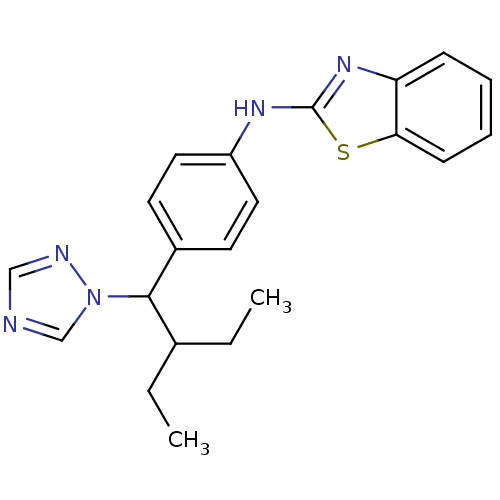
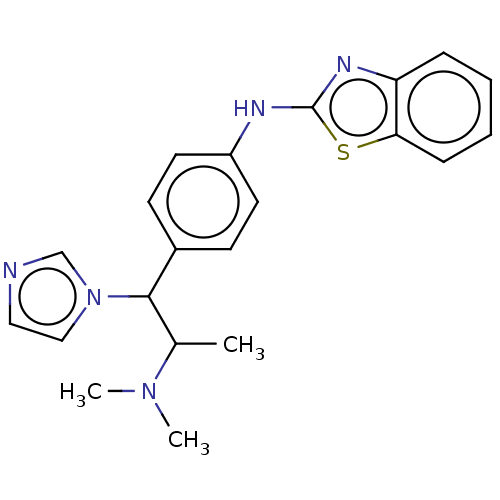
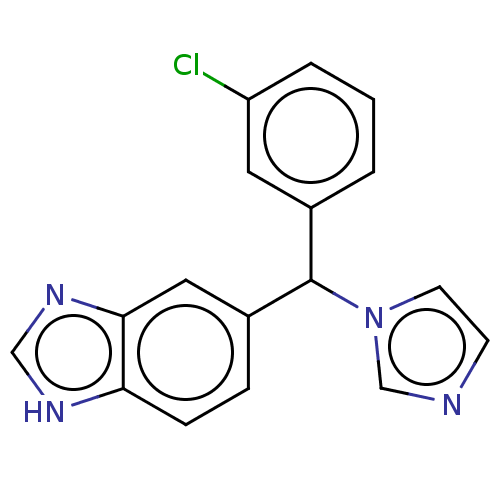
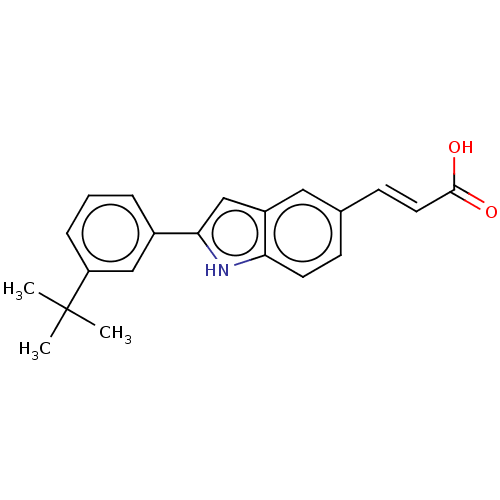
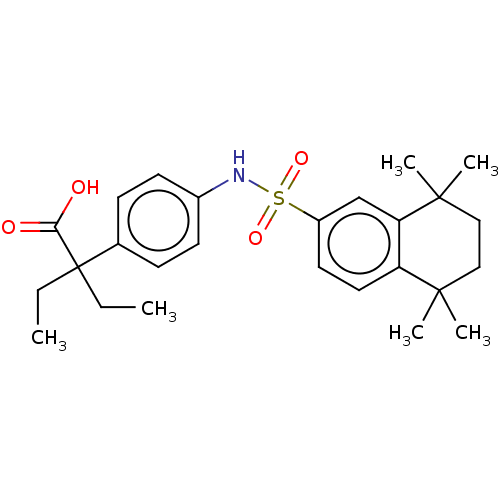
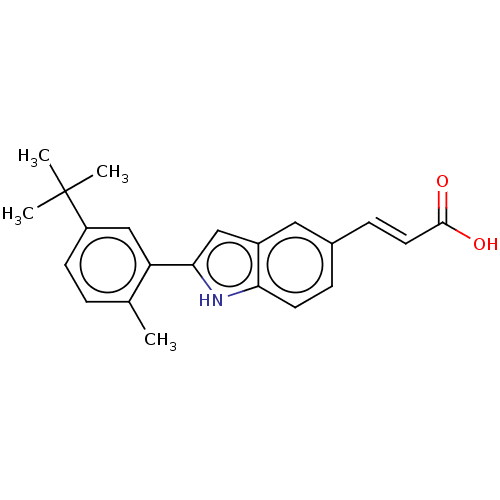
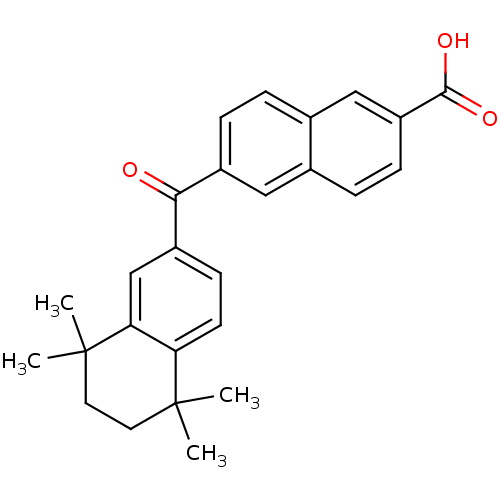
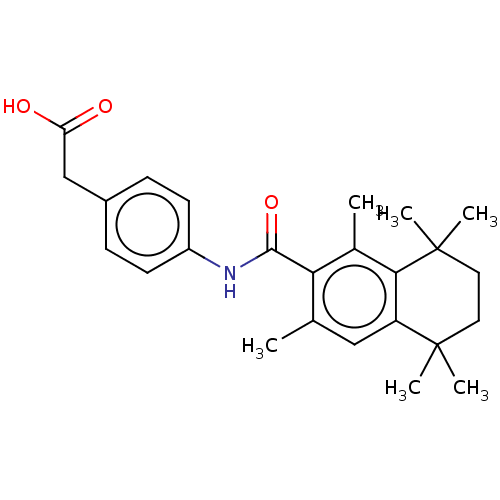
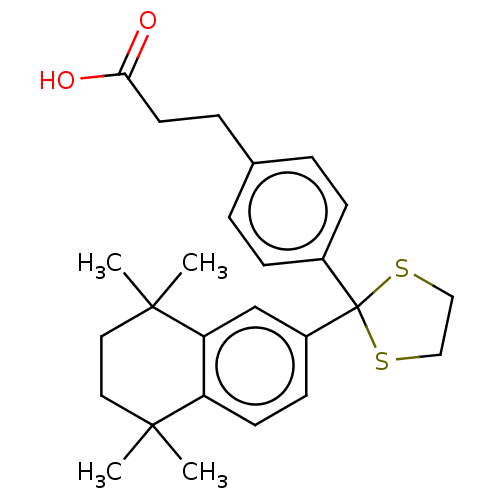
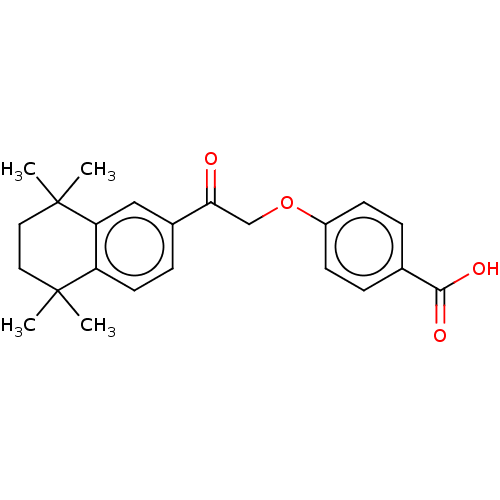
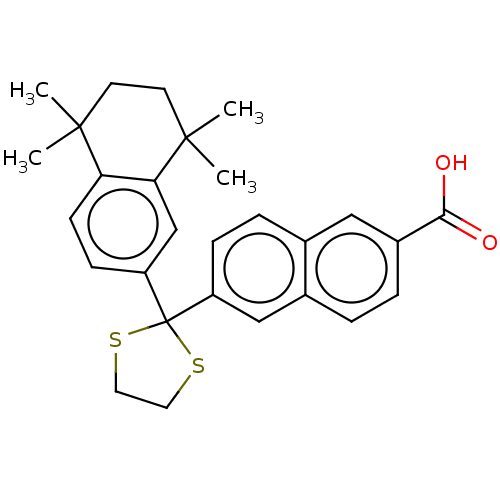
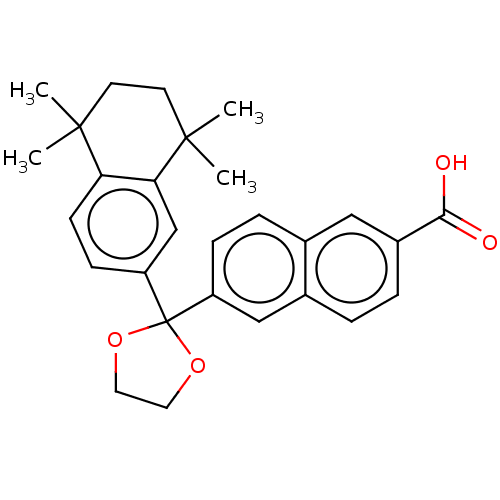
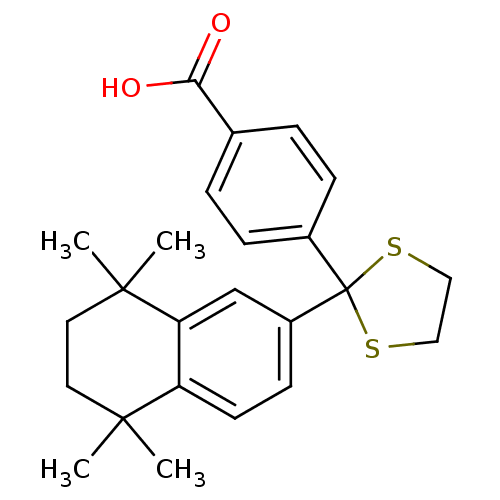
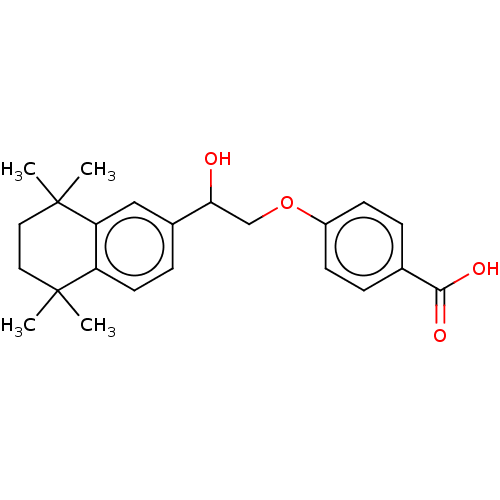
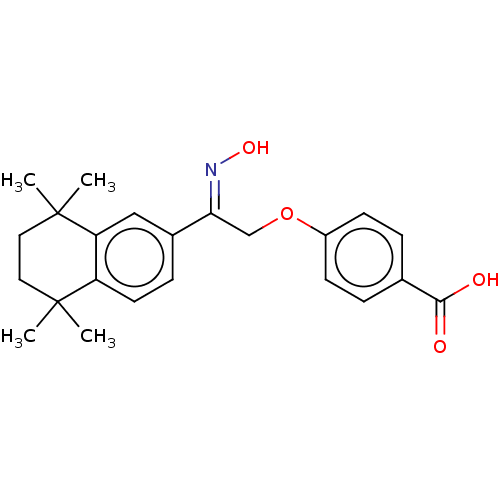
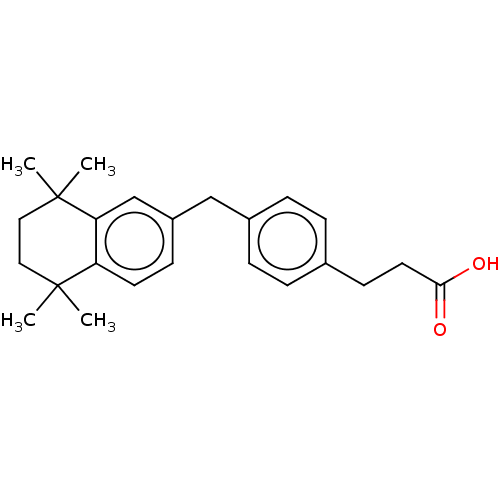
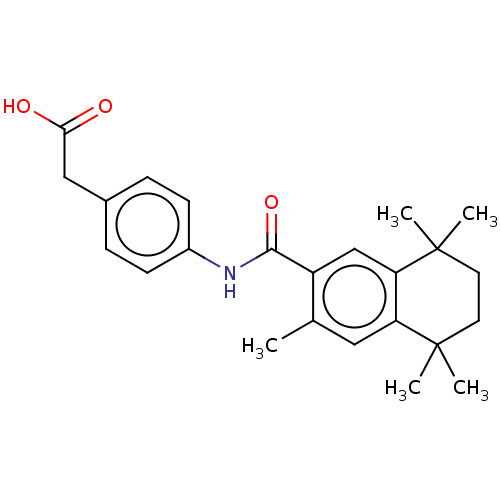
