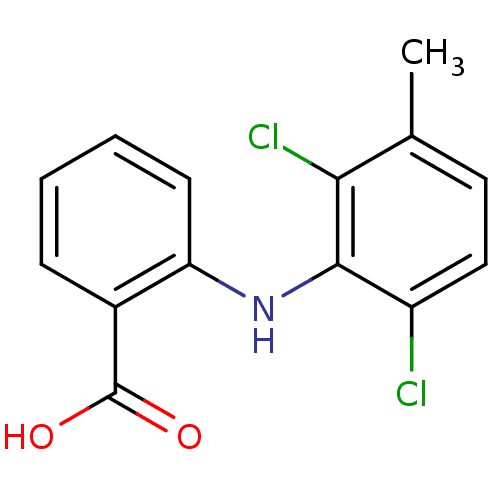
BDBM22971 2-[(2,6-dichloro-3-methylphenyl)amino]benzoic acid::CHEMBL509::Meclofenamate::Meclofenamic acid::US11337935, Compound Meclofenamic-acid::US20240002326, Compound Meclofenamic acid
SMILES Cc1ccc(Cl)c(Nc2ccccc2C(O)=O)c1Cl
InChI Key InChIKey=SBDNJUWAMKYJOX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 37 hits for monomerid = 22971
Found 37 hits for monomerid = 22971
TargetProstaglandin G/H synthase 2(Mus musculus (Mouse))
Vanderbilt University School Of Medicine
Curated by ChEMBL
Vanderbilt University School Of Medicine
Curated by ChEMBL
Affinity DataIC50: 400nMAssay Description:Inhibition of the murine Arg120Ala mutant type COX-2More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition 5-lipoxygenase mediated LTB4 formation in rat basophilic leukemia (RBL-1) cellsMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 2(Homo sapiens (Human))
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
Affinity DataIC50: 40nMAssay Description:Inhibition of the human Prostaglandin G/H synthase 2 was determined by thin-layer chromatography assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:Binding affinity to TTR in human plasma assessed as protein stabilization preincubated for 1 hr followed by urea-mediated denaturation by Western blo...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Compound was evaluated for its inhibitory activity against 5-LO(5-lipoxygenase)More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Compound was evaluated in an intact RBL-1 cell line for inhibition of 5-lipoxygenaseMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of 5-lipoxygenase measured by the reduction of leukotriene B4 (LTB4) in intact basophilic rat leukemia cellsMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C4(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human recombinant N-terminal His6-tagged AKR1C4 expressed in Escherichia coli BL21(DE3) cells using 8-Acetyl-2,3,5,6-tetrahydro-1H,4H-1...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 8.74E+3nMAssay Description:Inhibition of human recombinant N-terminal His6-tagged AKR1C2 expressed in Escherichia coli BL21(DE3) cells using 8-Acetyl-2,3,5,6-tetrahydro-1H,4H-1...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 3.16E+3nMAssay Description:Inhibition of human recombinant N-terminal His6-tagged AKR1C1 expressed in Escherichia coli BL21(DE3) cells using 8-Acetyl-2,3,5,6-tetrahydro-1H,4H-1...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C3(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 540nMAssay Description:Inhibition of human recombinant N-terminal His6-tagged AKR1C3 expressed in Escherichia coli BL21(DE3) cells using 8-Acetyl-2,3,5,6-tetrahydro-1H,4H-1...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Compound was evaluated in an intact RBL-1 cell line for inhibition of 5-CyclooxygenaseMore data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C4(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of AKR1C4 (unknown origin)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 2(Homo sapiens (Human))
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
Affinity DataIC50: 700nMAssay Description:Inhibition of COX2 (unknown origin)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Homo sapiens (Human))
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
Affinity DataIC50: 220nMAssay Description:Inhibition of COX1 (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C3(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 540nMAssay Description:Inhibition of AKR1C3 (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C2(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 8.74E+3nMAssay Description:Inhibition of AKR1C2 (unknown origin)More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 3.16E+3nMAssay Description:Inhibition of AKR1C1 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Inhibition of Prostaglandin G/H synthase mediated PGF2-alpha formation in rat basophilic leukemia (RBL-1) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Binding affinity at 5-hydroxytryptamine 3 receptor in rat posterior cortex by [3H]-BRL 43694 displacement.More data for this Ligand-Target Pair
