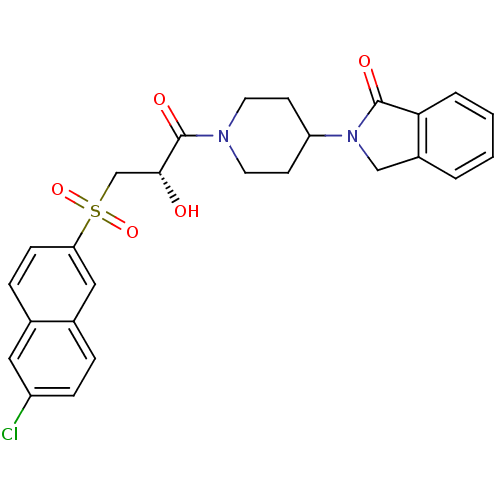
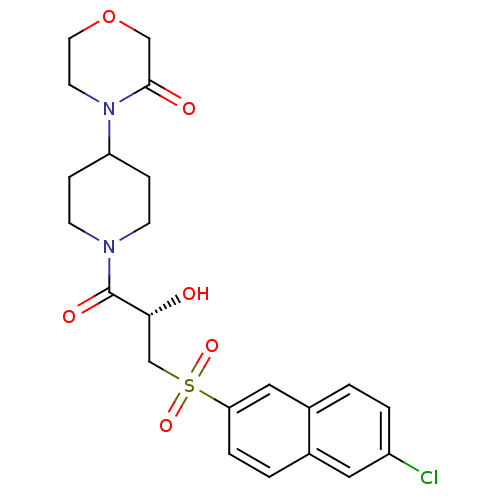
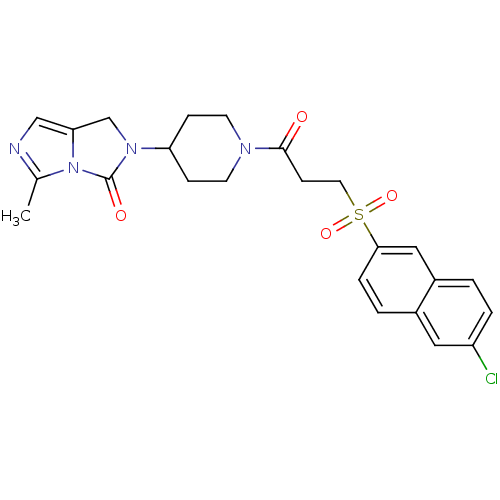
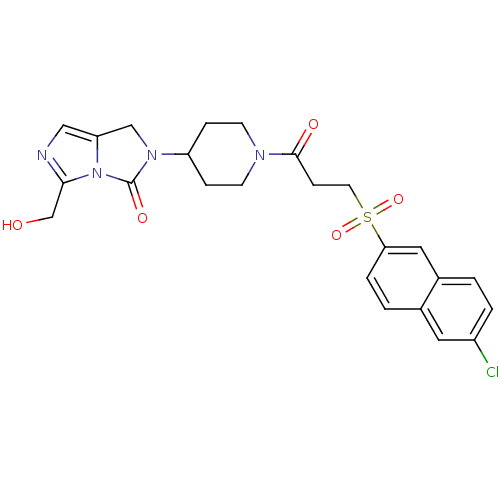
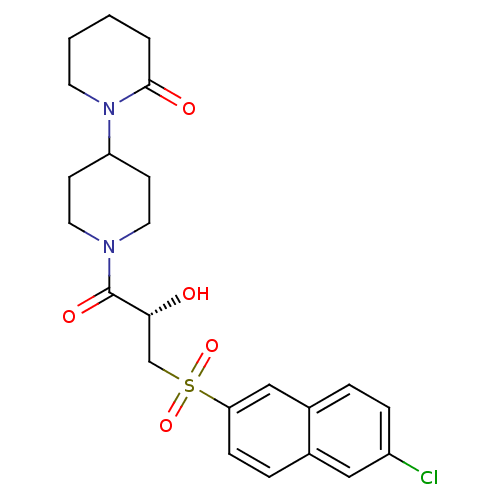
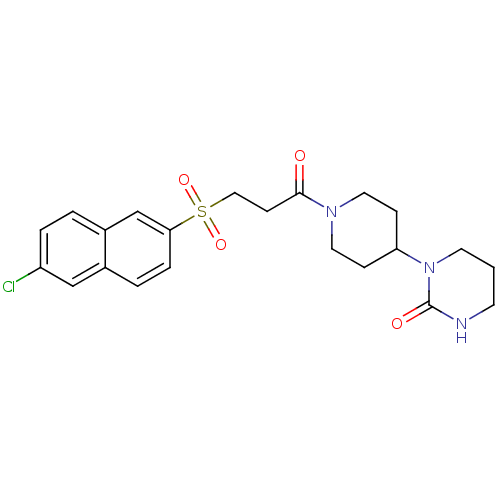
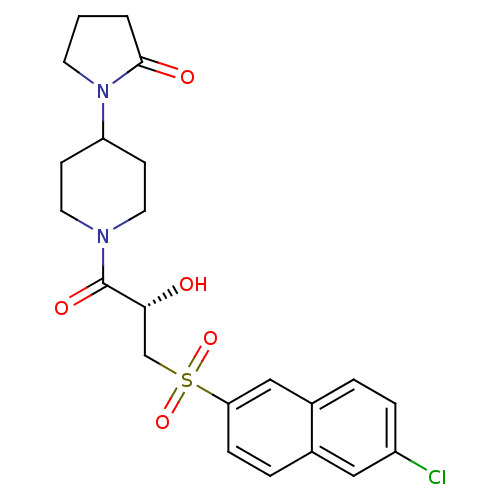
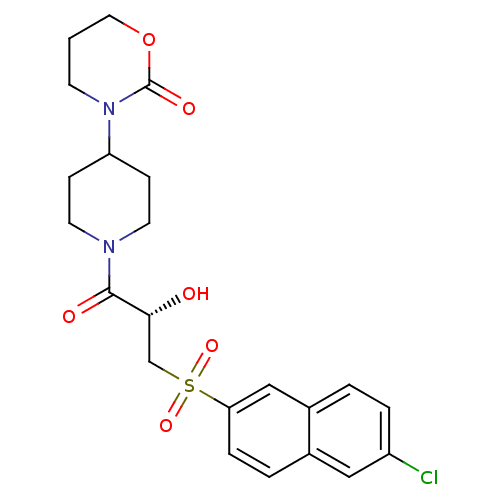
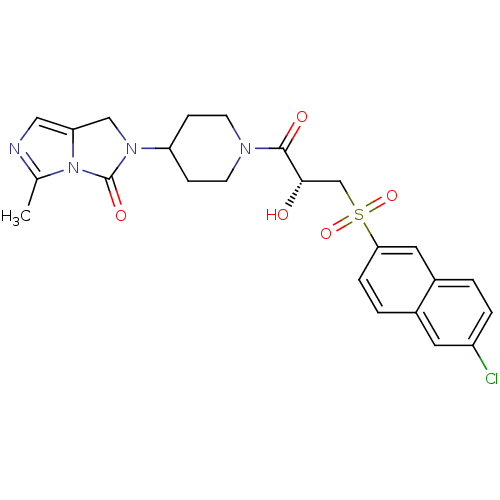
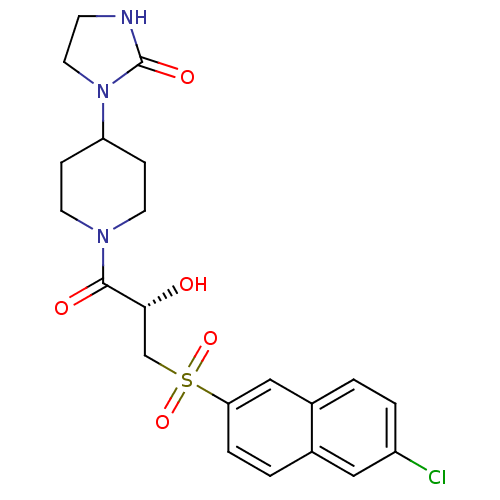
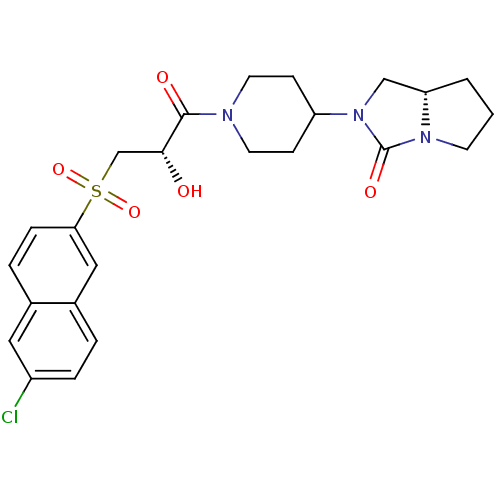
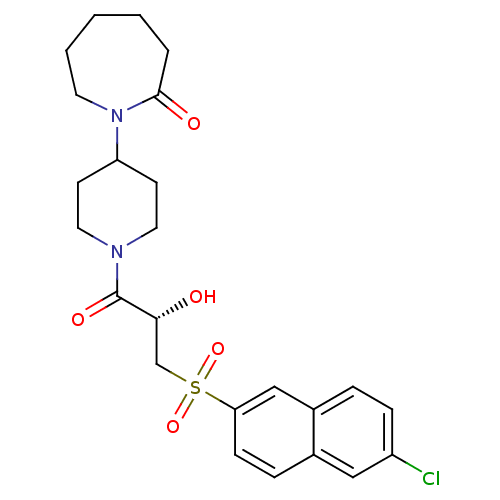
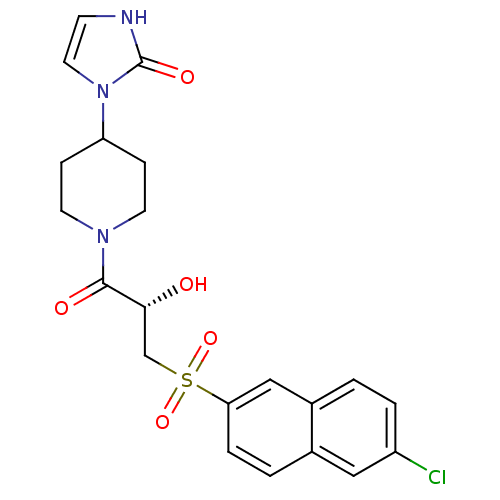
