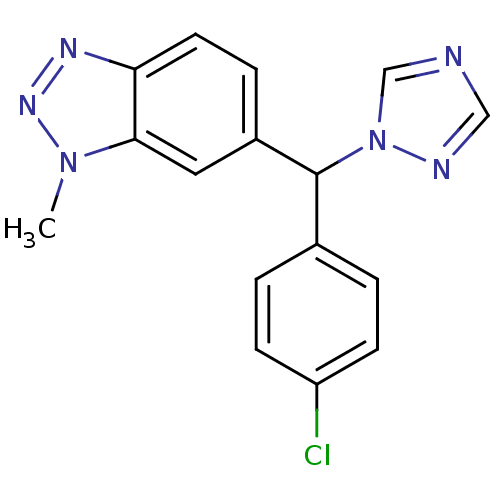
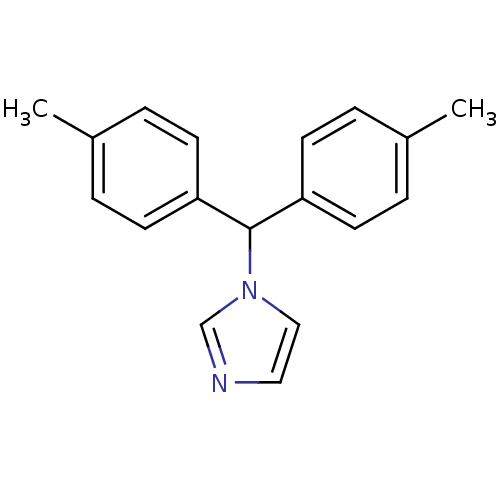
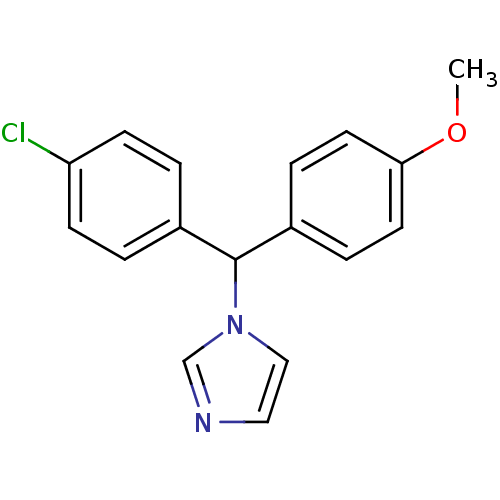
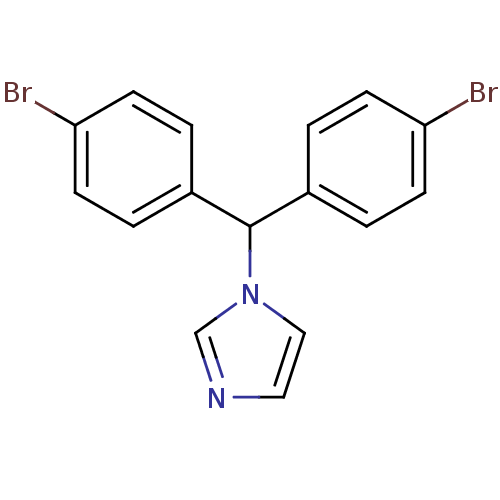
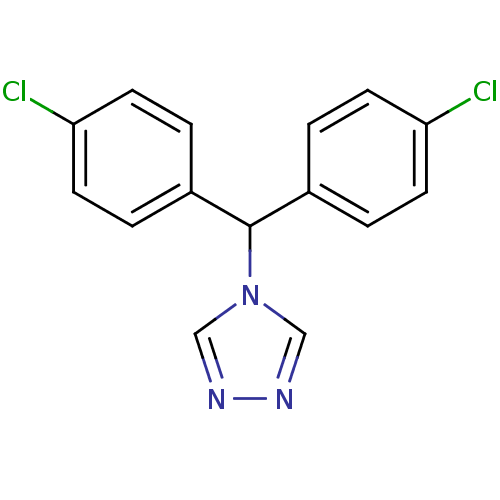
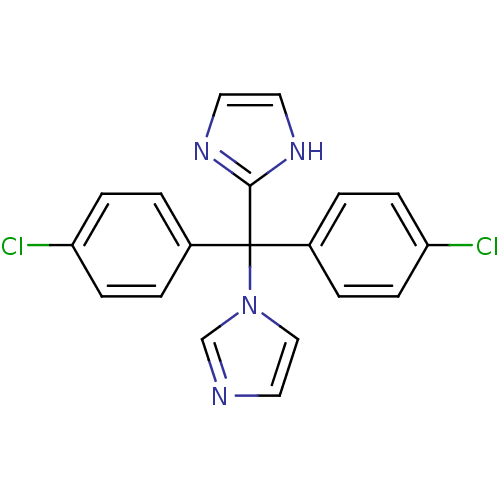
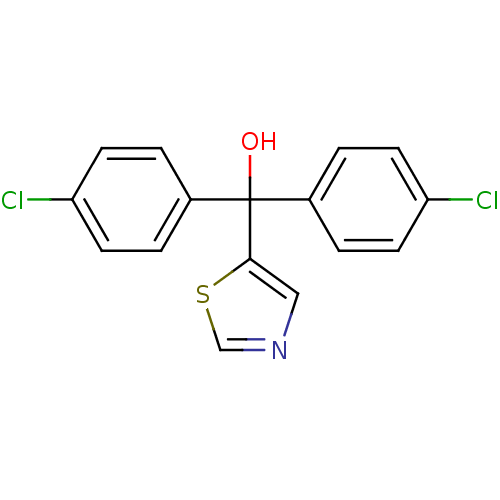
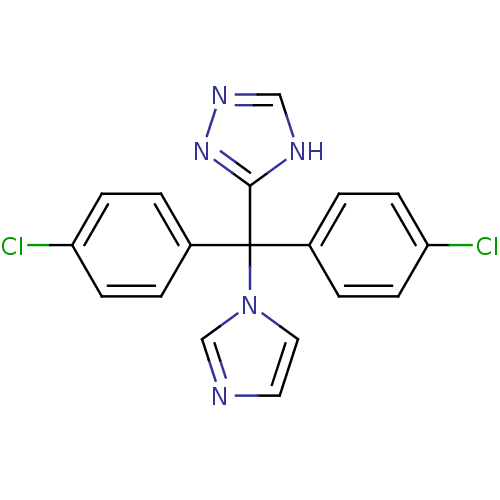
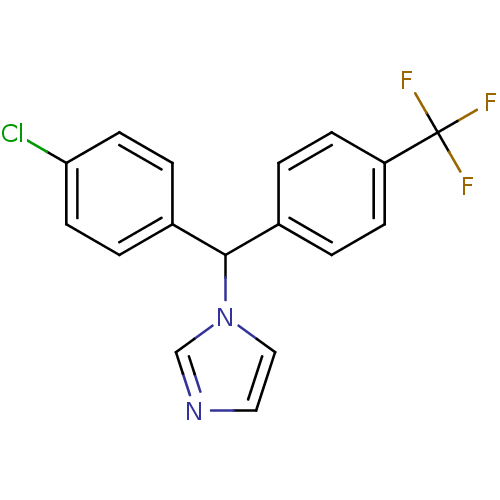
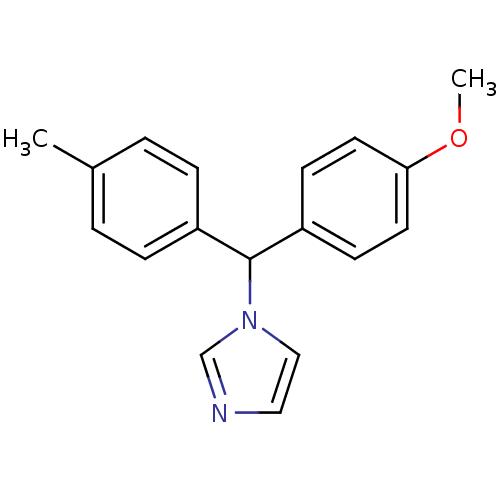
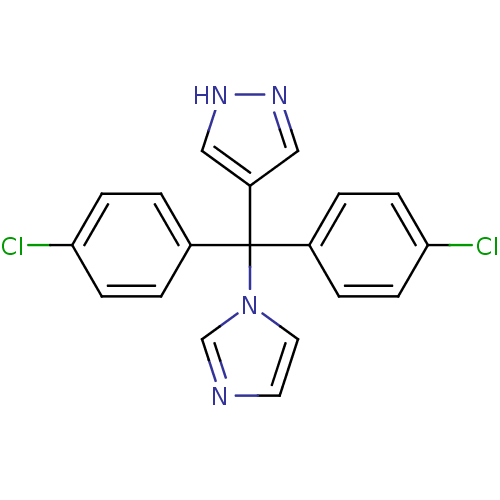
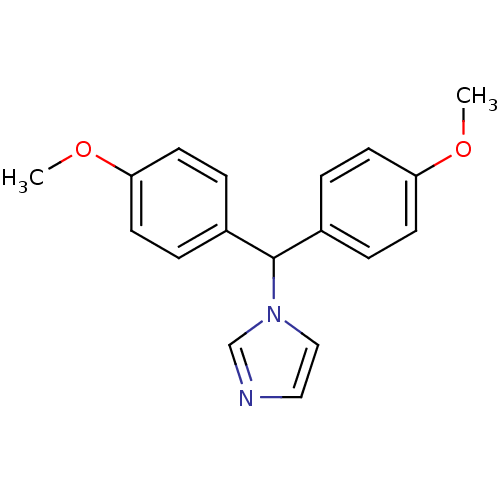
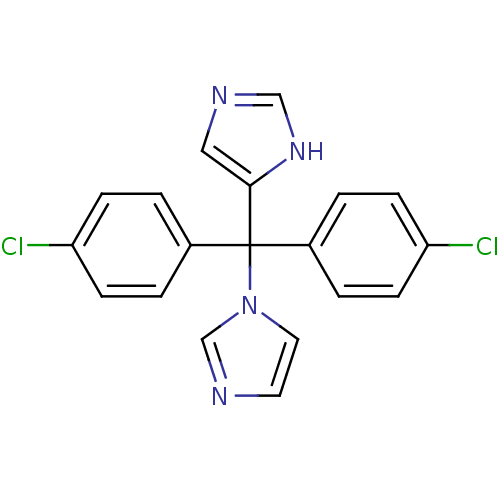
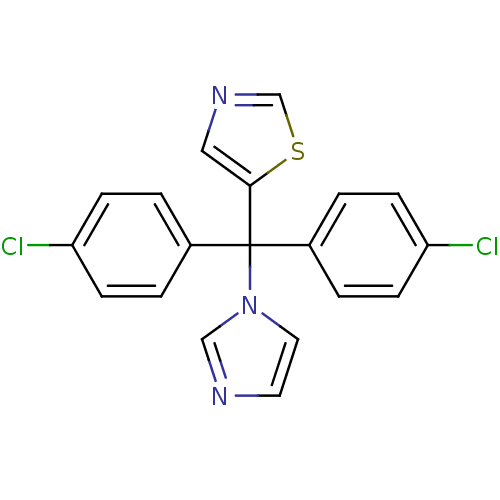
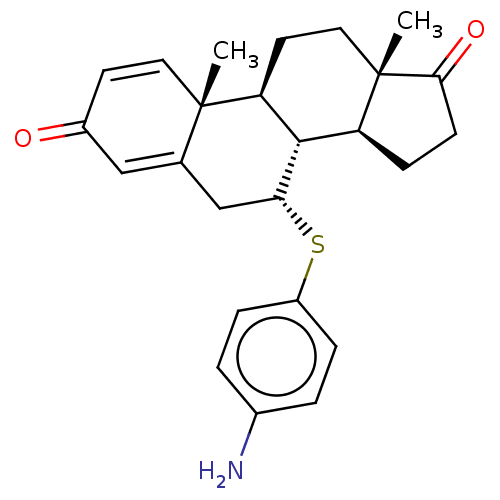
