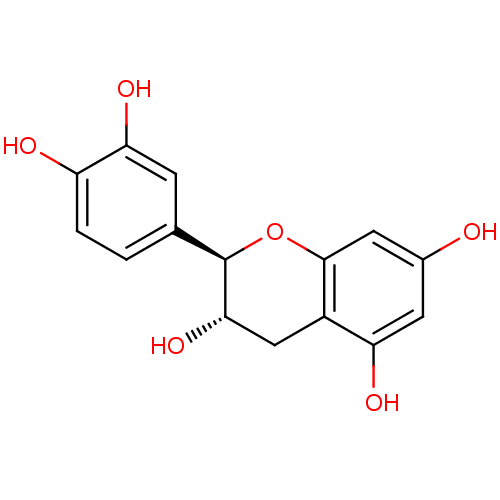
BDBM23416 α-CA inhibitor, 3::(+)-Catechin::(2R,3S)-2-(3,4-dihydroxyphenyl)-3,4-dihydro-2H-1-benzopyran-3,5,7-triol::CHEMBL311498::Catechin::D-Catechin
SMILES O[C@H]1Cc2c(O)cc(O)cc2O[C@@H]1c1ccc(O)c(O)c1
InChI Key InChIKey=PFTAWBLQPZVEMU-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 41 hits for monomerid = 23416
Found 41 hits for monomerid = 23416
Affinity DataIC50: 60nMAssay Description:Inhibition of mushroom tyrosinase monophenolase activityMore data for this Ligand-Target Pair
Affinity DataIC50: 70nMAssay Description:Inhibition of mushroom tyrosinase diphenolase activityMore data for this Ligand-Target Pair
Affinity DataKi: 450nMAssay Description:Inhibition of human carbonic anhydrase 7 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 600nMAssay Description:Noncompetitive inhibition of Leishmania amazonensis recombinant arginase expressed in Escherichia coli Rosetta (DE3) pLysS using L-arginine as substr...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+3nMAssay Description:Inhibition of Leishmania amazonensis recombinant arginase expressed in Escherichia coli Rosetta (DE3) pLysS using L-arginine as substrate incubated f...More data for this Ligand-Target Pair
Affinity DataKi: 1.84E+3nMAssay Description:Inhibition of human carbonic anhydrase 2 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 2.20E+3nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
Affinity DataKi: 2.42E+3nMAssay Description:Inhibition of human carbonic anhydrase 1 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 3.58E+3nMAssay Description:Inhibition of human carbonic anhydrase 3 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.02E+3nMAssay Description:Inhibition of human carbonic anhydrase 5b after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Sheep)
University of Pennsylvania School of Medicine
Curated by ChEMBL
University of Pennsylvania School of Medicine
Curated by ChEMBL
Affinity DataIC50: 4.10E+3nMAssay Description:Inhibition of peroxidase activity of COX1 in heep seminal vesicle by TMPD assayMore data for this Ligand-Target Pair
Affinity DataKi: 4.21E+3nMAssay Description:Inhibition of human carbonic anhydrase 5a after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.72E+3nMAssay Description:Inhibition of human carbonic anhydrase 12 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.90E+3nMAssay Description:Inhibition of human carbonic anhydrase 4 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.91E+3nMAssay Description:Inhibition of human carbonic anhydrase 6 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 5.03E+3nMAssay Description:Inhibition of human carbonic anhydrase 9 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 5.60E+3nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
Affinity DataKi: 6.80E+3nMpH: 7.4Assay Description:Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion over a ...More data for this Ligand-Target Pair
Affinity DataKi: 7.68E+3nMAssay Description:Inhibition of mouse carbonic anhydrase 15 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 9.71E+3nMpH: 7.4Assay Description:CA activity was assayed by following the change in absorbance at 348 nm of 4-NPA to 4-nitrophenylate ion over a period of 3 min at 25°C using a spect...More data for this Ligand-Target Pair
Affinity DataKi: 1.05E+4nMAssay Description:Inhibition of human carbonic anhydrase 13 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.16E+4nMAssay Description:Inhibition of human carbonic anhydrase 14 after 15 mins by stopped flow CO2 hydration methodMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Sheep)
University of Pennsylvania School of Medicine
Curated by ChEMBL
University of Pennsylvania School of Medicine
Curated by ChEMBL
