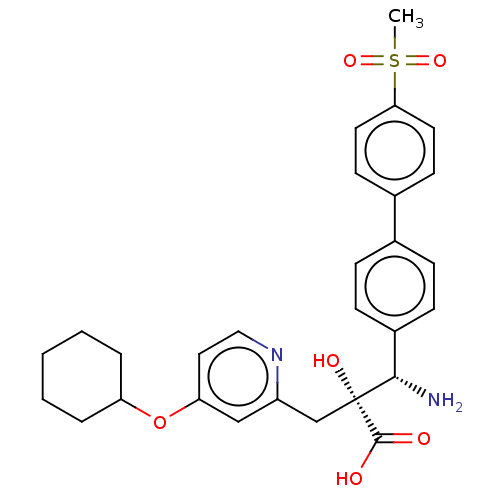
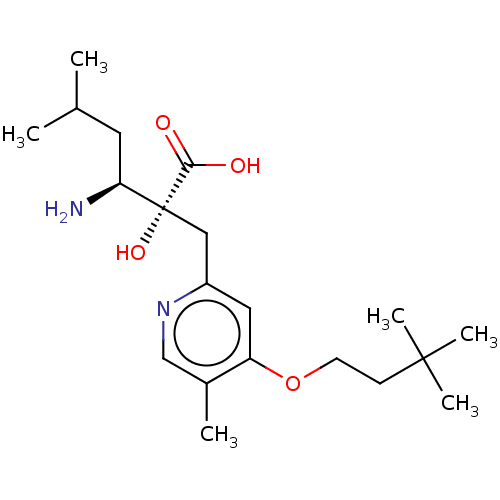
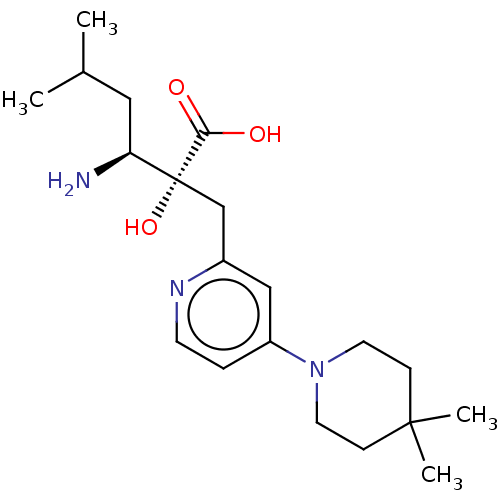
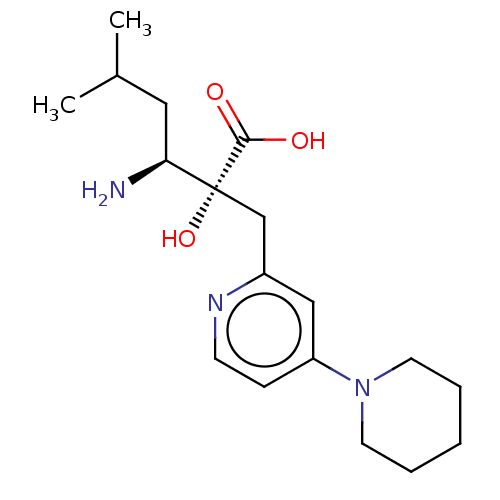
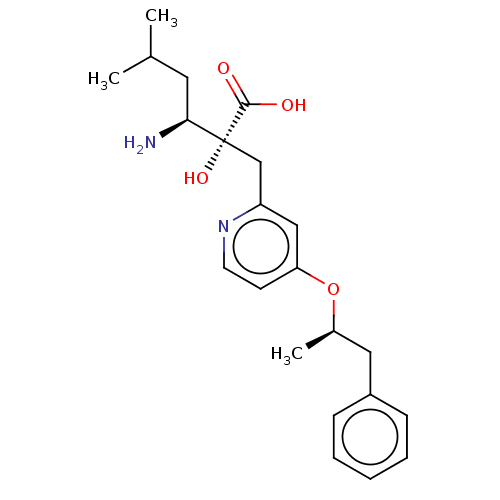
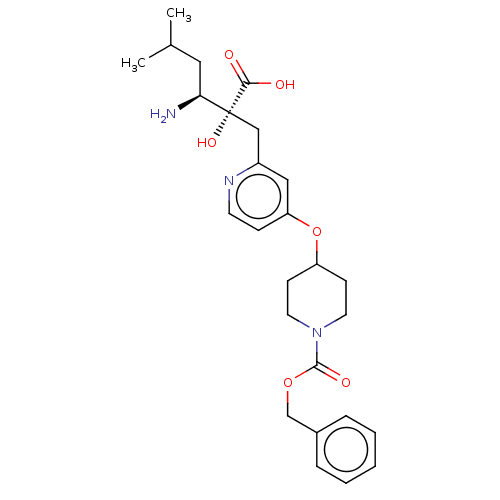
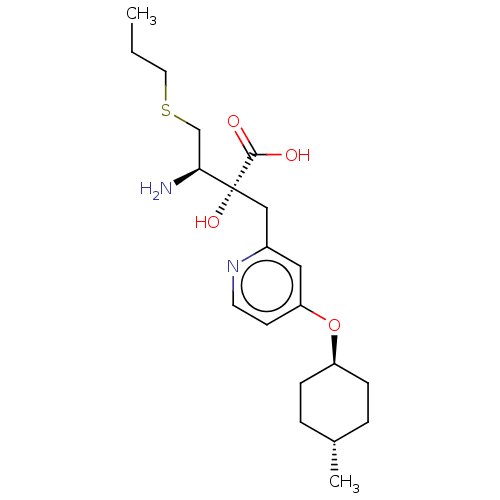
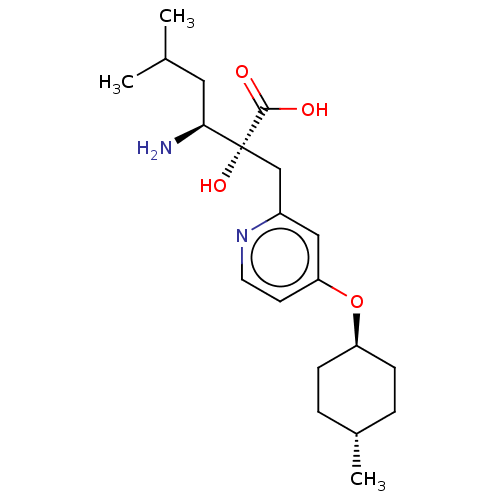
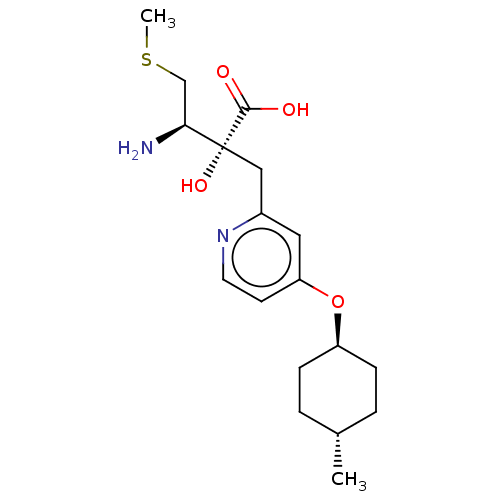
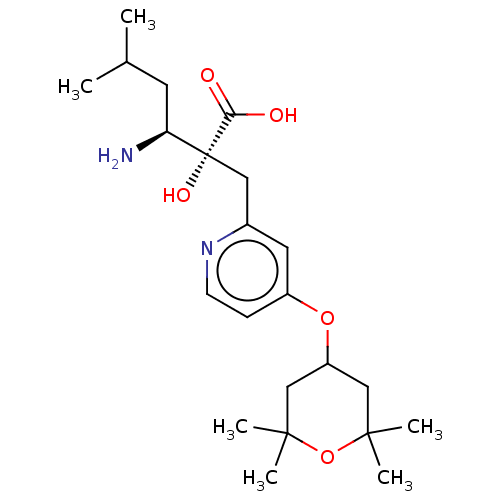
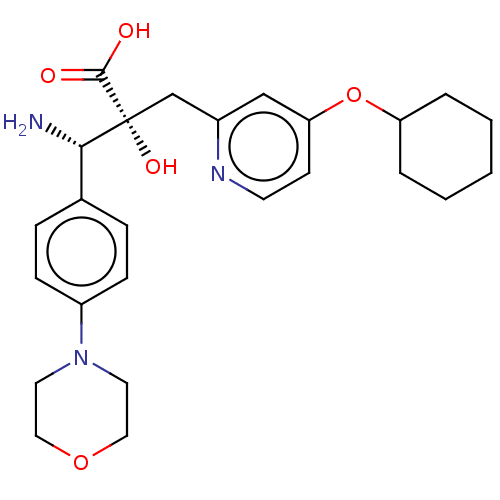
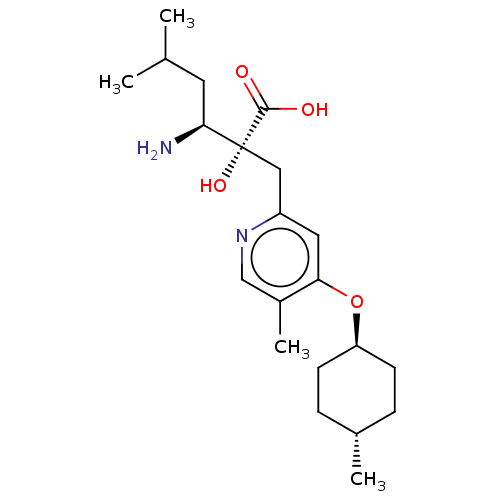
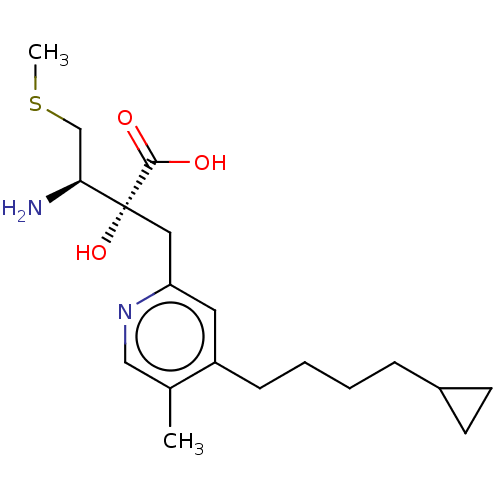