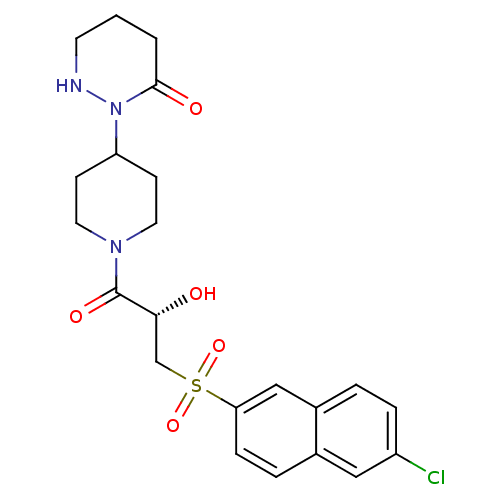
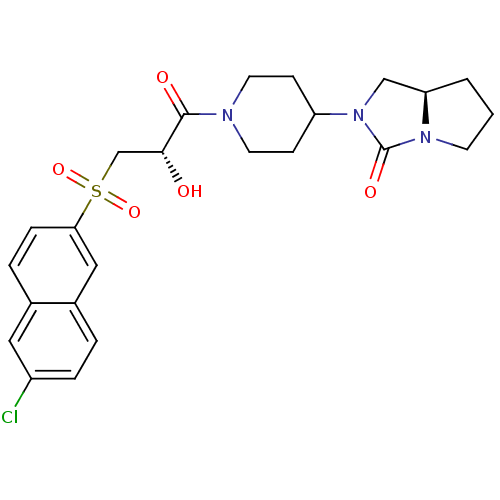
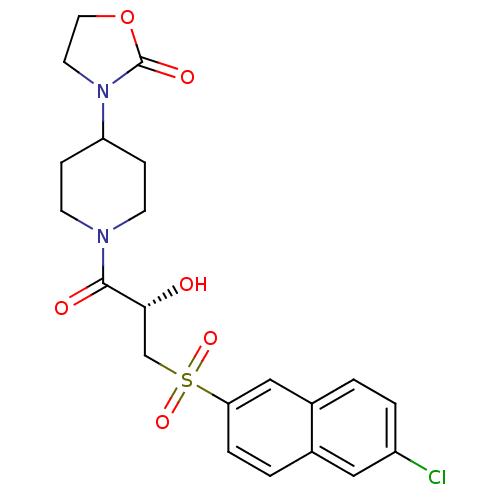
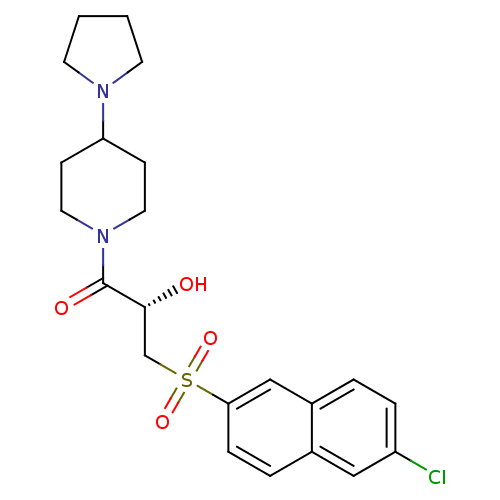
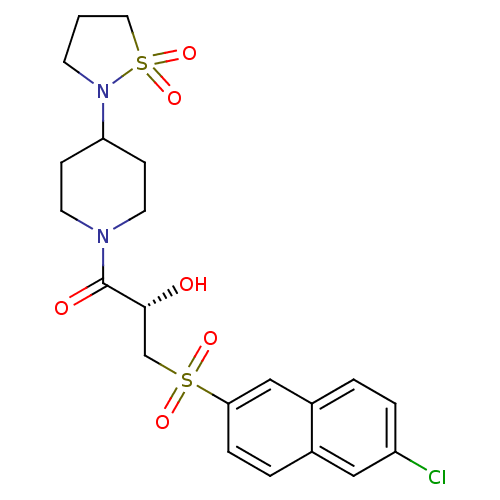
Report error Found 26 Enz. Inhib. hit(s) with all data for entry = 50031677
Affinity DataIC50: 1.70nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Inhibition of human factor 10a using S2222 as substrate after 10 mins by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.5nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.80nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 