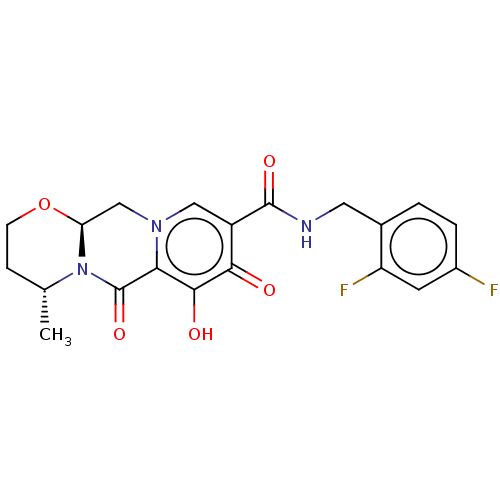
BDBM50062551 CHEBI:76010::Dolutegravir::GSK1349572::S-349572
SMILES [H][C@]12Cn3cc(C(=O)NCc4ccc(F)cc4F)c(=O)c(O)c3C(=O)N1[C@H](C)CCO2
InChI Key InChIKey=RHWKPHLQXYSBKR-BMIGLBTASA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 50 hits for monomerid = 50062551
Found 50 hits for monomerid = 50062551
Affinity DataKi: 18nMAssay Description:Displacement of [3H]-DTG from Sprague-Dawley rat liver Sigma 2 receptor incubated for 120 mins in presence of [3H]-(+)-pentazocine by liquid scintill...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Displacement of [3H]-di-o-tolylguanidine from sigma2 receptor (unknown origin) incubated for 1 hr in presence of (+)SKF10047 by liquid scintillation ...More data for this Ligand-Target Pair
Affinity DataKi: 30nMAssay Description:Displacement of [3H]DTG from sigma 2 receptor in Wistar Han rat liver membranes measured after 120 mins by scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 32nMAssay Description:Displacement of [3H]DTG from sigma2 receptor in rat liver membranes after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Cavia porcellus (Guinea pig))
University of Trieste
Curated by ChEMBL
University of Trieste
Curated by ChEMBL
Affinity DataKi: 71nMAssay Description:Displacement of (+)-[3H]pentazocine from guinea pig brain cortex sigma1 receptor by scintillation analyzerMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Cavia porcellus (Guinea pig))
University of Trieste
Curated by ChEMBL
University of Trieste
Curated by ChEMBL
Affinity DataKi: 124nMAssay Description:Displacement of [3H]-(+)-pentazocine from Dunkin-Hartley guinea pig brain cortex Sigma 1 receptor incubated for 150 mins by liquid scintillation coun...More data for this Ligand-Target Pair
Affinity DataKi: 217nMAssay Description:Displacement of (+)-[3H]pentazocine from sigma1 receptor in rat brain membranes after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.52E+3nMAssay Description:Displacement of (-)-[3H]vesamicol from rat VAChT expressed in rat PC12 cell membranes after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate preincubated for 20 mins with NADPH prior to initia...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2C8 in human liver microsomes using rosiglitazone as substrate preincubated for 20 mins with NADPH prior to ini...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2B6 in human liver microsomes using bupropion as substrate preincubated for 20 mins with NADPH prior to initiat...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2A6 in human liver microsomes using coumarin as substrate preincubated for 20 mins with NADPH prior to initiati...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate preincubated for 20 mins with NADPH prior to initia...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using nifedipine as substrate preincubated for 20 mins with substrate prior to initiation of reaction ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.40E+4nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using atorvastatin as substrate preincubated for 20 mins with substrate prior to initiation of reactio...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2D6 in human liver microsomes using bufuralol as substrate preincubated for 20 mins with substrate prior to initiation of reaction w...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2C19 in human liver microsomes using S-mephenytoin as substrate preincubated for 20 mins with substrate prior to initiation of react...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate preincubated for 20 mins with substrate prior to initiation of reaction ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2C8 in human liver microsomes using rosiglitazone as substrate preincubated for 20 mins with substrate prior to initiation of reacti...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2B6 in human liver microsomes using bupropion as substrate preincubated for 20 mins with substrate prior to initiation of reaction w...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2A6 in human liver microsomes using coumarin as substrate preincubated for 20 mins with substrate prior to initiation of reaction wi...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate preincubated for 20 mins with substrate prior to initiation of reaction ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of MDR1 (unknown origin) expressed in MDCK2 cells assessed as basolateral to apical transport of [3H]digoxin after 90 mins by liquid scint...More data for this Ligand-Target Pair
TargetBroad substrate specificity ATP-binding cassette transporter ABCG2(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: >3.00E+4nMAssay Description:Inhibition of BCRP (unknown origin) expressed in MDCK2 cells assessed as basolateral to apical transport of [14C]cimetidine after 90 mins by liquid s...More data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 2(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human MRP2 expressed in baculovirus-infected Sf9 cell membrane vesicle using [3H]estradiol 17beta-D-glucuronide as substrate preincubat...More data for this Ligand-Target Pair