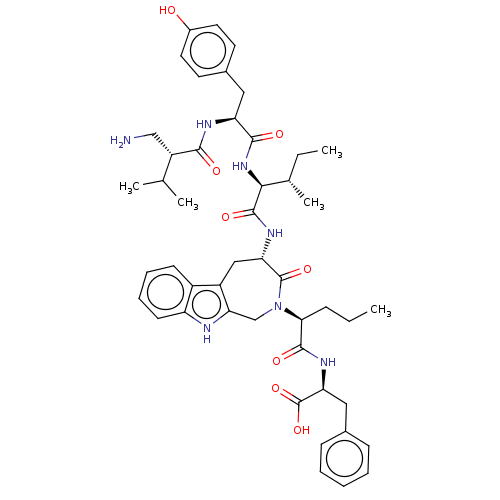
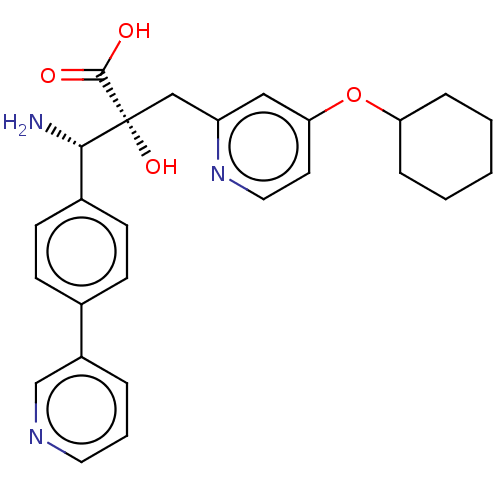
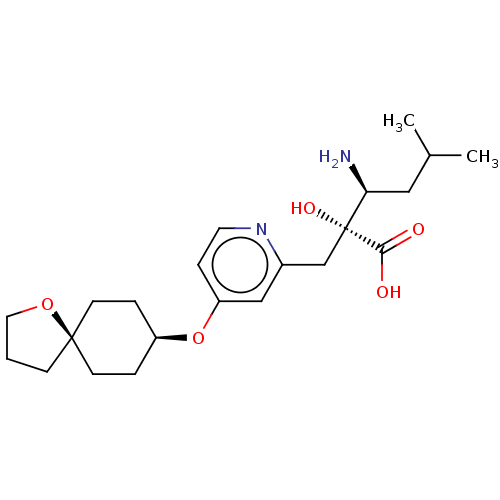
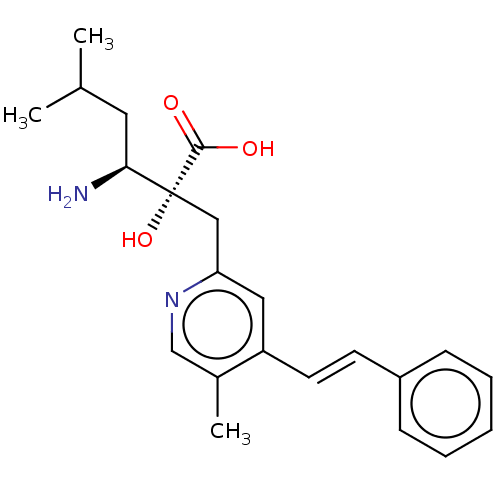
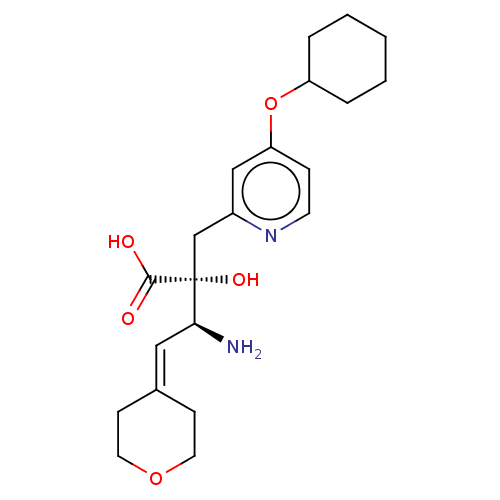
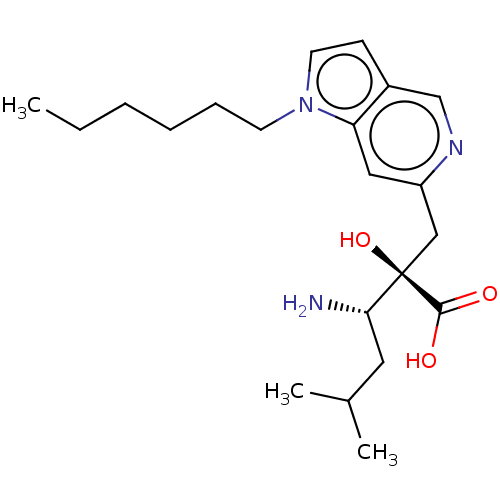
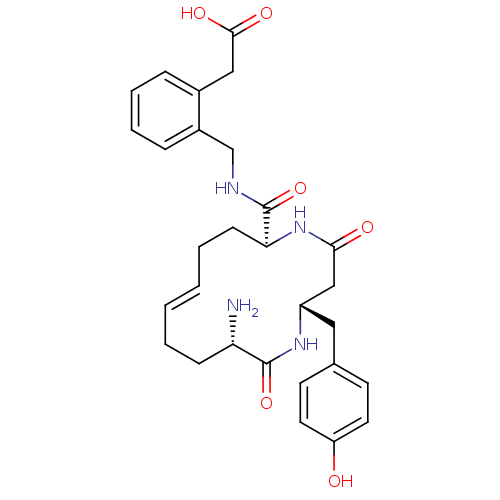
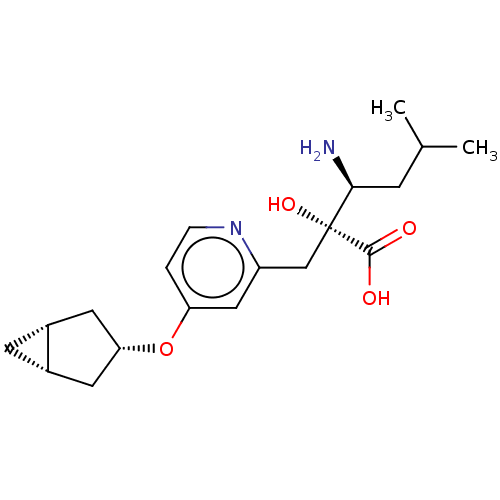
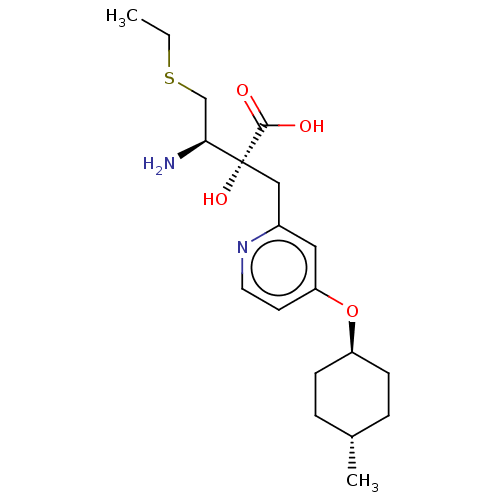
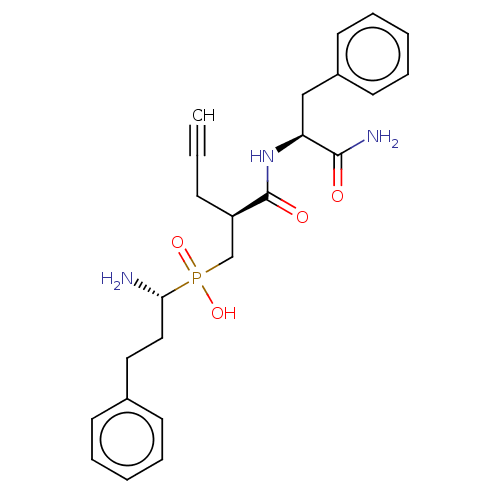
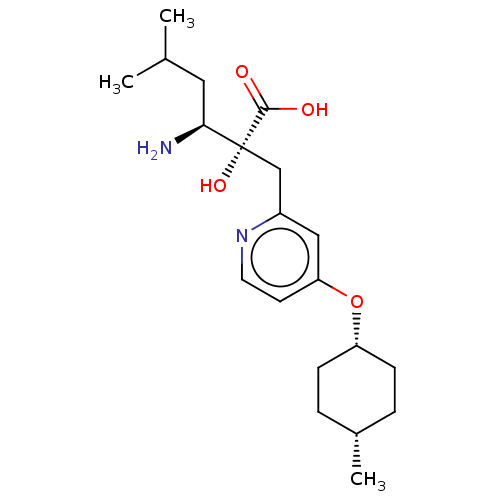
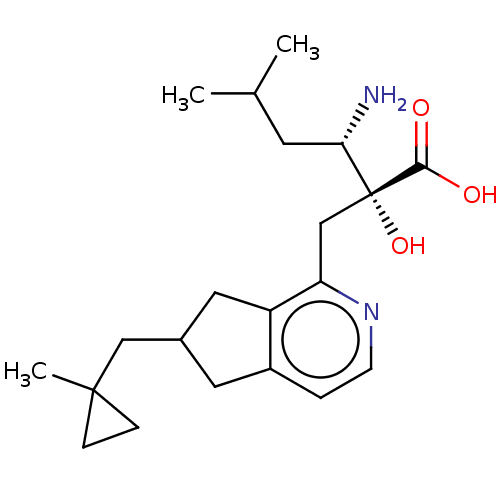