Affinity DataIC50: 2.72E+4nMAssay Description:Inhibition of human BSEP expressed in baculovirus transfected fall armyworm Sf21 cell membranes vesicles assessed as reduction in ATP-dependent [3H]-...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C1(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 740nMAssay Description:Inhibition of recombinant human AKR1C1 expressed in Escherichia coli BL21 cells in presence of 9,10-phenanthrenequinone and NADPH by fluorescence ass...More data for this Ligand-Target Pair
TargetAldo-keto reductase family 1 member C3(Homo sapiens (Human))
University Of Auckland
Curated by ChEMBL
University Of Auckland
Curated by ChEMBL
Affinity DataIC50: 512nMAssay Description:Inhibition of recombinant human AKR1C3 expressed in Escherichia coli BL21 cells in presence of 9,10-phenanthrenequinone and NADPH by fluorescence ass...More data for this Ligand-Target Pair
Affinity DataIC50: 504nMAssay Description:Table 3: The FP assay was then adapted for HTS and used to screen 120,000 small molecule library for compounds that displaced probe 5 from the T4 bin...More data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+5nMAssay Description:Different concentration ranges were tested in the FP-based DNA binding competition assay (0.2-200 uM, 10-500 uM, 10 uM-3 mM), with DMSO concentration...More data for this Ligand-Target Pair
Affinity DataIC50: 7.94E+3nMAssay Description:Inhibition of FTO (unknown origin) demethylation activity using m6A7-Broccoli RNA as substrate incubated for 3 mins under shaking condition and measu...More data for this Ligand-Target Pair
Affinity DataIC50: 8.00E+3nMAssay Description:Inhibition of N-terminal 31 residues truncated His-tagged human FTO demethylase activity expressed in Escherichia coli BL21(DE3) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human ALKBH2 demethylase activity by restriction endonuclease digestion assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human ALKBH3 demethylase activity by restriction endonuclease digestion assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of N-terminal His-tagged human ALKBH5 (66 to 292 residues)More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Ovis aries (Sheep))
Vanderbilt University School Of Medicine
Curated by ChEMBL
Vanderbilt University School Of Medicine
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of the ovine Prostaglandin G/H synthase 1 was determined by thin-layer chromatography assayMore data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 2(Mus musculus (Mouse))
Vanderbilt University School Of Medicine
Curated by ChEMBL
Vanderbilt University School Of Medicine
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibition of the murine wild type Prostaglandin G/H synthase 2More data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+5nMpH: 7.5 T: 2°CAssay Description:The DNA-binding competition assay was performed in 25 μL, in black 384-well plates, using either 30 mM HEPES (N-2-Hydroxyethylpiperazine-N'-2-Etha...More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 2(Mus musculus (Mouse))
Vanderbilt University School Of Medicine
Curated by ChEMBL
Vanderbilt University School Of Medicine
Curated by ChEMBL
Affinity DataIC50: 130nMT: 2°CAssay Description:Inhibition assays in triplicate were performed by preincubating enzyme (60-80 nM) and inhibitors (0-5 mM) for 20 min at 25°C followed by the add...More data for this Ligand-Target Pair
Affinity DataIC50: 1.74E+4nMAssay Description:Displacement of dm6A-containing ssDNA from truncated N-terminal his-tagged human FTO (31 residues) mediated demethylation activity expressed in Esche...More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 1(Ovis aries (Sheep))
Vanderbilt University School Of Medicine
Curated by ChEMBL
Vanderbilt University School Of Medicine
Curated by ChEMBL
Affinity DataIC50: 40nMpH: 8.0 T: 2°CAssay Description:For the time-dependent inhibition studies, COX enzyme was incubated with test compounds for 20 min and then analyzed for remaining COX activity by tr...More data for this Ligand-Target Pair
TargetProstaglandin G/H synthase 2(Homo sapiens (Human))
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
St. Bartholomew'S and The Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
Affinity DataIC50: 50nMT: 2°CAssay Description:For the time-dependent inhibition studies, COX enzyme was incubated with test compounds for 20 min and then analyzed for remaining COX activity by tr...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)