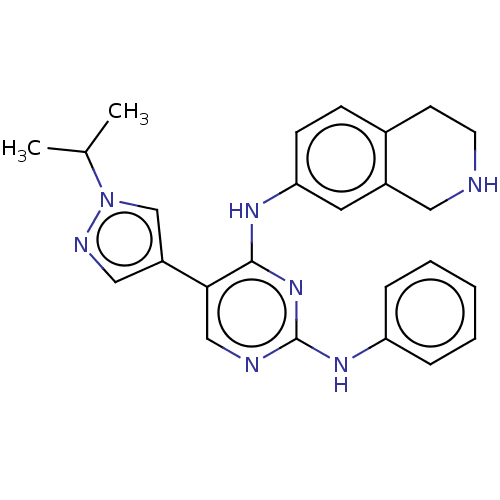
Affinity DataIC50: 0.0100nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
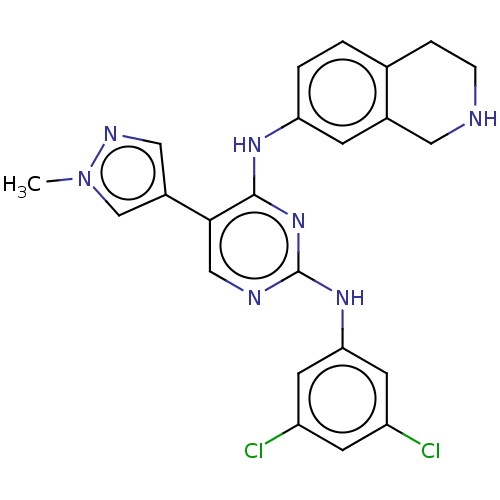
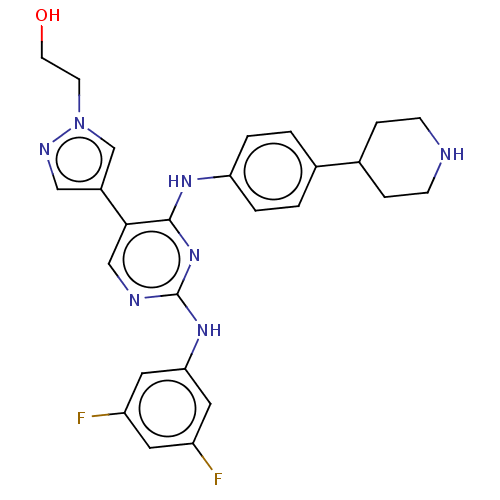
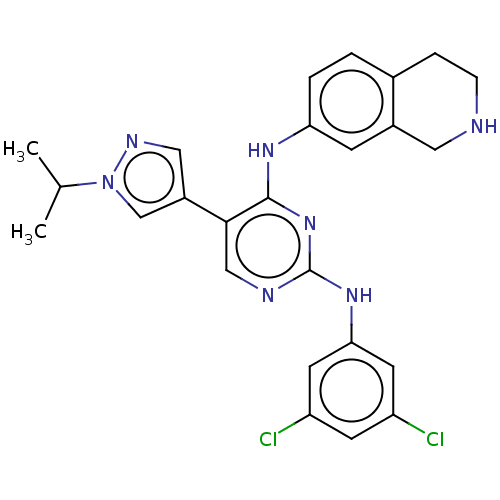
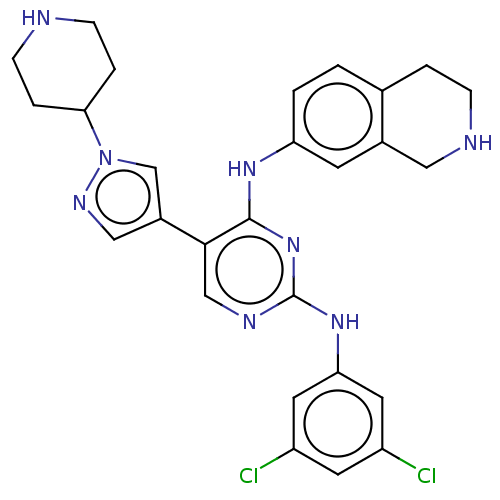
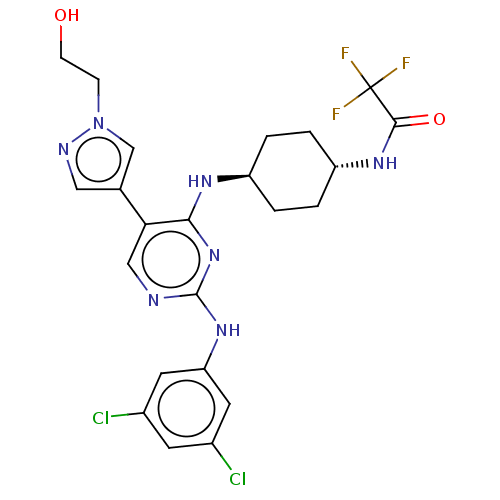
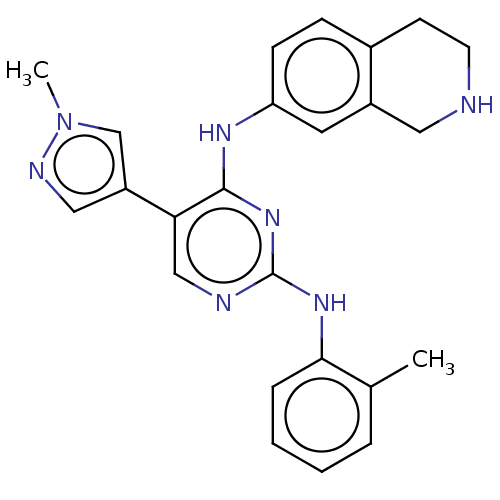
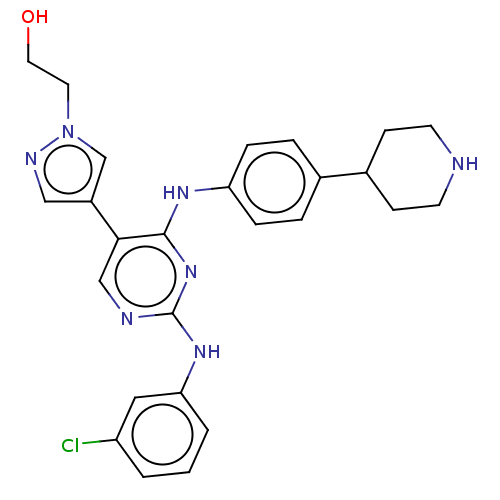
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0120nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
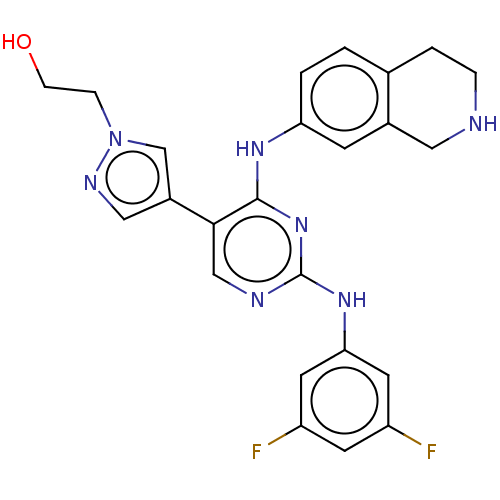
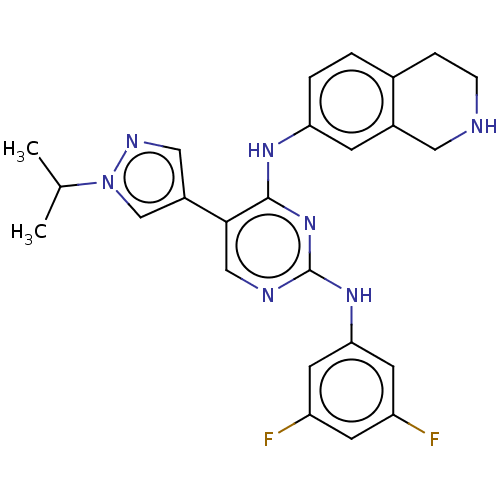
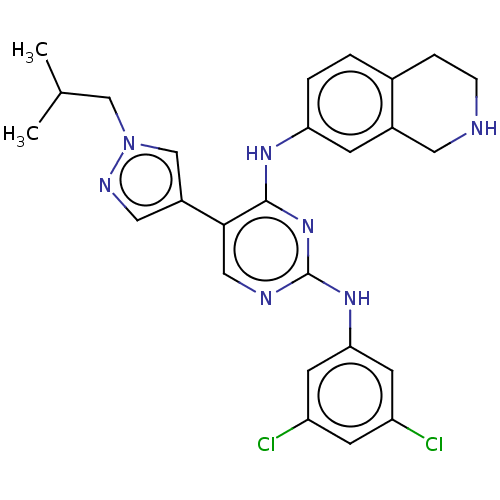
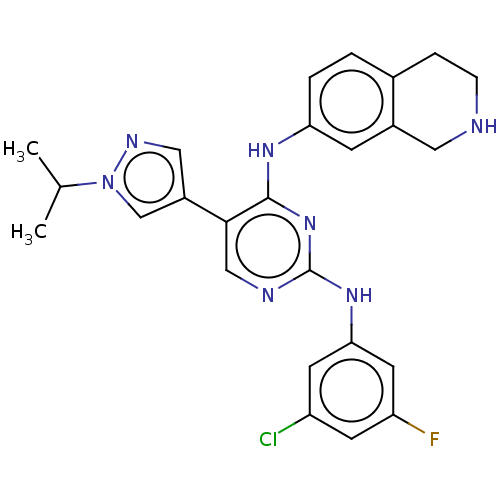
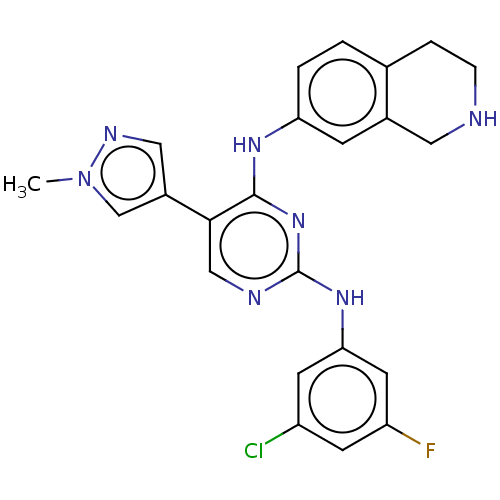
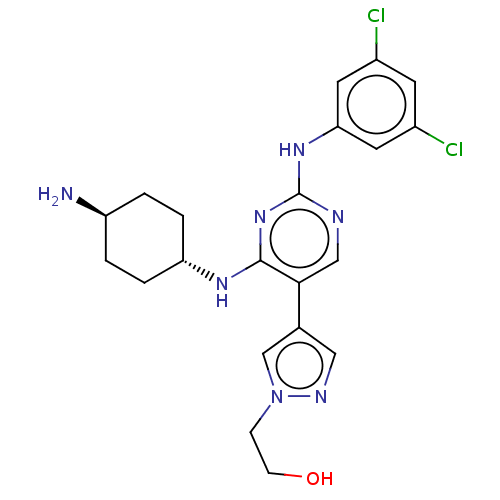
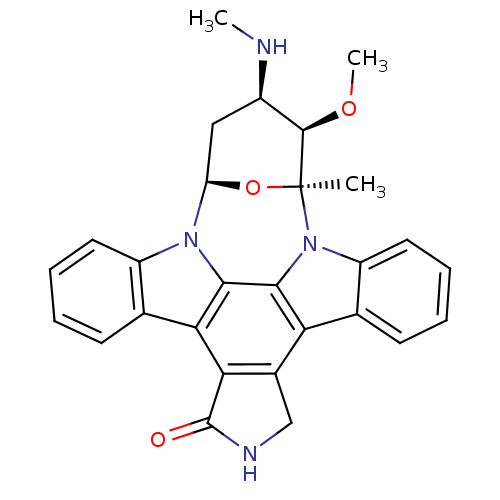
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0120nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0120nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0120nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0410nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0410nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0520nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0600nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.0970nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.100nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.130nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.130nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.130nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.140nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.140nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.160nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.160nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.160nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.170nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.170nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.170nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.190nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.200nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.230nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.270nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.270nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.280nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.300nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.320nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.340nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.340nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.370nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.420nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.480nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.580nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.600nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.700nMAssay Description:Inhibition of Sky (unknown origin) by ELISA kinase assay in presence of 60 uM ATPMore data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 0.900nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human RSE (451 to end residues) using KVEKIGEGTYGVVYK as substrate incubated for 40 mins in presence of [gamma33P]ATP by sc...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.20nMAssay Description:Inhibition of SKY (unknown origin)More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.20nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.30nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.40nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.5nMAssay Description:Inhibition of human TYRO3 using poly[Glu:Tyr] (4:1) as substrate by [gamma-33P]-ATP assayMore data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.60nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.60nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair
TargetTyrosine-protein kinase receptor TYRO3(Human)
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1.60nMAssay Description:For evaluation of inhibitory activities of the compounds according to the present invention on Tyro 3, Axl, and Mer, the following test was carried o...More data for this Ligand-Target Pair