Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cysteine protease
Ligand
BDBM50268553
Substrate
n/a
Meas. Tech.
ChEMBL_591545 (CHEMBL1062951)
IC50
700±n/a nM
Citation
 Mallari, JP; Shelat, AA; Kosinski, A; Caffrey, CR; Connelly, M; Zhu, F; McKerrow, JH; Guy, RK Structure-guided development of selective TbcatB inhibitors. J Med Chem 52:6489-93 (2009) [PubMed] Article
Mallari, JP; Shelat, AA; Kosinski, A; Caffrey, CR; Connelly, M; Zhu, F; McKerrow, JH; Guy, RK Structure-guided development of selective TbcatB inhibitors. J Med Chem 52:6489-93 (2009) [PubMed] Article More Info.:
Target
Name:
Cysteine protease
Synonyms:
Rhodesain
Type:
PROTEIN
Mol. Mass.:
48425.78
Organism:
Trypanosoma brucei rhodesiense
Description:
ChEMBL_619861
Residue:
450
Sequence:
MPRTEMVRFVRLPVVLLAMAACLASVALGSLHVEESLEMRFAAFKKKYGKVYKDAKEEAFRFRAFEENMEQAKIQAAANPYATFGVTPFSDMTREEFRARYRNGASYFAAAQKRLRKTVNVTTGRAPAAVDWREKGAVTPVKDQGQCGSCWAFSTIGNIEGQWQVAGNPLVSLSEQMLVSCDTIDFGCGGGLMDNAFNWIVNSNGGNVFTEASYPYVSGNGEQPQCQMNGHEIGAAITDHVDLPQDEDAIAAYLAENGPLAIAVDATSFMDYNGGILTSCTSEQLDHGVLLVGYNDSSNPPYWIIKNSWSNMWGEDGYIRIEKGTNQCLMNQAVSSAVVGGPTPPPPPPPPPSATFTQDFCEGKGCTKGCSHATFPTGECVQTTGVGSVIATCGASNLTQIIYPLSRSCSGLSVPITVPLDKCIPILIGSVEYHCSTNPPTKAARLVPHQ
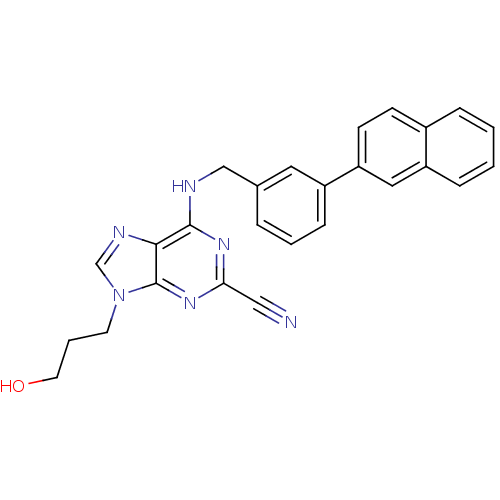
Inhibitor
Name:
BDBM50268553
Synonyms:
9-(3-hydroxypropyl)-6-(3-(naphthalen-2-yl)benzylamino)-9H-purine-2-carbonitrile | 9-(3-hydroxypropyl)-6-(naphthalen-2-ylmethylamino)-9H-purine-2-carbonitrile | CHEMBL497234
Type:
Small organic molecule
Emp. Form.:
C26H22N6O
Mol. Mass.:
434.4925
SMILES:
OCCCn1cnc2c(NCc3cccc(c3)-c3ccc4ccccc4c3)nc(nc12)C#N