TargetSolute carrier organic anion transporter family member 1B1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human OATP1B1 expressed in CHO cells using [3H]estradiol 17beta-D-glucuronide as substrate by liquid scintillation counting analysisMore data for this Ligand-Target Pair
TargetSolute carrier organic anion transporter family member 1B3(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human OATP1B3 expressed in BacMam baculovirus virus infected HEK MSR2 cells using [3H]estradiol 17beta-D-glucuronide as substrate by li...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of human OCT2 expressed in MDCK2 cells using [14C]metformin as substrate by liquid scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Inhibition of HIV integrase strand transfer activity using 5'-biotin/3'-Cy5-labeled DNA substrate preincubated for 5 mins followed by substrate addit...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus 1)
University of Minnesota
Curated by ChEMBL
University of Minnesota
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of HIV1 reverse transcriptase RNaseH activity using 3'-fluorescein/5'-Dabcyl labeled HTS-1 substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 2.70nMAssay Description:Inhibition of HIV-1 integrase strand transfer activity using [3H]-DNA as substrate preincubated for 60 mins prior to substrate addition measured afte...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Inhibition of HIV integrase strand transfer activityMore data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus 1)
University of Minnesota
Curated by ChEMBL
University of Minnesota
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of HIV1 reverse transcriptase RNase H expressed in Escherichia coli JM109 using RNA/DNA duplex substrate HTS-1More data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Inhibition of HIV1 integrase strand transfer activity using 5'-biotinylated oligonucleotide as substrate preincubated for 10 mins followed by substra...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus 1)
University of Minnesota
Curated by ChEMBL
University of Minnesota
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of RNase H activity of full length HIV1 reverse transcriptase using RNA-DNA duplex HTS-1 substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Inhibition of HIV1 recombinant integrase expressed in Escherichia coli using [32P]-labeled oligonucleotide as substrate after 60 mins by strand trans...More data for this Ligand-Target Pair
Affinity DataEC50: 3.60nMAssay Description:Inhibition of HIV-1 NL4-3 integrase E92Q/N155H double mutantMore data for this Ligand-Target Pair
Affinity DataEC50: 2.30nMAssay Description:Inhibition of wild type HIV-2 ROD9 integraseMore data for this Ligand-Target Pair
Affinity DataEC50: 1.5nMAssay Description:Inhibition of HIV-1 NL4-3 integrase T97A/Y143C double mutantMore data for this Ligand-Target Pair
Affinity DataEC50: 6.80nMAssay Description:Inhibition of HIV-1 NL4-3 integrase G140S/Q148R double mutantMore data for this Ligand-Target Pair
Affinity DataEC50: 1.5nMAssay Description:Inhibition of wild type HIV-1 NL4-3 integraseMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2C19 in human liver microsomes using S-mephenytoin as substrate preincubated for 20 mins with NADPH prior to in...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Metabolism-dependent inhibition of CYP2D6 in human liver microsomes using bufuralol as substrate preincubated for 20 mins with NADPH prior to initiat...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Metabolism-dependent inhibition of CYP3A4 in human liver microsomes using atorvastatin as substrate preincubated for 20 mins with NADPH prior to init...More data for this Ligand-Target Pair
Affinity DataIC50: 6.50E+4nMAssay Description:Metabolism-dependent inhibition of CYP3A4 in human liver microsomes using nifedipine as substrate preincubated for 20 mins with NADPH prior to initia...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human recombinant UGT1A1 expressed in supersomes assessed as scopoletin glucuronidation by fluorescence analysisMore data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+4nMAssay Description:Inhibition of human MDR1More data for this Ligand-Target Pair
TargetATP-binding cassette sub-family C member 2(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: >9.00E+4nMAssay Description:Inhibition of human MRP2More data for this Ligand-Target Pair
Affinity DataIC50: 0.680nMAssay Description:Inhibition of HIV-1 integrase Q148K mutantMore data for this Ligand-Target Pair
TargetBroad substrate specificity ATP-binding cassette transporter ABCG2(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 6.70E+4nMAssay Description:Inhibition of human BCRPMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)