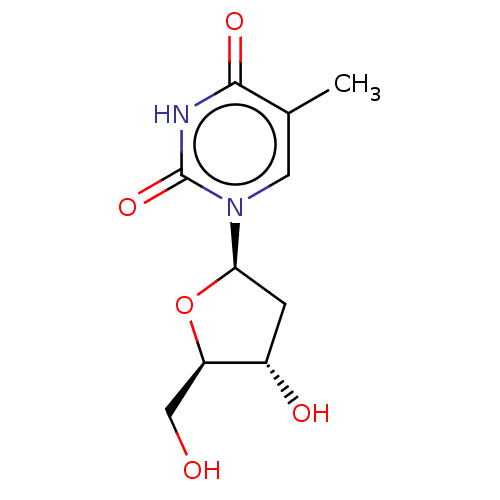
BDBM1 dT::thymidine
SMILES CC1=CN(C(=O)NC1=O)[C@H]2C[C@@H]([C@H](O2)CO)O
InChI Key InChIKey=IQFYYKKMVGJFEH-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 21 hits for monomerid = 1
Found 21 hits for monomerid = 1
Affinity DataIC50: 16nMAssay Description:Inhibitory activity (50 uM) against HSV-1 Thymidine kinase in OST-TK-/HSV-1 TK+ cell line in combination with BVAraUMore data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Inhibitory activity (50 uM) against HSV-1 Thymidine kinase in OST-TK-/HSV-1 TK+ cell line in combination with gancicclovirMore data for this Ligand-Target Pair
Affinity DataKi: 200nMAssay Description:Binding affinity constant against HSV-1 thymidine kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Binding affinity towards HSV-1 thymidine kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibitory concentration against HSV-1 TK (WT) catalyzed [3H]-GCV phosphorylationMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Inhibition of HSV-1 thymidine kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: 4.10E+3nMAssay Description:Inhibition of Herpes B virus recombinant thymidine kinase-mediated [3H]TdR phosphorylationMore data for this Ligand-Target Pair
TargetTransmembrane and immunoglobulin domain-containing 3(Rat)
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
TargetAdenosine receptor A1(Rat)
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
TargetAdenosine receptor A2a(Rat)
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
National Institute of Diabetes, Digestive and Kidney Diseases
Curated by PDSP Ki Database
Affinity DataKi: 1.54E+4nM IC50: 2.10E+4nMAssay Description:Ketotifen, dacarbazine, thiocolchicoside, meloxicam, methotrexate, furosemide, olanzapine, methylprednizolone acetate, paricalcitol, ritodrine hydroc...More data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:Inhibition of Mycobacterium tuberculosis recombinant TMPKMore data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:Inhibitory activity against thymidine monophosphate kinase (TMPK) in Mycobacterium tuberculosisMore data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:Binding affinity towards mycobacterium tuberculosis thymidine monophosphate kinaseMore data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:Inhibition of Mycobacterium tuberculosis TMPK by coupled spectrophotometric assayMore data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:Inhibitory activity against thymidine monophosphate kinase (TMPK) in Mycobacterium tuberculosisMore data for this Ligand-Target Pair
TargetThymidylate kinase(Mycobacterium tuberculosis (strain ATCC 25618 / H3...)
Cnrs
Curated by ChEMBL
Cnrs
Curated by ChEMBL
Affinity DataKi: 2.70E+4nMAssay Description:In Vitro inhibition of Thymidine Monophosphatase Kinase of Mycobacterium tuberculosis (TMPKm)More data for this Ligand-Target Pair
Affinity DataKi: 1.80E+5nMAssay Description:Inhibition of human TMPKMore data for this Ligand-Target Pair
Affinity DataKi: 1.80E+5nMAssay Description:Inhibition of human TMPK by coupled spectrophotometric assayMore data for this Ligand-Target Pair
Affinity DataKi: 2.57E+5nMAssay Description:Inhibition of Staphylococcus aureus CCM 885 recombinant thymidine kinaseMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+5nMAssay Description:Inhibitory concentration against HSV-1 TK (A167Y) catalyzed [3H]-GCV phosphorylationMore data for this Ligand-Target Pair
Activity Spreadsheet -- ITC Data from BindingDB
 Found 4 hits for monomerid = 1
Found 4 hits for monomerid = 1
ITC DataΔG°: -7.22kcal/mole −TΔS°: 12.0kcal/mole ΔH°: -19.1kcal/mole logk: 1.90E+5
pH: 7.5 T: 25.00°C
pH: 7.5 T: 25.00°C
ITC DataΔG°: -7.12kcal/mole −TΔS°: 10.2kcal/mole ΔH°: -17.5kcal/mole
pH: 7.5 T: 20.00°C
pH: 7.5 T: 20.00°C
ITC DataΔG°: -7.02kcal/mole −TΔS°: 8.81kcal/mole ΔH°: -15.8kcal/mole
pH: 7.5 T: 15.00°C
pH: 7.5 T: 15.00°C
ITC DataΔG°: -7.12kcal/mole −TΔS°: 6.49kcal/mole ΔH°: -13.6kcal/mole
pH: 7.5 T: 10.00°C
pH: 7.5 T: 10.00°C
 3D Structure (crystal)
3D Structure (crystal)