Affinity DataIC50: 2.02E+4nMAssay Description:Inhibition of cyclooxygenase activity of COX1 in sheep seminal vesicle in presence of 1 mM phenol by cyclooxygenase assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.58E+4nMAssay Description:Agonist activity at human melatonin MT1 receptor stably expressing in human HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataEC50: 2.58E+4nMAssay Description:Agonist activity at human MT1R stably expressed in HEK293 cells by FlexStation3 Bench-top MultiMode Microplate ReaderMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of aldose reductase in rat lens homogenateMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Sheep)
University of Pennsylvania School of Medicine
Curated by ChEMBL
University of Pennsylvania School of Medicine
Curated by ChEMBL
Affinity DataKd: 4.07E+4nMAssay Description:Binding affinity to COX1 in sheep seminal vesicleMore data for this Ligand-Target Pair
Affinity DataEC50: 4.73E+4nMAssay Description:Agonist activity at human MT2R stably expressed in HEK293 cells by FlexStation3 Bench-top MultiMode Microplate ReaderMore data for this Ligand-Target Pair
Affinity DataIC50: 4.73E+4nMAssay Description:Agonist activity at human melatonin MT2 receptor stably expressing in human HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKd: 4.92E+4nMAssay Description:Inhibition of human thrombin assessed as equilibrium dissociation constant at 50 to 1000 uM by BIAcore analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+4nMAssay Description:Inhibition of bovine seminal vesicle microsomal COX1-mediated prostaglandin productionMore data for this Ligand-Target Pair
TargetDihydroorotate dehydrogenase (fumarate)(Leishmania major)
University of S£O Paulo
Curated by ChEMBL
University of S£O Paulo
Curated by ChEMBL
Affinity DataIC50: 9.83E+4nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
TargetADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1(Human)
University of Strasburg
Curated by ChEMBL
University of Strasburg
Curated by ChEMBL
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human CD38 using 20 uM 1, N6-etheno NAD+ as substrate by continuous fluorimetric methodMore data for this Ligand-Target Pair
TargetDihydroorotate dehydrogenase (fumarate)(Leishmania major)
University of S£O Paulo
Curated by ChEMBL
University of S£O Paulo
Curated by ChEMBL
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of recombinant oligo-histidine-tagged Leishmania major DHODH expressed in Escherichia coli BL21(DE3) cells using DHO as substrate measured...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of xanthine oxidase assessed as decrease in uric acid production by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.25E+5nMAssay Description:Inhibition of human thrombin amidolytic activity using D-Phe-Pip-Arg-pNA as substrate preincubated for 10 mins followed by substrate addition measure...More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+5nMAssay Description:Displacement of ANS from DAPK1 catalytic domain (1 to 285) (unknown origin) after 30 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+5nMAssay Description:Inhibition of rat intestinal sucrase using p-nitrophenyl-alpha-d-glucopyranoside as substrate incubated for 10 mins prior to substrate addition measu...More data for this Ligand-Target Pair
Affinity DataIC50: 3.41E+5nMAssay Description:Inhibition of porcine pancreatic lipase using pNPB as substrate measured after 30 mins using plasma treated sampleMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+5nMAssay Description:Inhibition of porcine pancreatic lipase using pNPB as substrate measured after 30 minsMore data for this Ligand-Target Pair
Affinity DatapH: 6.0 T: 2°CAssay Description:The assay was carried out at room temperature for 10 min with salivary alpha-amylase, starch, and test compounds. The reducing sugar was determined b...More data for this Ligand-Target Pair