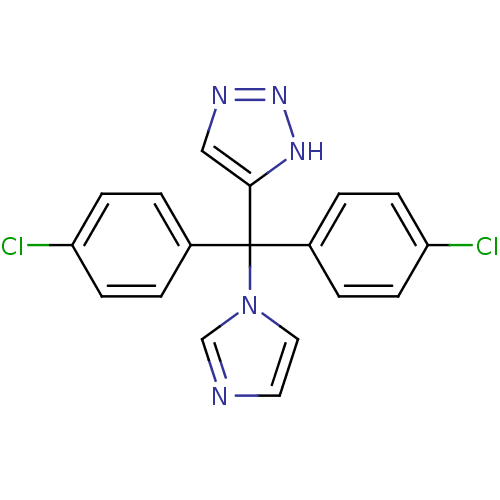
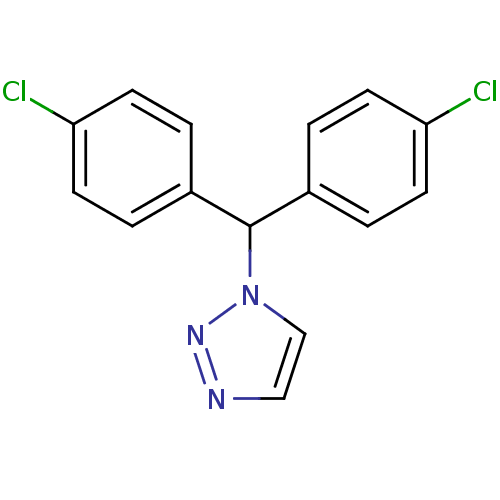
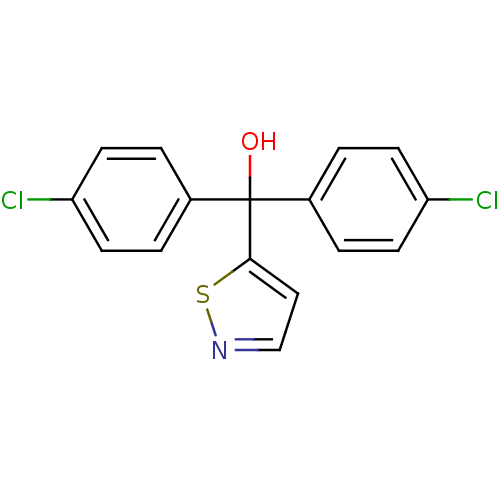
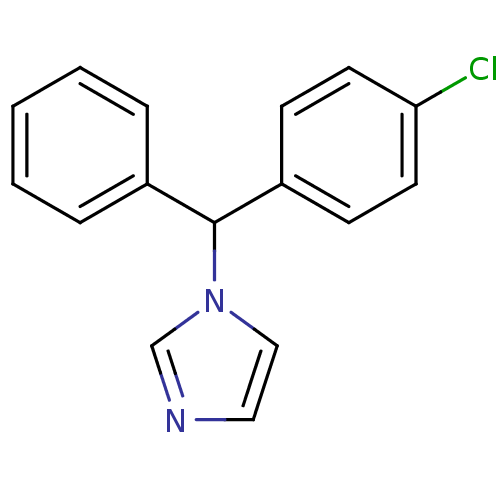
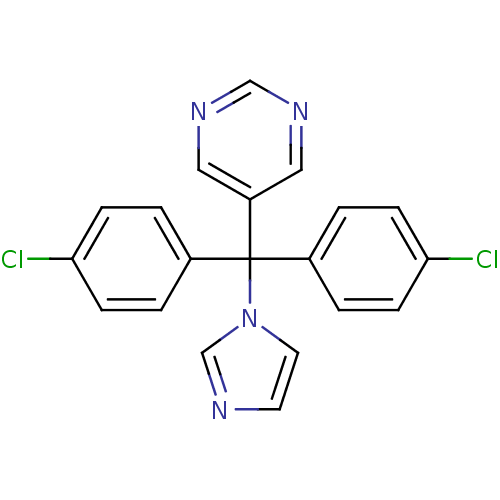
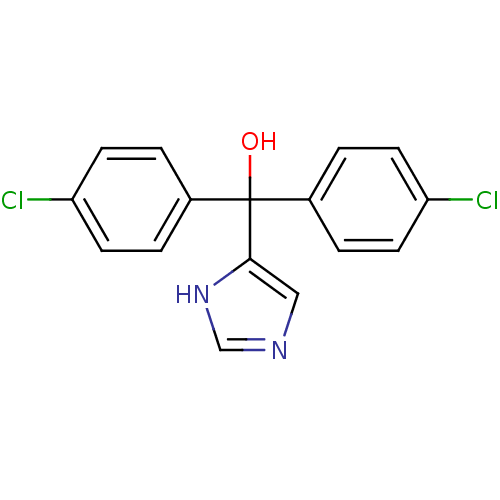
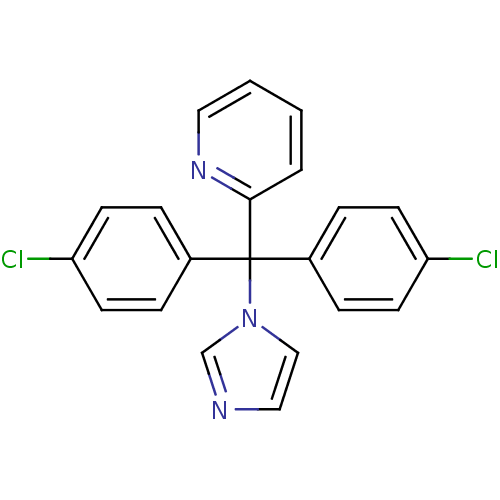
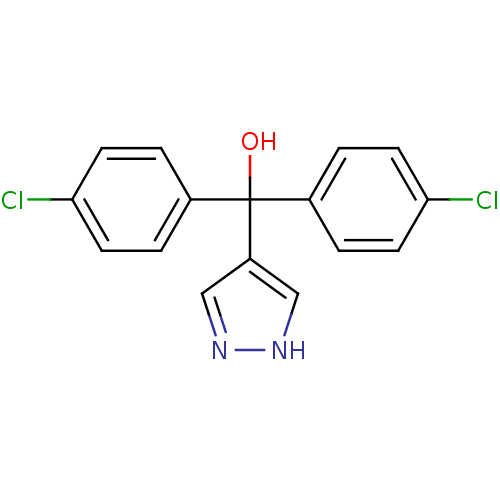
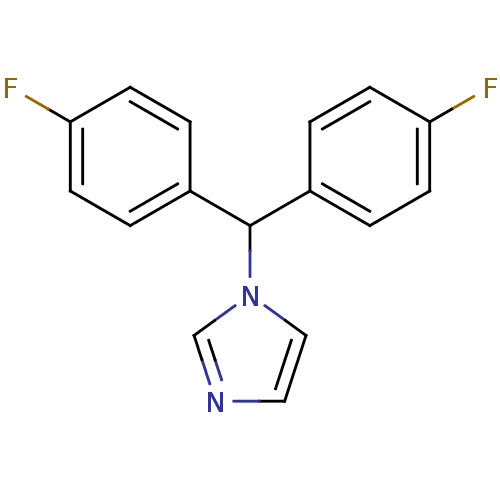
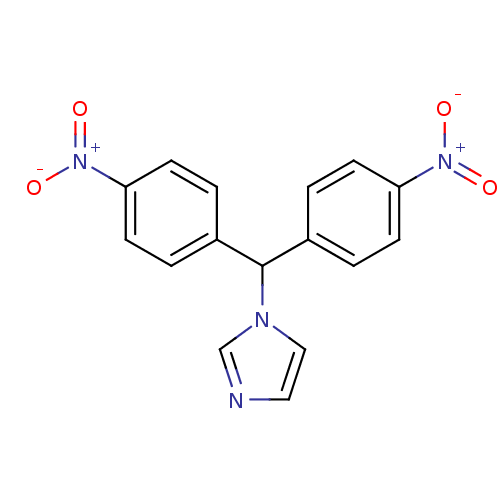
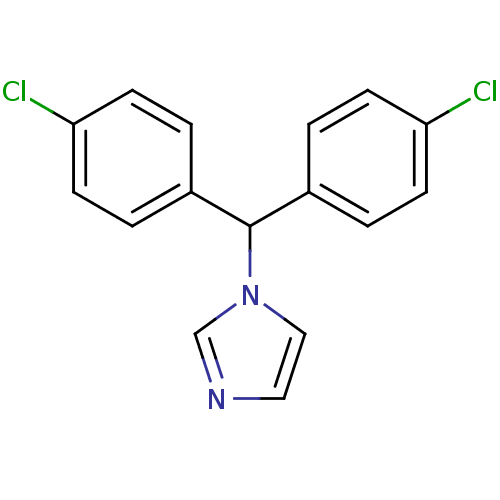
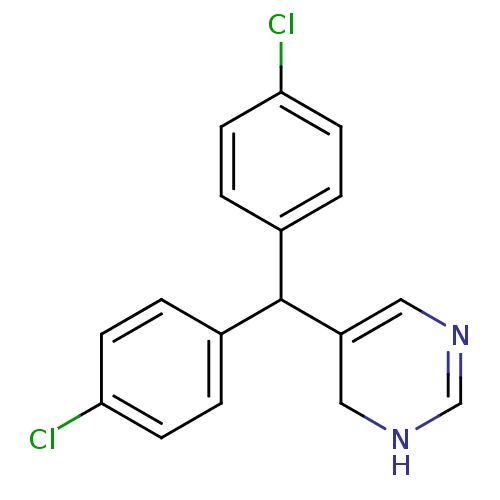
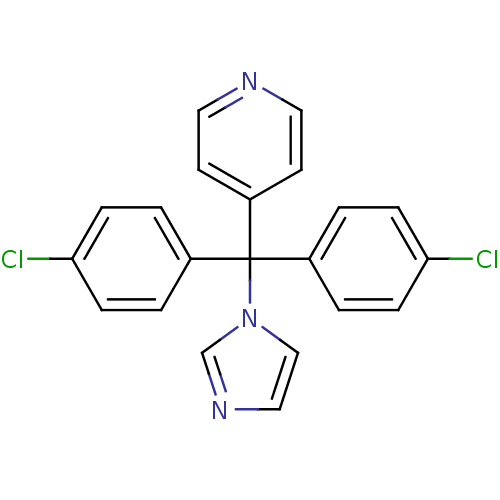
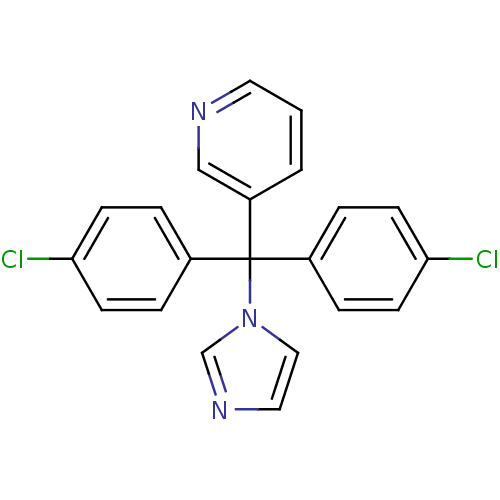