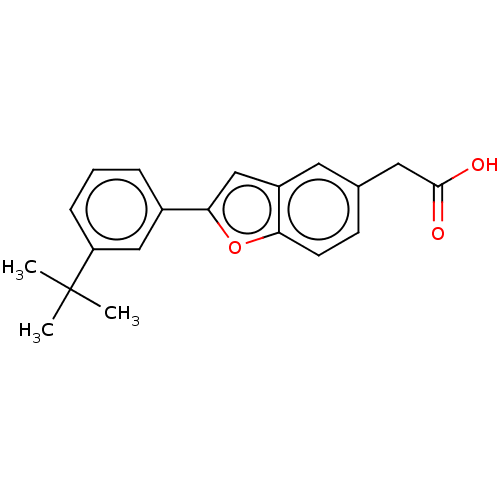
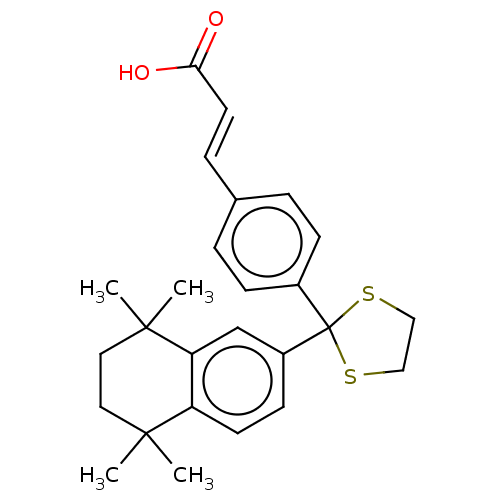
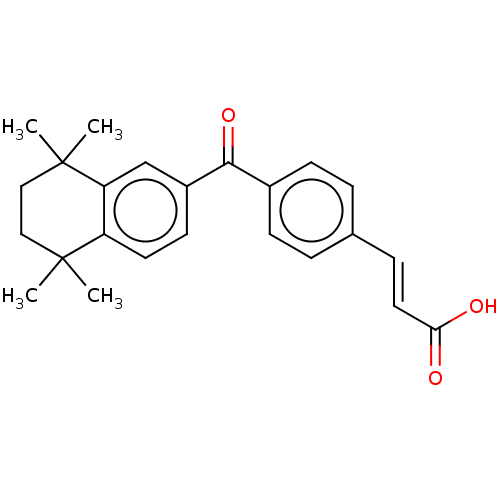
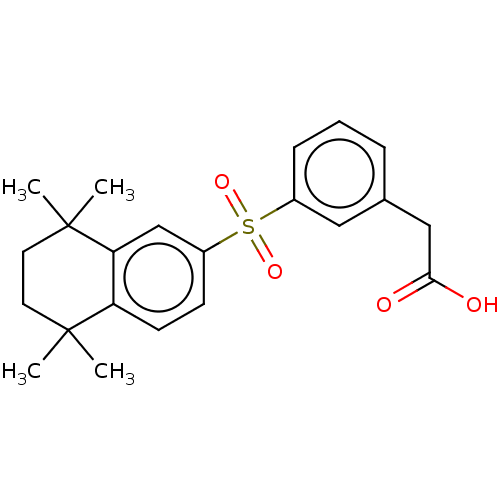
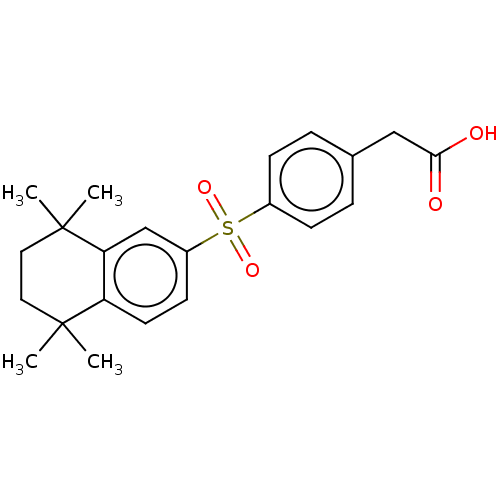
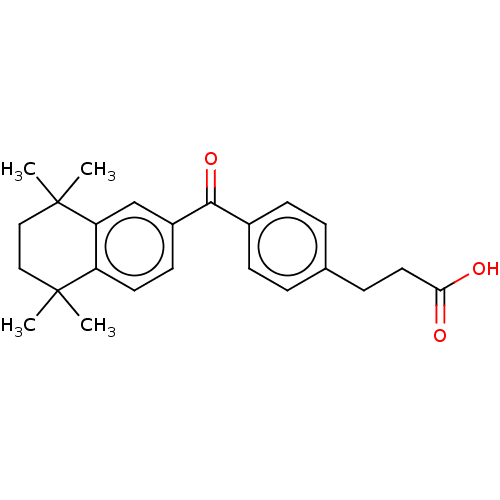
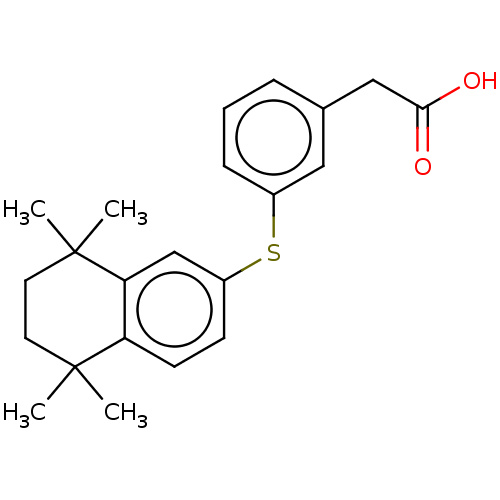
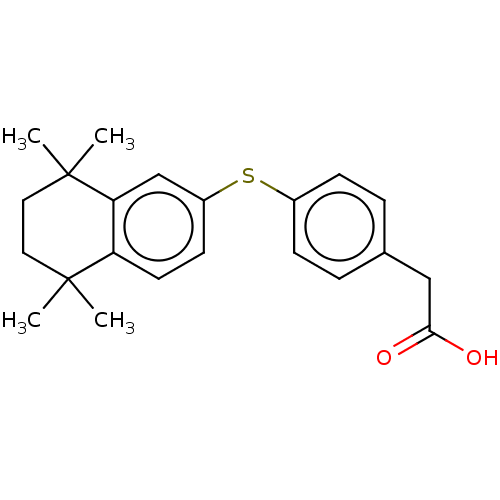
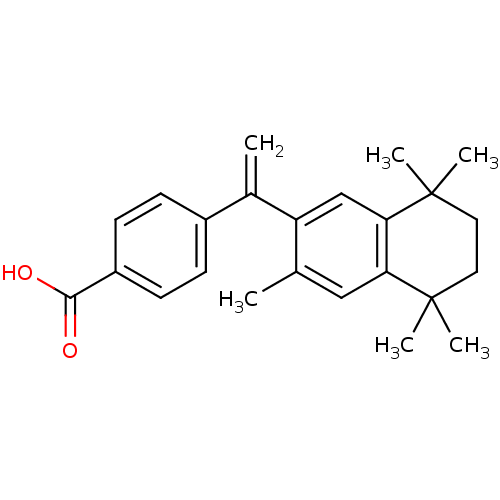
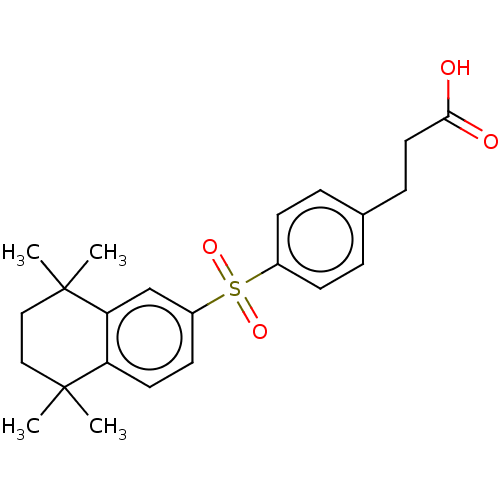
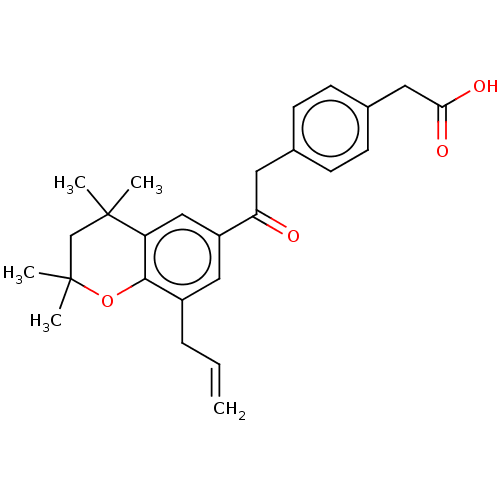
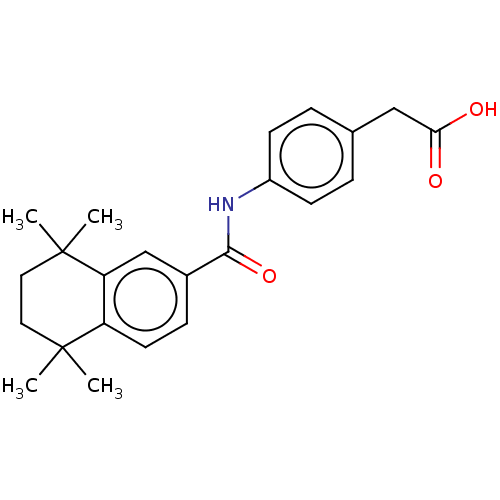
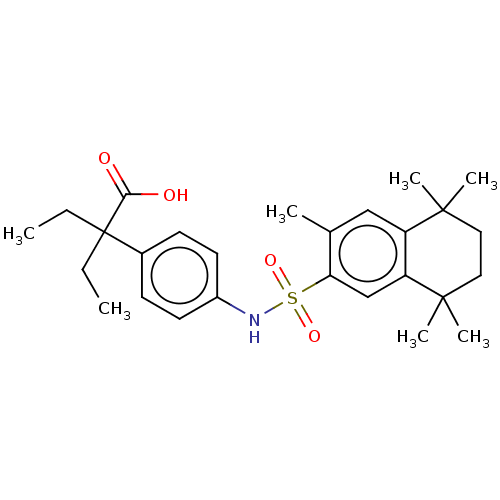
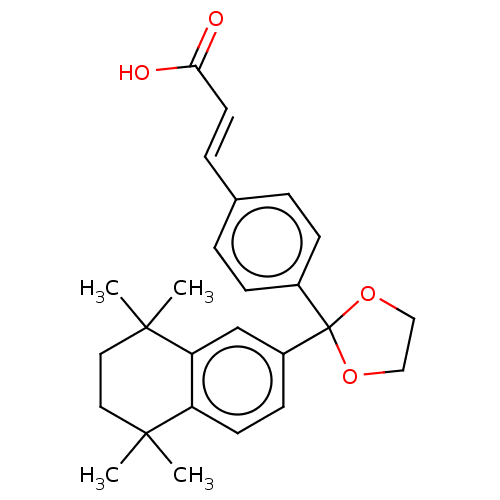
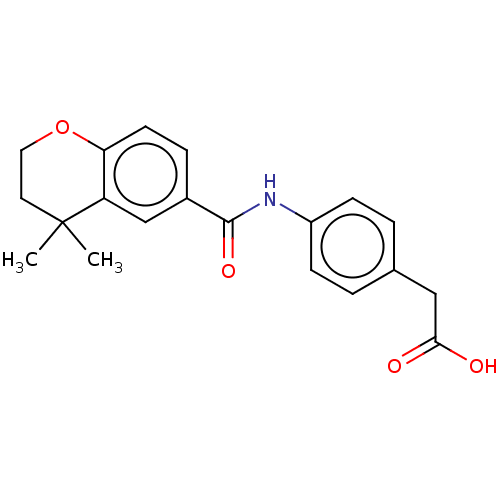
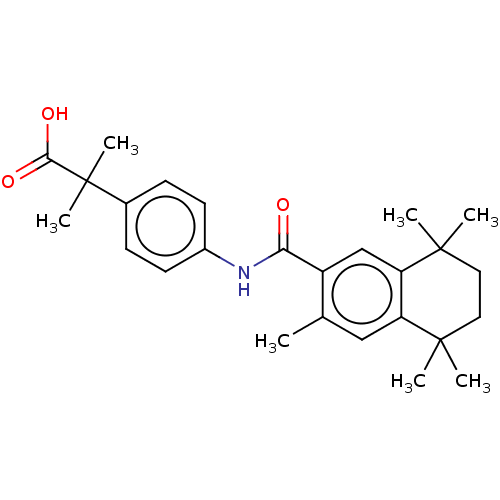
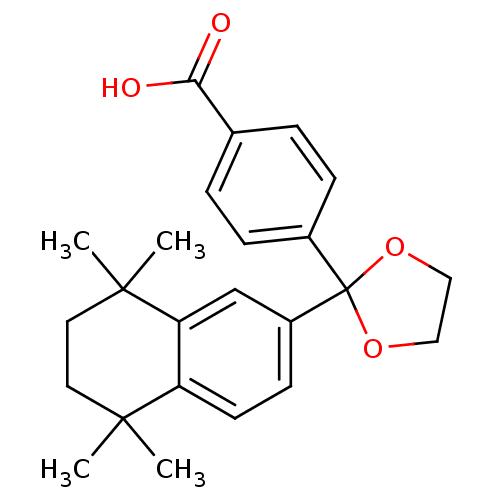