9.10nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 46nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 51nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 147nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 198nMAssay Description:Inhibition of human factor 10a assessed as p-nitroanilide release using S2765 as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibition of human thrombin after 10 mins by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.50E+3nMAssay Description:Inhibition of human factor 9a after 10 mins by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.40E+4nMAssay Description:Inhibition of human tissue plasminogen activator after 5 mins by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+4nMAssay Description:Inhibition of human plasmin after 5 mins by chromogenic assayMore data for this Ligand-Target Pair
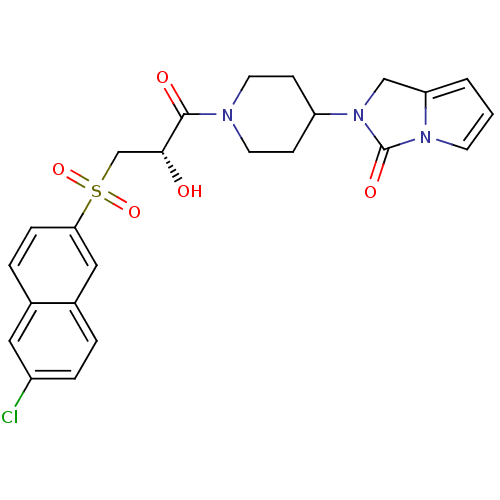
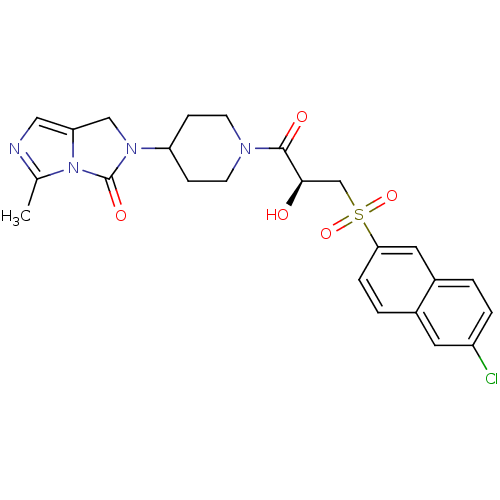

 3D Structure (crystal)
3D Structure (crystal)