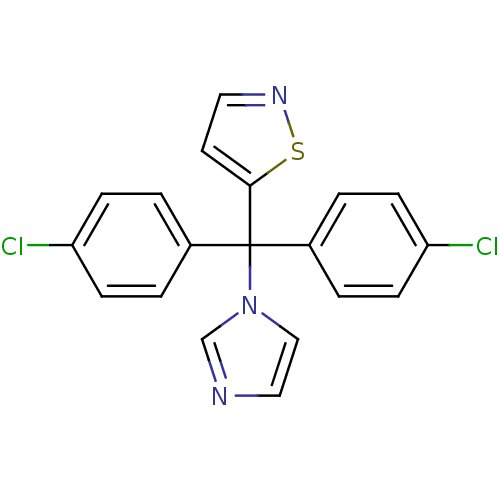
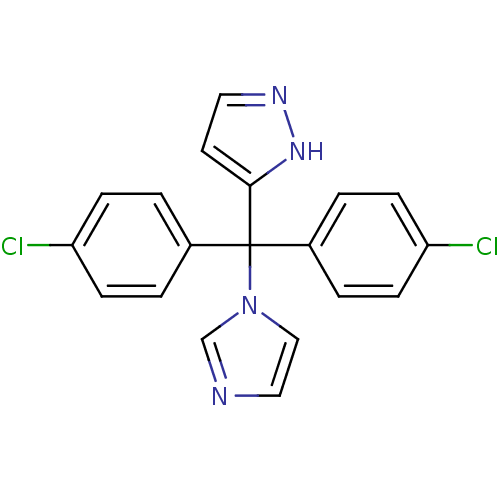
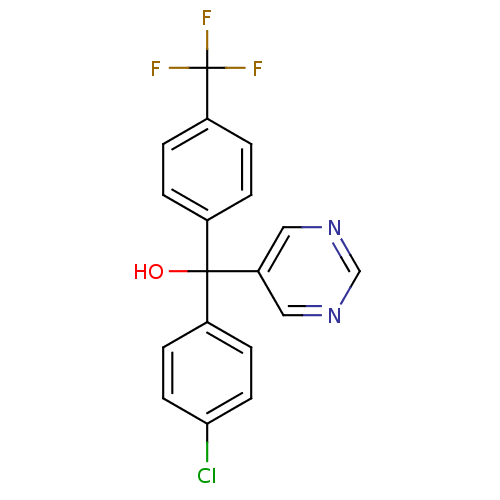
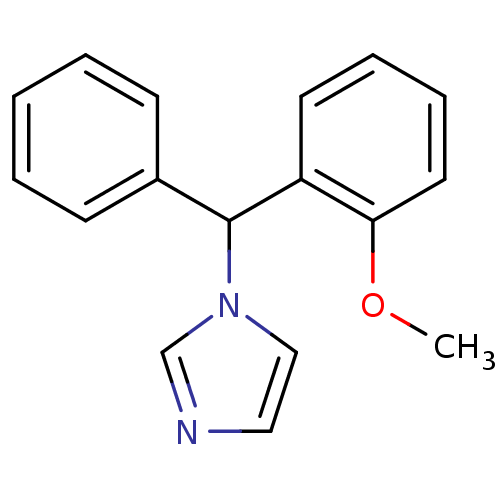
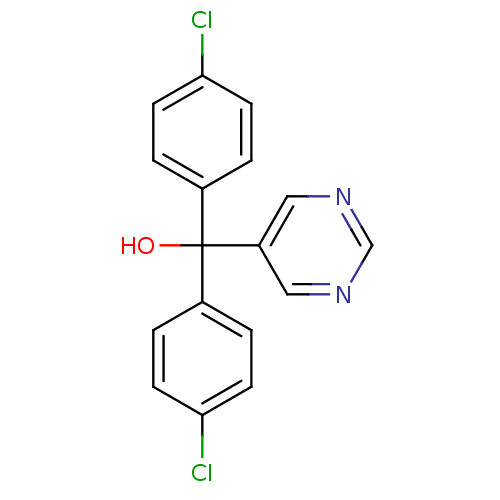
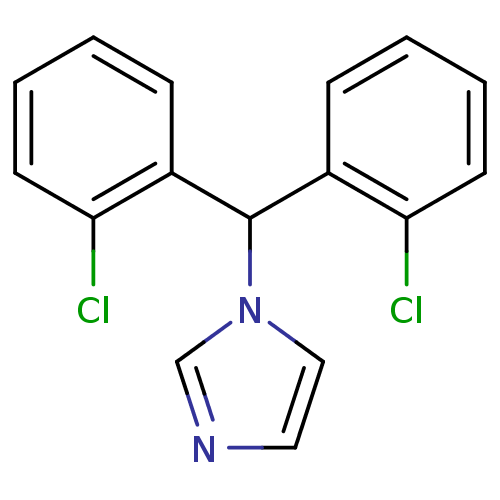
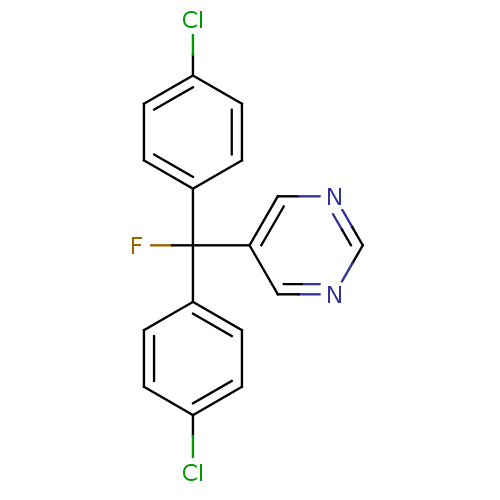
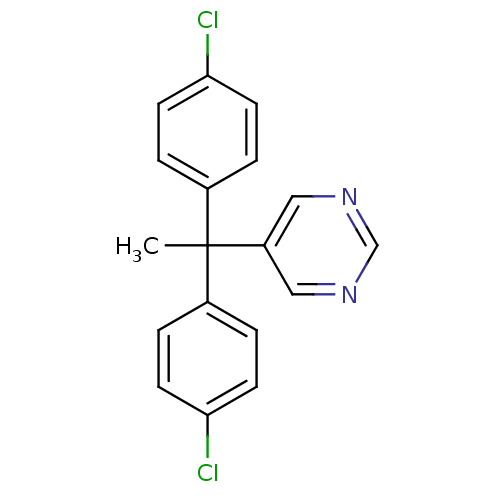
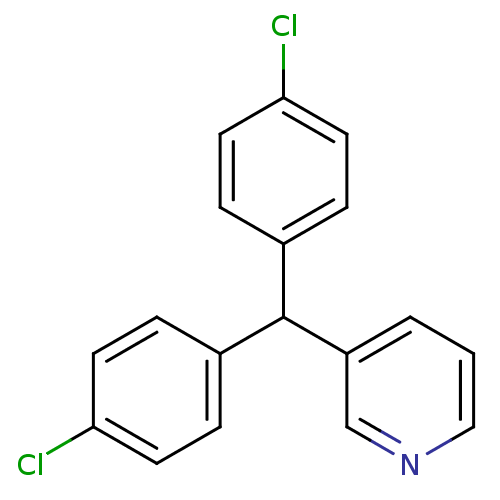
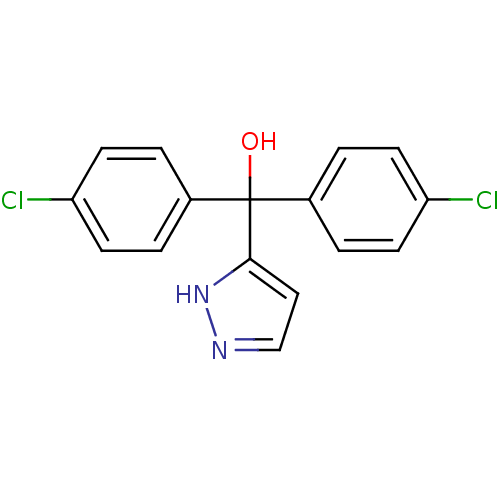
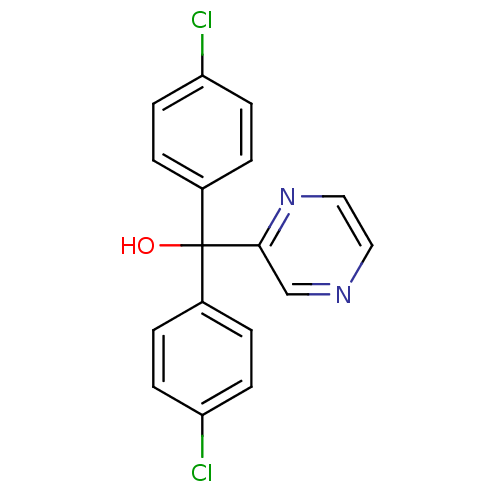
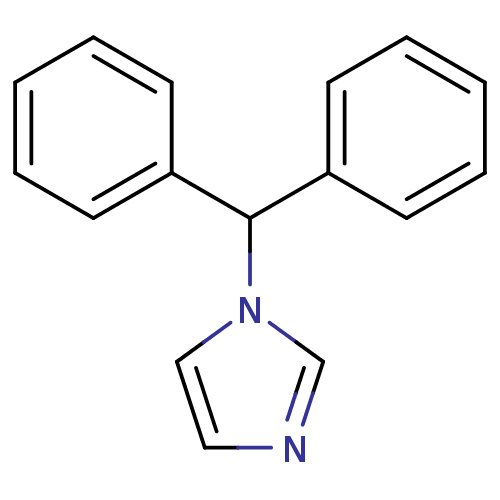
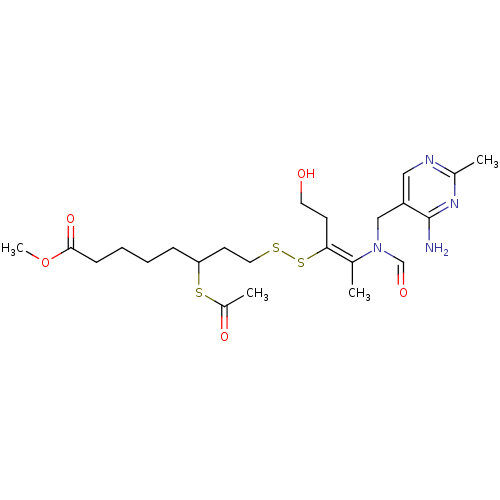
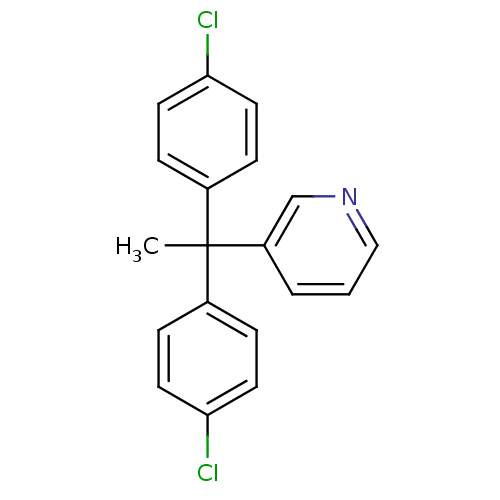