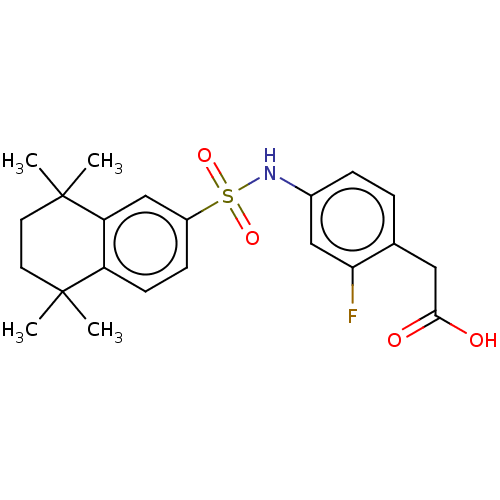
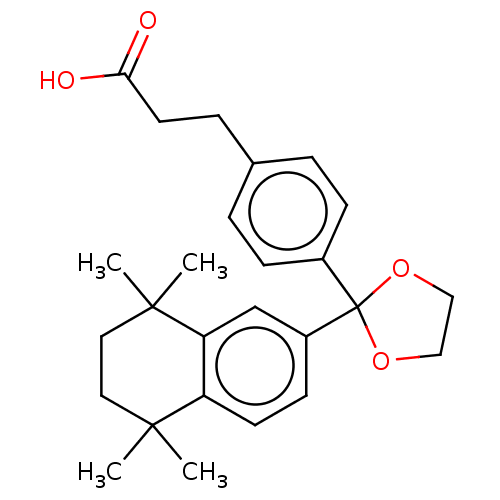
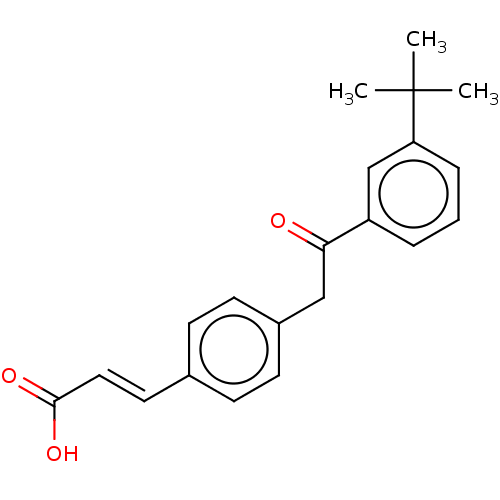
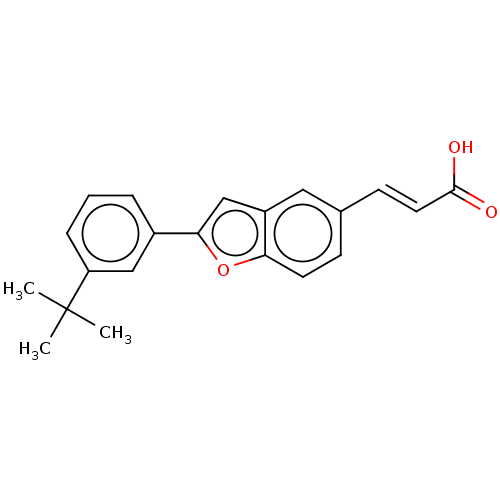
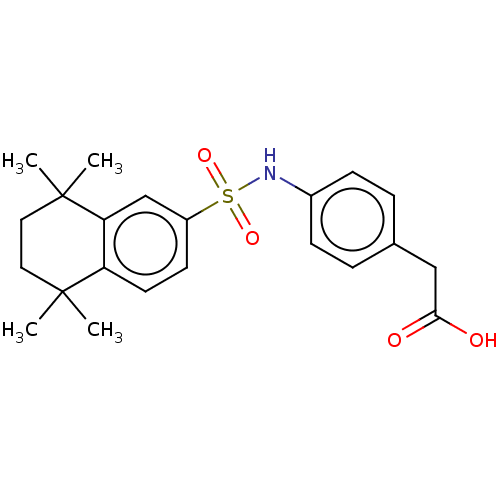
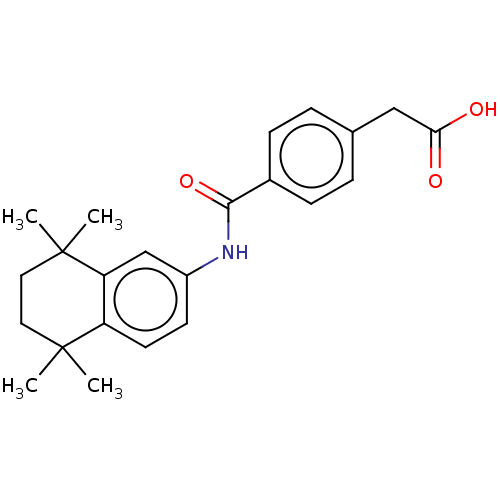
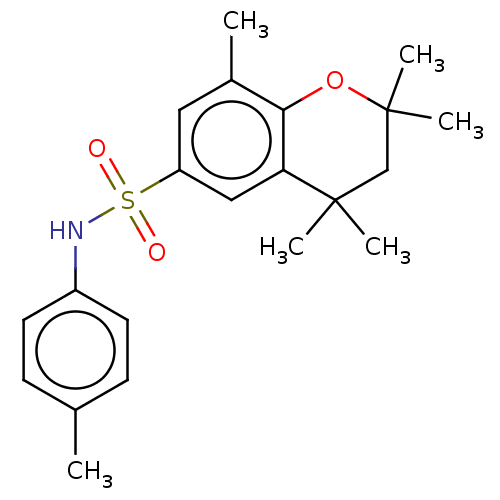
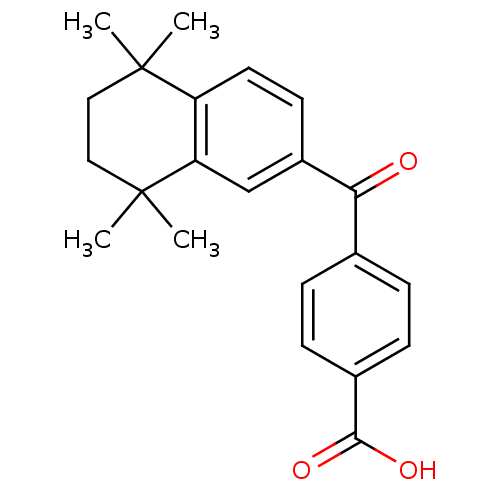
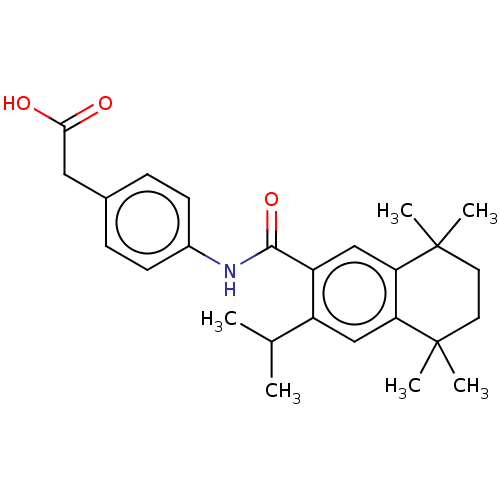
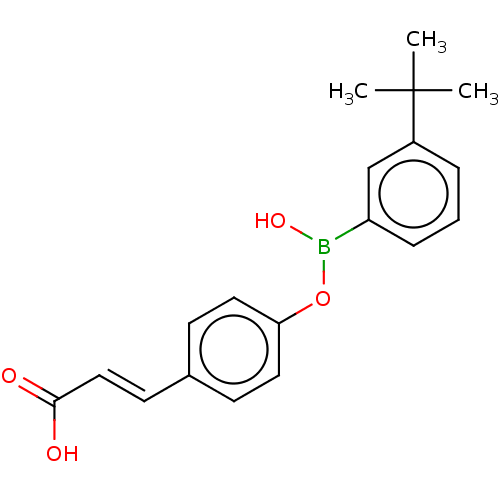
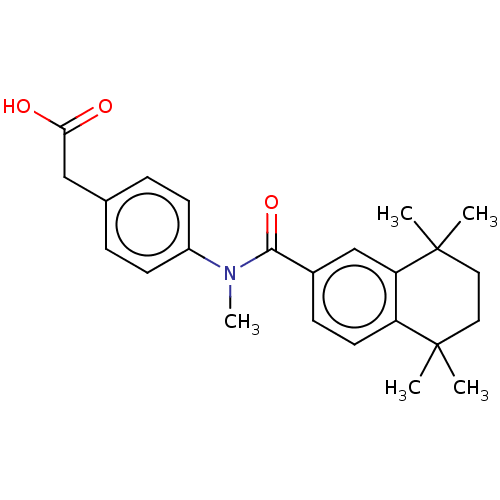
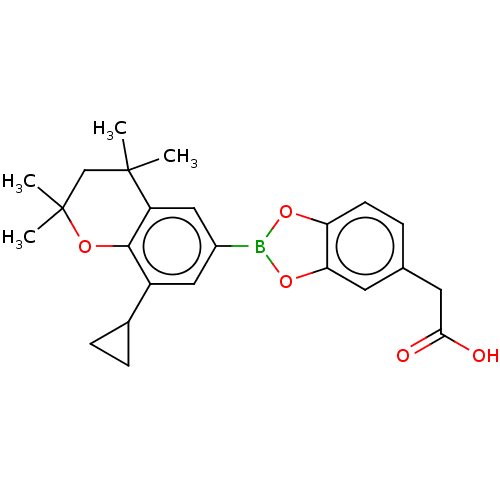
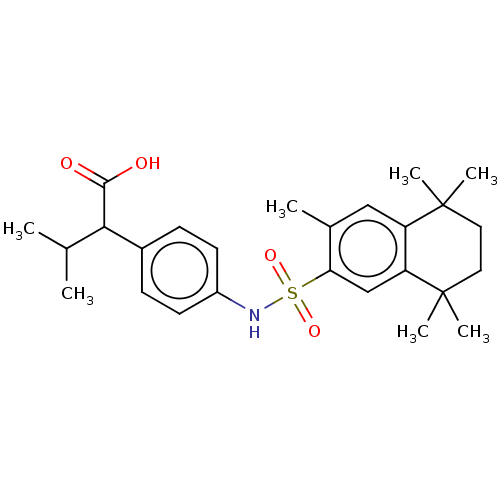
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 0.460nMAssay Description:Eighteen compounds were tested as potential inhibitors of CYP26A1 and CYP26B1. The formation of 9-cis-4-OH-RA metabolite was monitored and the percen...