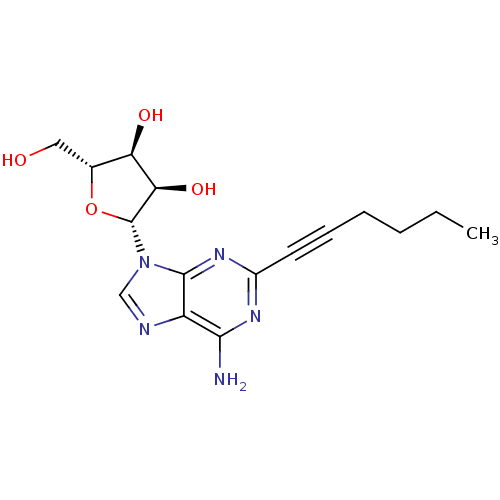
BDBM50119168 (2R,3R,4S,5R)-2-(6-Amino-2-hex-1-ynyl-purin-9-yl)-5-hydroxymethyl-tetrahydro-furan-3,4-diol::(2R,3R,4S,5R)-2-(6-amino-2-(hex-1-ynyl)-9H-purin-9-yl)-5-(hydroxymethyl)-tetrahydrofuran-3,4-diol::(2R,3R,4S,5R)-2-(6-amino-2-(hex-1-ynyl)-9H-purin-9-yl)-5-(hydroxymethyl)tetrahydrofuran-3,4-diol::2-(l-hexyn-l-yl)adenosine (7)::CHEMBL319387
SMILES CCCCC#Cc1nc(N)c2ncn([C@@H]3O[C@H](CO)[C@@H](O)[C@H]3O)c2n1
InChI Key InChIKey=AHJRSKVOELIMOX-RVXWVPLUSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50119168
Found 4 hits for monomerid = 50119168
Affinity DataEC50: >1nMAssay Description:Adenosine A1 receptor mediated negative chronotropic activity in spontaneously beating rat atriaMore data for this Ligand-Target Pair
Affinity DataEC50: 128nMAssay Description:Maximal NECA stimulation of adenylate cyclase via adenosine A2 receptor in human platelet membranesMore data for this Ligand-Target Pair
TargetAdenosine receptor A2b(Homo sapiens (Human))
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
National Institute Of Diabetes And Digestive And Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 1.00E+4nMAssay Description:Agonist activity at human adenosine A2B receptor expressed in CHO cells assessed as stimulation of adenylate cyclaseMore data for this Ligand-Target Pair
Affinity DataEC50: 79nMAssay Description:Vasorelaxation as Adenosine A2A receptor activity in rat aortaMore data for this Ligand-Target Pair