Affinity DataIC50: 3nMAssay Description:Binding affinity for Cytochrome P450 19A1More data for this Ligand-Target Pair
Affinity DataEC50: 6.40nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 8.40nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 8.80nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 8.90nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 9.30nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 9.40nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 9.60nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 13nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 16nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 17nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Inhibition of aromatase in rat mammary tumorMore data for this Ligand-Target Pair
Affinity DataEC50: 18nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 19nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 19nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 20nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataKi: 24nMAssay Description:Binding affinity for Cytochrome P450 19A1More data for this Ligand-Target Pair
Affinity DataEC50: 26nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 28nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataKi: 29nMAssay Description:Binding affinity for Cytochrome P450 19A1More data for this Ligand-Target Pair
Affinity DataEC50: 29nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 30nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 40nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 42nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 50nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 55nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 55nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 57nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 61nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 66nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 66nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 71nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 71nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 76nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 78nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 82nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 84nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 84nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 90nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 94nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 125nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair
Affinity DataEC50: 140nMAssay Description:In vitro inhibition of cytochrome P450 19A1 by rat ovarian microsomes incubated with [3H]androstenedione and NADPH-generating system.More data for this Ligand-Target Pair
Affinity DataEC50: 145nMAssay Description:In vitro Cytochrome P450 19A1 inhibition concentration to decrease aromatization of androstenedione in rat ovarian microsomeMore data for this Ligand-Target Pair