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 0.460nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 3.10nMAssay Description:Eighteen compounds were tested as potential inhibitors of CYP26A1 and CYP26B1. The formation of 9-cis-4-OH-RA metabolite was monitored and the percen...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 18nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 18nMAssay Description:Eighteen compounds were tested as potential inhibitors of CYP26A1 and CYP26B1. The formation of 9-cis-4-OH-RA metabolite was monitored and the percen...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 140nMAssay Description:Eighteen compounds were tested as potential inhibitors of CYP26A1 and CYP26B1. The formation of 9-cis-4-OH-RA metabolite was monitored and the percen...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 200nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 290nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 500nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 520nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 550nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 680nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 930nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.03E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.03E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.40E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.40E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.70E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 2.06E+3nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 2.30E+3nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 4.40E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.90E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 6.20E+3nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 8.80E+3nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 8.81E+3nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 9.07E+3nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.15E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.19E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.26E+4nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.42E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.45E+4nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.79E+4nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.83E+4nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.89E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.92E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 2.18E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of microsomal fraction of human CYP26B1 expressed in Sf9 cells using 9-cis-RA as substrate preincubated for 5 mins followed by NADPH addit...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 4.23E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 5.49E+4nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 6.54E+4nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 8.81E+4nMAssay Description:CYP26 inhibitory activities of various compounds.More data for this Ligand-Target Pair
TargetCytochrome P450 26B1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 1.00E+5nMAssay Description:This retinoid activity assay measures the ability of test compounds to induce expression of a transiently transfected RA sensitive reporter construct...More data for this Ligand-Target Pair