Affinity DataIC50: 0.890nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 0.890nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
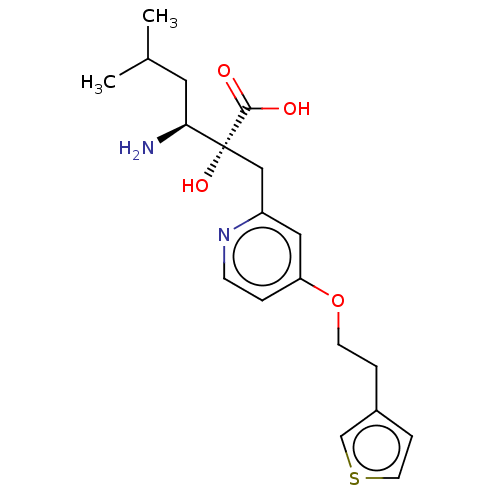
Affinity DataIC50: 0.940nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 0.940nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
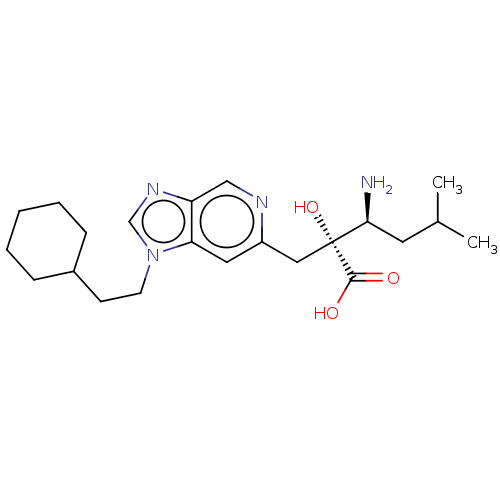
Affinity DataIC50: 0.950nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 0.950nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
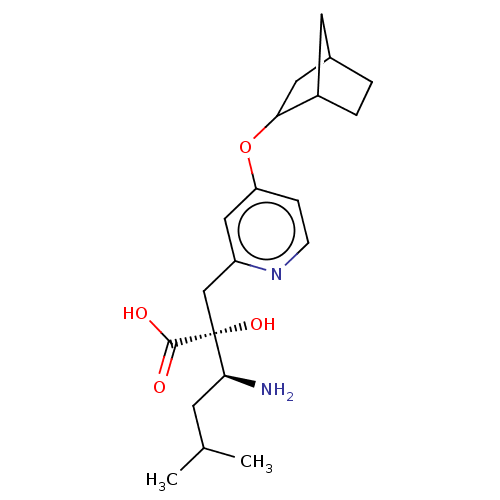
Affinity DataIC50: 1nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:The inventors have assumed that inhibition of nocturnal activity of placental leucine aminopeptidase (P-LAP), i.e. aminopeptidase that cleaves AVP, w...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:Binding affinity to insulin regulated aminopeptidase (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:The inventors have assumed that inhibition of nocturnal activity of placental leucine aminopeptidase (P-LAP), i.e. aminopeptidase that cleaves AVP, w...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Inhibition of catalytic activity of recombinant human IRAP transfected in human HEK-293 cells assessed as cleavage of substrate L-leucine-p-nitroanil...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of IRAP (unknown origin) expressed in HEK 293S GnTI(-) cells by in vitro fluorimetric assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:HEK293 cells forced to transiently express hP-LAP (J Biol Chem 1996; 271: 56-61) were prepared by lipofection, homogenized, and then subjected to ult...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:hP-LAP: HEK293 cells forced to transiently express hP-LAP were prepared by lipofection, homogenized, and then subjected to ultracentrifugation at 100...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:The inventors have assumed that inhibition of nocturnal activity of placental leucine aminopeptidase (P-LAP), i.e. aminopeptidase that cleaves AVP, w...More data for this Ligand-Target Pair